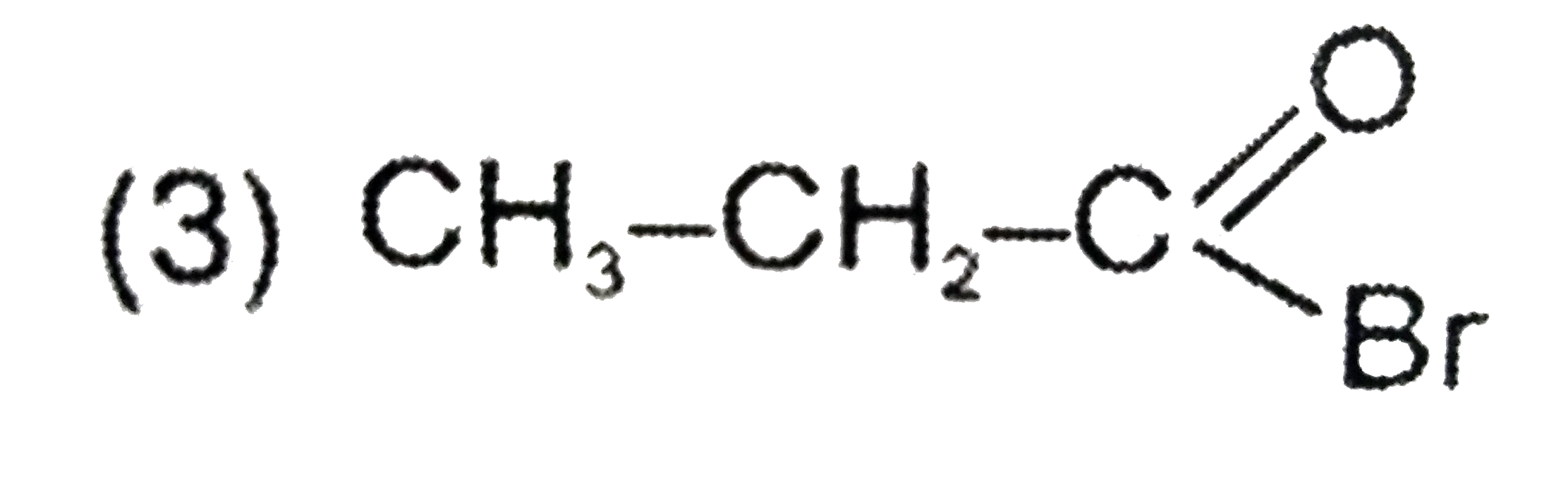Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
State and explain Kohlrausch's law ofindependent migration of ions. |
|
Answer» Solution :Statement of Kohlrausch's LAW: This statesthat of INFINITE dilutionof the solution,each ionof an electrolytemigratesindependentlyof its co-ionsand contributesindependentlyto thetotalmolarconductivityof the electrolyte, irrespectiveof the natureof otherions presentin te solution . Explanation :Both theions, cationand anionof the electrolytemake a definitecontributionto the molar conductivity ofthe electrolyte at infinite dilution or ZERO concentration`(wedge_(0))`. Of `lambda_(+)^(0)` and `lambda_(-)^(0)`are the molarconductivities of cation and nation respectively at infinite dilution, then `wedge_(0) = lambda_(+)^(0) + lambda_(-)^(0)` . This is known as Kohlrausch's law of independentmigrationof ions. `wedge_(0KCl) = lambda_(K^(+))^(0) + lambda_(Cl^(-))^(0)` and `wedge_(0NaCl) = lambda_(Na^(+))^(0) + lambda_(Cl^(-))^(0)` `:. wedge_(0KCl) - wedge_(0NaCl)` `= (lambda_(K^(+))^(0) + lambda_(Cl^(-))^(0)) - (lambda_(Na^(+))^(0) + lambda_(Cl^(-))^(0)) = lambda_(K^(+))^(0) - lambda_(Na^(+))^(0) + lambda_(I^(-))^(0)` `:. wedge_(0KI) - wedge_(0NAl) = (lambda_(K^(+))^(0) + lambda_(I^(-))^(0)) - (lambda_(Na^(+))^(0) + lambda_(I^(-))^(0))` `= lambda_(K^(+))^(0) - lambda_(Na^(+))^(0) = 150.3- 126.9 = 23.4` Thus the difference in `wedge_(0)` valuesfor `K^(+)` and `Na^(+)` saltsis constant and is independentof the nature of other ANIONS. This provesthe validity of Kohlrausch's law. |
|
| 2. |
State and explain Freundlich adsorption isotherm. |
| Answer» | |
| 3. |
State and explainFaraday's secondlaw of electrolysis . |
Answer» Solution :Explanation :  (1) Faraday's second law of ELECTROLYSIS : It states thatwhen the same quantity of electricityis passedthroughdifferentelectrolyticcells containingdifferentelectrolytes,arrangedin series,then the amounts of the substancesproducedby oxidationor reductionat therespectiveelectrodesare directly proportional to theirrespectivechemical equivalent masses. (2) Mathematical EXPRESSION : If `W_(1)` and `W_(2)` are themasses of two substances A and B havingmolar masses`M_(1)` and `M_(2)` respectively. Number of moles of `A = W_(1)//M_(1)` Number of moles of `B = W_(2)//M_(2)` Now from half cell reactions at two ELECTRODES, Moles of A produced = Moles of electrons passed `xx` mole RATIO of A in half reaction Moles of B produced = Moles of electrons passed `xx` molesratio of B in half reaction Hence Faraday's second law can be mathematicallyrepresentedas. `("moles of A produced")/("moles of B produced") = ("moles ratio of A halfreaction")/("moles ratio of B half reaction")` |
|
| 4. |
State Faraday's first law of electrolysis. |
|
Answer» |
|
| 5. |
State and derive Ostwaid dilution law for a weak electrolyte. |
|
Answer» SOLUTION :At constant temperature the degree of dissociation of weak electrolyte PROCESS with dilution in such a way that the value of the `(alpah^2 c)/(1- alpha)` remains constant. LET US CONSIDER weak acid `HA leftrightarrow H^(+)+ A^-` c 0 0 `c-a c ca ca ` Initial concentration Equilibrium concentration Applying law of mass action `K_a=([H^+][A^-])/([HA])` `K_a=(ca times ca)/(c (1-a))=(c a^2)/(1-a)` |
|
| 6. |
State an example and function of the following: wide spectrum antibiotics. |
| Answer» Solution :Chloramphenicol is a WIDE spectrum antibiotic. It is used to treat acute fever, typhoid, dysentry, urinary INFECTIONS, MENINGITIS and pneumonia. VANCOMYCIN and OFLOXACIN are other broad spectrum antibiotics. | |
| 7. |
State an example and function of the following : wide spectrum antibiotics. |
| Answer» Solution :Chloramphenicol is a wide spectrum antibiotic. It is used for the treatment of TYPHOID, dysentery, meningitis and acute FEVER. It is effective against a NUMBER of MICROORGANISMS. | |
| 8. |
Why the use of Aspartame is limited to cold foods and soft drinks? |
| Answer» SOLUTION :It is UNSTABLE at HIGH TEMPRATURE. | |
| 9. |
State a consequence of lanthanoid contraction shown by transition elements. |
| Answer» SOLUTION :The PROPERTIES of 4D and 5d-transition ELEMENTS rasemble CLOSELY. | |
| 10. |
Starting with toluene, outline a synthesis of (a) 1-bromo- 2-trichloromethylbenzene,(b) 1-bromo-3-trichloromethylbenzene, and (c) 1-bromo-4-trichloromethylbenzene. |
Answer» Solution : Compounds (a) and (c) can be obtained by ring BROMINATION of toluene followed by benzylic radical CHLORINATION of the side CHAIN USING three molar equivalents of CHLORINE:
|
|
| 11. |
State a condition under which a bimolecular reaction is kinetically first order reaction. |
| Answer» SOLUTION :A bimolecular reaction BECOMES first order reaction when one of the reactants is in EXCESS. | |
| 12. |
Starting with three different amino acid molecules, how many different tripeptide molecules are formed |
|
Answer» 12 |
|
| 13. |
Starting with one mole of a compound X it is found that the reaction is 3/4 complete in 1 min . What is the rate constant if the reaction follows first order ? |
|
Answer» `0.213 SEC^(-1)` `:. K = (2.303)/(60 sec)"log" 4 = (2.303xx0.602)/(60)` `=0.0231 sec^(-1)` |
|
| 14. |
Starting with alcohol, outline a synthesis of cyclohexyl chloride |
Answer» SOLUTION :
|
|
| 15. |
Starting with alcohol, outline a synthesis ofbutyl bromide |
Answer» SOLUTION :
|
|
| 16. |
Starting with alcohol, outline a synthesis of benzyl bromide |
Answer» SOLUTION :
|
|
| 17. |
Starting materials for the preperation of inorganic benzene is |
|
Answer» `B_2 H_6 + HN_3` |
|
| 18. |
Starting with 10 g of a radioactive substance 0.1 gis left after 10 days. The rate constant for the decay is: |
|
Answer» `0.9212"DAY"^(-1)` |
|
| 19. |
Starting from the following reagents, how will your prepare ethanenitrile. CH_3CONH_2 |
|
Answer» SOLUTION :By DEHYDRATION of primary amides and ALDOXIMES with `P_2O_5` `{:(CH_3-CONH_2underset(-H_2O)OVERSET(P_2O_5)(to)CH_3-CN),("AcetamideEthanenitrile"):}`. |
|
| 20. |
Starting materials for nylon -66 are |
|
Answer» hexmethylene diamine and ethylene GLYCOL |
|
| 21. |
Starting material for Nylon - 6 is |
|
Answer» `ALPHA` - CAPROLACTUM 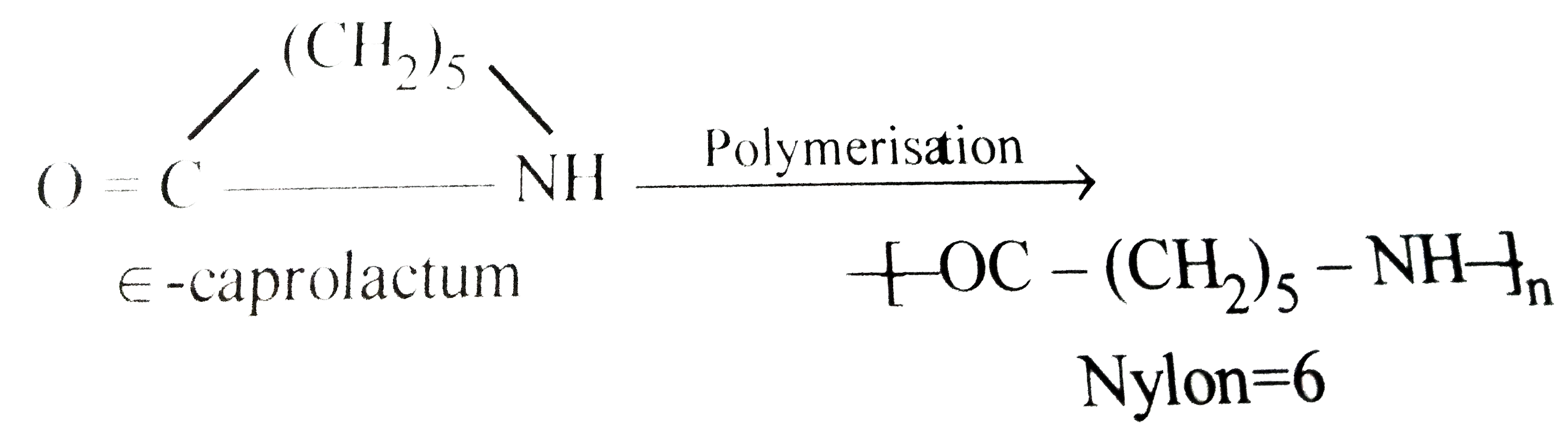
|
|
| 22. |
Starting from the following reagents, how will your prepare ethanenitrile. CH_3CH=NOH |
| Answer» SOLUTION :`{:(CH_3-CH=NOHunderset(-H_2O)OVERSET(P_2O_5)(to)CH_3-CN),("AcetaldoximesEthanenitrile"):}` | |
| 23. |
Starting from SiCl_(4), prepare the following in steps not exceeding the number given in parentheses. Silicon |
| Answer» SOLUTION :`3SiCl_(4)+4AL to 4AlCl_(3)+3Si` | |
| 24. |
Starting from the following reagents, how will your prepare ethanenitrile. CH_3Br |
|
Answer» Solution :Aryl cyanide cannot be prepared in this METHOD because of their less reactivity TOWARDS NUCLEOPHILIC substitution. Aryl cyanides are prepared using Sandmeyers reactions. `{:(KCN+CH_3-Br to CH_3-CN+KBr),("methyl bromideethanenitrile"):}`. |
|
| 25. |
Starting from SiCl_(4), prepare the following in steps not exceeding the number given in parentheses. Na_(2)SiO_(3). |
|
Answer» Solution :`SiCl_(4)+4H_(2)O to underset("orthosilicic acid")(H_(4)SiO_(4)+4HCL)` `H_(4)SiO_(4)OVERSET(Delta)to SiO_(2)+2H_(2)O` `SiO_(2)+Na_(2)CO_(3)to Na_(2)SiO_(3)+H_(2)O`. |
|
| 26. |
Starting from SiCl_(4), prepare the following in steps not exceeding the number given in parentheses. Linear silicon containing methyl groups only |
| Answer» Solution :`n(CH_(3))_(2)SI(OH)_(2)OVERSET(Delta)to-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(Si)-O-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(Si)-O-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(Si)-O-` | |
| 27. |
Starting from radium, the radioactive disintegration process terminates when the following is obtained |
|
Answer» Lead |
|
| 28. |
Starting from propanoic acid, the following reactions were carried out what is the compound Z |
|
Answer» `CH_(3)-CH_(2)-BR` |
|
| 29. |
Starting from radium, the radioactive disintegration process terminates when the following is obtained: |
| Answer» Answer :A | |
| 30. |
Starting from phenol, how would you prepare the following compounds. (i)Benzene (ii) Aniline (iii) Anisole (iv) 1,4, benzoquinone (v) Cyclohexanol |
Answer» SOLUTION :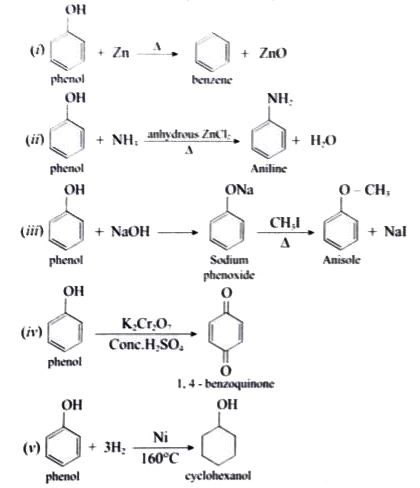
|
|
| 31. |
Starting from methyl magnesium iodide, how would you prepare acetic acid? |
Answer» SOLUTION :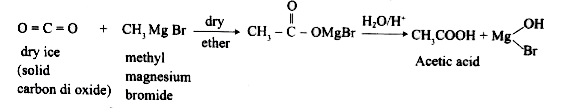
|
|
| 32. |
Starting from diethyl malonate, what type of acids can prepared from it? |
|
Answer» Succinic ACID |
|
| 33. |
Starting from ethyl ethanoate, how would you prepare ethanol? |
| Answer» SOLUTION :`UNDERSET("ETHYL ethanoate")(CH_3COOCH_2CH_3)underset(H_2O)OVERSET(LiAlH_4)to underset("ETHANOL")(2CH_3-CH_2OH)` | |
| 34. |
Starting from methyl magnesium bromide, how would you obtain ethane nitrile? |
Answer» Solution :`underset("METHYL magnesium bromide")(CH_(3)MGBR)+underset("Cyanogaen chloride")(Cl-CN)tounderset("Ethane nitrile")(CH_(3)-CN)+ ` 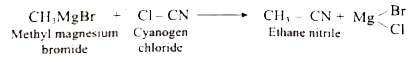
|
|
| 35. |
Starting from butyl magnesium bromide, how would you obtain 2-methyl hexan-2-o1? |
Answer» SOLUTION :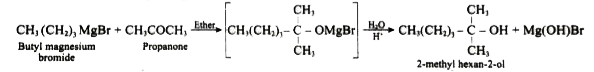
|
|
| 36. |
Starting from benzene diazonium chloride how would you get bright orange azo dye? |
Answer» SOLUTION :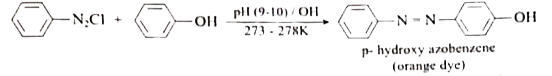
|
|
| 37. |
Starting frombenzene diazonium chloride, how would you prepare (i) Biphenyl (ii) Phenyl hydrazine (iii) p-hydroxy azo benzene (iv) p-amino azo benzene (v) Chloro benzene |
|
Answer» SOLUTION :(i) `underset("Benzene DIAZONIUM chlorid")(C_(6)H_(5)N_(2)^(+)Cl^(-))+underset("Benzene")(C_(6)H_(6))overset(NaOH)(to)underset("BIPHENYL")(C_(6)H_(5))-C_(6)H_(5)+N_(2)uarr+HCl` (ii) `C_(6)H_(5)N_(2)^(+)Cl^(-)underset(("or")Na_(2)SO_(3))overset(Sncl_(2)+HCl)(to)underset("Phenol hydrazine")(C_(6)H_(5)NH-NH_(2))` 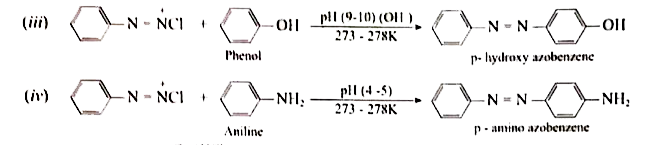 (V) `C_(6)H_(5)N_(2)^(+)Cl^(-)overset(Cu//HCl)(to) underset("Chloro benzene")(C_(6)H_(5)Cl)+N_(2)uarr` |
|
| 38. |
Starting from Benzene diazonium chloride, how would you prepare (i) Benzen (ii) Phenol (iii) Nitro benzene (iv)( Benzoic acid (v) Fluorobenzene |
|
Answer» SOLUTION :(i) `underset("Benzene diazonium chloride")(C_(6)H_(5)N_(2)^(+)Cl^(-))+H_(3)PO_(2)+H_(2)Otounderset("Benzene")(C_(6)H_(6))+H_(3)PO_(3)+HCl+N_(2)uarr` (ii) `C_(6)H_(5)N_(2)^(+)Cl^(-)+H_(2)O overset(DELTA//283K)(to)underset("Phenol")(C_(6)H_(5)OH)+N_(2)uarr+HCl` (iii) `C_(6)H_(5)N_(2)^(+)Cl^(-)+HBF_(4)toC_(6)H_(5)N_(2)^(+)BF_(4)^(-)underset(Delta)overset(NaNO_(2)//Cu)(to)underset("Nitro benzene")(C_(6)H_(5)NO_(2))+N_(2)uarr +NaBF_(4)` (iv) `C_(6)H_(5)N_(2)^(+)Cl^(-)+HBF_(4)toC_(6)H_(5)N_(2)^(+)BF_(4)^(-)+CH_(3)COOHtounderset(Benzoic acid")(C_(6)H_(5)COOH)+BF_(3)+CH_(3)F` (v) `C_(6)H_(5)N_(2)^(+)Cl^(-)+HBF_(4)toC_(6)H_(5)N_(2)^(+)BF_(4)^(-) overset(Delta)(to) underset("FLUORO benzene")(C_(6)H_(5)F)+BF_(3)+N_(2)uarr` |
|
| 39. |
Starting from aniline how can the following be prepared ? (a) Chloro benzene (b) p-hydroxy azobenzene (c) Benzonitrile (d) (d) p-amino azo benzene |
|
Answer» SOLUTION :(a) `C_(6)H_(5)NH_(2) OVERSET(HNO_(2))underset(HCl)rarr C_(6)H_(5)N_(2)CL overset(HCl)underset(Cu_(2)Cl_(2))rarr C_(6)H_(5)Cl` (b) `C_(6)H_(5)NH_(2) overset(HNO_(2))underset(HCl)rarr C_(5)H_(5)N_(2)Cl overset("Phenol")rarr C_(6)H_(5)-N=N-C_(6)H_(4)-OH` (c ) `C_(6)H_(5)NH_(2) overset (HNO_(2))underset(HCl)rarr C_(6)H_(5)N_(2)Cl overset(KCN) underset(Cu_(2)(CN)_(2))rarr C_(6)H_(5)CN` (d) `C_(6)H_(5)NH_(2) overset (HNO_(2)) underset(HCl)rarr C_(6)H_(5)N_(2)Cl overset("Aniline")rarr C_(6)H_(5)N=NC_(6)H_(4)NH_(2)` |
|
| 40. |
Starting from aniline how can the following be prepared ? (a) Chloro benzene (b) p-hydroxy azobenzene (c) Benzonitrile (d) p-amino azo benzene |
|
Answer» Solution :(a) `C_(6)H_(5)NH_(2) OVERSET(HNO_(2))underset(HCl)rarr C_(6)H_(5)N_(2)CL overset(HCl)underset(Cu_(2)Cl_(2))rarr C_(6)H_(5)Cl` (b) `C_(6)H_(5)NH_(2) overset(HNO_(2))underset(HCl)rarr C_(5)H_(5)N_(2)Cl overset("PHENOL")rarr C_(6)H_(5)-N=N-C_(6)H_(4)-OH` (c ) `C_(6)H_(5)NH_(2) overset (HNO_(2))underset(HCl)rarr C_(6)H_(5)N_(2)Cl overset(KCN) underset(Cu_(2)(CN)_(2))rarr C_(6)H_(5)CN` (d) `C_(6)H_(5)NH_(2) overset (HNO_(2)) underset(HCl)rarr C_(6)H_(5)N_(2)Cl overset("ANILINE")rarr C_(6)H_(5)N=NC_(6)H_(4)NH_(2)` |
|
| 41. |
Starting form Benzene diazonium chloride, how will you get Nitrobenzene? |
|
Answer» Solution :When benzene diazonium CHLORIDE is treated with fluororic acid we get diazonium fluorobroate which on treated with sodium nitrite solution in the PRESENCE of copper, nirobenzene is obtained. `underset("Benzene diazonium chloride")(C_(6)H_(5)N_(2)""^(+)Cl^(-))+underset("Hydro fluoro boric acid")(HBF_(4))to underset("Benzenediazonium fluoroborate")(C_(6)H_(5)-N_(2)^(+)BF_(4)^(-)BF_(4)^(-)underset(DELTA)overset(NaNO_(2)//Cu)(to) underset("Nitrobenzene")(C_(6)H_(5)NO_(2))+N_(2)uarr+NaBF_(4)` |
|
| 42. |
Starch when heated with enzyme diastase yields |
|
Answer» GLUCOSE |
|
| 43. |
Starch reacts with iodine to give : |
|
Answer» blueblack COLOUR |
|
| 44. |
Starch paper is used to test for the presence of |
|
Answer» Iodine as: `I^(-)+" oxidant "rarr I_(2)` `I_(2)+" starch "rarr "BLUE COLOUR"` |
|
| 45. |
Starch paper moistened with KI solution turns blue in ozone because of: |
|
Answer» IODINE liberation |
|
| 46. |
Starch undergoes hydrolysis in presence of mineral acids to give fructose.True/false |
|
Answer» |
|
| 47. |
starch on hydrolysis produces |
|
Answer» `ALPHA`-D-glucose |
|
| 48. |
Starch on hydrolysis by a dilute inorganic mineral acid gives : |
|
Answer» SUCROSE |
|
| 49. |
Starch is used to test even a small of |
|
Answer» Urea in BLOOD |
|
| 50. |
Starch is used for …………………………in plants. |
| Answer» SOLUTION :energystorage | |