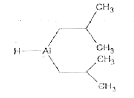
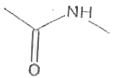
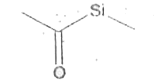
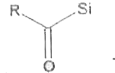
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Reagent 'A' can be, |
|
Answer» `CF_(3)CO_(3)H` 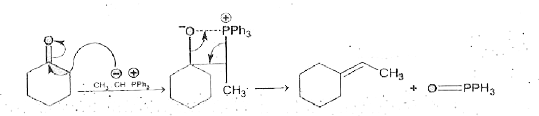 Hence, (B) and ( C ) are the correct ANSWERS. |
|
| 2. |
Read the passage given below and answer the questions that follows: Nitrogen differs from the rest of the members of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d-orbitals. Nitrogen has unique ability to form p pi- p pimultiple bonds with itself and with other elements having small size and high electronegativity (For example, C, O). Heavier elements of this group do not form p pi - p pibonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diatomic molecule with a triple bond (one s and two p) between the two atoms. Consequently, its bond enthalpy (941.4 kJ "mol"^(-1)) is very high. On the contrary, phosphorus, arsenic and antimony form single bonds as P-P, As-As and Sb-Sb while bismuth forms metallic bonds in elemental state, However, the single N-N bond is weaker than the single P-P bond because of high interelectronic repulsion of the non-bonding electrons, owing to the small bond length. Why is the single N- N bond weaker than the single P - P bond ? |
| Answer» Solution : It is because of the HIGH interelectronic REPULSIONS between non-bonding ELECTRONS on nitrogen ATOMS owing to small bond length between nitrogen atoms. | |
| 3. |
Read the passage given below and answer the questions that follows: Nitrogen differs from the rest of the members of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d-orbitals. Nitrogen has unique ability to form p pi- p pimultiple bonds with itself and with other elements having small size and high electronegativity (For example, C, O). Heavier elements of this group do not form p pi - p pibonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diatomic molecule with a triple bond (one s and two p) between the two atoms. Consequently, its bond enthalpy (941.4 kJ "mol"^(-1)) is very high. On the contrary, phosphorus, arsenic and antimony form single bonds as P-P, As-As and Sb-Sb while bismuth forms metallic bonds in elemental state, However, the single N-N bond is weaker than the single P-P bond because of high interelectronic repulsion of the non-bonding electrons, owing to the small bond length.Heavier elements of Group 15 do not form p pi - p pibonds. |
| Answer» Solution :ATOMIC orbitals of heavier ELEMENTS are so LARGE and diffused that they cannot have EFFECTIVE OVERLAPPING | |
| 4. |
Read the passage given below and answer the questions that follows: Nitrogen differs from the rest of the members of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d-orbitals. Nitrogen has unique ability to form p pi- p pimultiple bonds with itself and with other elements having small size and high electronegativity (For example, C, O). Heavier elements of this group do not form p pi - p pibonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diatomic molecule with a triple bond (one s and two p) between the two atoms. Consequently, its bond enthalpy (941.4 kJ "mol"^(-1)) is very high. On the contrary, phosphorus, arsenic and antimony form single bonds as P-P, As-As and Sb-Sb while bismuth forms metallic bonds in elemental state, However, the single N-N bond is weaker than the single P-P bond because of high interelectronic repulsion of the non-bonding electrons, owing to the small bond length. What kind of bond is formed in the molecule of nitrogen ? |
|
Answer» <P> SOLUTION :It is `p PI - p pi ` BOND. |
|
| 5. |
Read the passage given below and answer the questions that follows: Nitrogen differs from the rest of the members of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d-orbitals. Nitrogen has unique ability to form p pi- p pimultiple bonds with itself and with other elements having small size and high electronegativity (For example, C, O). Heavier elements of this group do not form p pi - p pibonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diatomic molecule with a triple bond (one s and two p) between the two atoms. Consequently, its bond enthalpy (941.4 kJ "mol"^(-1)) is very high. On the contrary, phosphorus, arsenic and antimony form single bonds as P-P, As-As and Sb-Sb while bismuth forms metallic bonds in elemental state, However, the single N-N bond is weaker than the single P-P bond because of high interelectronic repulsion of the non-bonding electrons, owing to the small bond length. Why does nitrogen differ from rest of the members of the Group ? |
| Answer» Solution :This is due to small size, high ELECTRONEGATIVITY, high ionisation ENTHALPY and ABSENCE of d-orbitals in nitrogen. | |
| 6. |
Read the passage given below and answer the questions that follows: Nitrogen differs from the rest of the members of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d-orbitals. Nitrogen has unique ability to form p pi- p pimultiple bonds with itself and with other elements having small size and high electronegativity (For example, C, O). Heavier elements of this group do not form p pi - p pibonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diatomic molecule with a triple bond (one s and two p) between the two atoms. Consequently, its bond enthalpy (941.4 kJ "mol"^(-1)) is very high. On the contrary, phosphorus, arsenic and antimony form single bonds as P-P, As-As and Sb-Sb while bismuth forms metallic bonds in elemental state, However, the single N-N bond is weaker than the single P-P bond because of high interelectronic repulsion of the non-bonding electrons, owing to the small bond length. Give two examples of compounds that nitrogen forms with oxygen using p pi- p pibond. |
Answer» SOLUTION : 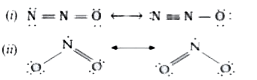
|
|
| 7. |
Read the passage given below and answer the questions that follow: The growing focus on environmental issues within the arena of global politics was firmly consolidated at the United Nations Conference on Environment and Development held at Rio de Janerio, Brazil in June 1992. This was also called the Earth Summit….What was obvious at the Rio Summit was the rich and the developed countries of the first world generally referred to as the “Global North” were pursuing a different environmental agenda than the poor and the developing countries of the III world called the “Global South”...The differences in their approach culminated into the adoption of the principle called “Common but differentiated responsibilities.” a) How was the environmental concern of the first world different from that of the third world? b) In what ways did the principle of “Common but differentiated responsibilities’ help in addressing and overcoming the differences between the “Global North” and ‘Global South”? |
|
Answer» Solution :a) The developed countries of the first world generally referred to as ‘Global North’ were concerned with the ozone depletion and global warming, WHEREAS the developing countries of the third world called the ‘Global South” were anxious to address the relationship between economic development and environmental MANAGEMENT. b) i) There were major differences between the countries of the North and the South over environmental issues. The developed countries of the North wanted everyone to be made EQUALLY responsible for ecological conservation. The Global South on the other hand felt that much of the environmental degradation caused TODAY is the result of economic activities pursued by the Global North. Hence, they believed that the Global North owes a greater responsibility of undoing the damage now. ii) Moreover, the developing countries are undergoing a process of industrialisation and they should not be subjected to the same restrictions which apply to the developed countries. iii)These two concerns were effectively addressed by the Principle of “Common but differentiated responsibilities. It accepted that the special needs of the developing countries be taken into CONSIDERATION and in view of different contributions of global environmental degradation, states have common but differentiated responsibilities. |
|
| 8. |
Read the paragraph carefully and answer the questions given below it,Stereo//space isomerism in coordination compoundsGcometrical isomerism: Exhibited by square planer and octahedral complexes (not by tetrahedral complexes because relative positions are similar). If opposite positions have similar groups, it is trans, otherwise it is cis, Optical isomerism: Shown when mirror image is non-superimposable. Cis-isomer shows but trans isomer does notQWhich of the following types of octahedral complexes will exhibit geometrical isomerism (where M = metal and a, b are achiral ligands) |
| Answer» Answer :D | |
| 9. |
Read the paragraph carefully and answer the questions given below it,Stereo//space isomerism in coordination compoundsGcometrical isomerism: Exhibited by square planer and octahedral complexes (not by tetrahedral complexes because relative positions are similar). If opposite positions have similar groups, it is trans, otherwise it is cis, Optical isomerism: Shown when mirror image is non-superimposable. Cis-isomer shows but trans isomer does notQBoth geometrical and optical isomerism are shown by |
|
Answer» `[Co(en)_(2) Cl_(2)]^(+)` |
|
| 10. |
Read the paragraph carefully and answer the questions given below it,Stereo//space isomerism in coordination compoundsGcometrical isomerism: Exhibited by square planer and octahedral complexes (not by tetrahedral complexes because relative positions are similar). If opposite positions have similar groups, it is trans, otherwise it is cis, Optical isomerism: Shown when mirror image is non-superimposable. Cis-isomer shows but trans isomer does notQWhich of the following types of square planar complexes can show geometrical isomerism (where M = metal and a, b = achiral ligands) |
|
Answer» `Ma_(4)` |
|
| 11. |
Read the paragraph carefully and answer the questions given below it,Stereo//space isomerism in coordination compoundsGcometrical isomerism: Exhibited by square planer and octahedral complexes (not by tetrahedral complexes because relative positions are similar). If opposite positions have similar groups, it is trans, otherwise it is cis, Optical isomerism: Shown when mirror image is non-superimposable. Cis-isomer shows but trans isomer does notQHow many geometrical isomers are possible for the square planar complex[Pt(NO_(2))(Py)(NH_(3))(NH_(2)OH)]NO_(2) |
|
Answer» FOUR |
|
| 12. |
Read the paragraph carefully and answer the questions given below it,Stereo//space isomerism in coordination compoundsGcometrical isomerism: Exhibited by square planer and octahedral complexes (not by tetrahedral complexes because relative positions are similar). If opposite positions have similar groups, it is trans, otherwise it is cis, Optical isomerism: Shown when mirror image is non-superimposable. Cis-isomer shows but trans isomer does notQ |
|
Answer» |
|
| 13. |
Read the paragraph carefully and answer the following questions:The property of hydrides of p-block elements mostly depends on(i) (electronegativity difference between central atom and hydrogen..(ii) size of central atom and (iii)number of valence electrons in central atom. Some undergo hydrolysis in which central atom is less electronegative, Teal with OH to give hydrogen. While acidic property of hydride in a period depends on electronegativity of central atoms, I.e. more electronegative is the atom, more acidic is hydride. In a group, acidic property is proportional to the size of central atom. Some electron deficient hydride behaves as Lewis acid while only one hydride of an element in pblock behaves as Lewis base with lone pair of electrons Hydrides in which central atom's electronegativity is close to hydrogen has no reaction with water.QWhich one undergoes detonation when struck? |
|
Answer» `P_(2)H_(4)` |
|
| 14. |
Read the paragraph carefully and answer the following questions:The property of hydrides of p-block elements mostly depends on(i) (electronegativity difference between central atom and hydrogen..(ii) size of central atom and (iii)number of valence electrons in central atom. Some undergo hydrolysis in which central atom is less electronegative, Teal with OH to give hydrogen. While acidic property of hydride in a period depends on electronegativity of central atoms, I.e. more electronegative is the atom, more acidic is hydride. In a group, acidic property is proportional to the size of central atom. Some electron deficient hydride behaves as Lewis acid while only one hydride of an element in pblock behaves as Lewis base with lone pair of electrons Hydrides in which central atom's electronegativity is close to hydrogen has no reaction with water.QWhich one undergoes spontaneous combustion with exposure to air? |
|
Answer» `PH_(3)` |
|
| 15. |
Read the paragraph carefully and answer the following questions:The property of hydrides of p-block elements mostly depends on(i) (electronegativity difference between central atom and hydrogen..(ii) size of central atom and (iii)number of valence electrons in central atom. Some undergo hydrolysis in which central atom is less electronegative, Teal with OH to give hydrogen. While acidic property of hydride in a period depends on electronegativity of central atoms, I.e. more electronegative is the atom, more acidic is hydride. In a group, acidic property is proportional to the size of central atom. Some electron deficient hydride behaves as Lewis acid while only one hydride of an element in pblock behaves as Lewis base with lone pair of electrons Hydrides in which central atom's electronegativity is close to hydrogen has no reaction with water.QWhich one is the weakest acid among the following? |
|
Answer» HE |
|
| 16. |
Read the paragraph carefully and answer the following questions:The property of hydrides of p-block elements mostly depends on(i) (electronegativity difference between central atom and hydrogen..(ii) size of central atom and (iii)number of valence electrons in central atom. Some undergo hydrolysis in which central atom is less electronegative, Teal with OH to give hydrogen. While acidic property of hydride in a period depends on electronegativity of central atoms, I.e. more electronegative is the atom, more acidic is hydride. In a group, acidic property is proportional to the size of central atom. Some electron deficient hydride behaves as Lewis acid while only one hydride of an element in pblock behaves as Lewis base with lone pair of electrons Hydrides in which central atom's electronegativity is close to hydrogen has no reaction with water.QWhich one is highly flammable in nature? |
|
Answer» `NaBH_(4)` |
|
| 17. |
Read the paragraph carefully and answer the following questions:The property of hydrides of p-block elements mostly depends on(i) (electronegativity difference between central atom and hydrogen..(ii) size of central atom and (iii)number of valence electrons in central atom. Some undergo hydrolysis in which central atom is less electronegative, Teal with OH to give hydrogen. While acidic property of hydride in a period depends on electronegativity of central atoms, I.e. more electronegative is the atom, more acidic is hydride. In a group, acidic property is proportional to the size of central atom. Some electron deficient hydride behaves as Lewis acid while only one hydride of an element in pblock behaves as Lewis base with lone pair of electrons Hydrides in which central atom's electronegativity is close to hydrogen has no reaction with water.QWhich one is strongest base? |
|
Answer» `OH^(-)` |
|
| 18. |
Read the paragraph carefully and answer the following questions:Silicon is the second most abundant element occurring in earth crust. It is always found in combined state. It is obtained by reduction of silica and exists in two allotropic forms, i.e. amorphous and crystalline. Amorphous form is obtained by heating dry powdered silica with magnesium. Crystalline form is obtained by heating sand or quartz in a furnace. Silicon is used in manufacturing of certain alloys and polymers such as ferrosilicon, silicon bronze etc. The compounds of silicon are carborundum, silicone, silicates, glass etc. Silicones are organosilicon polymer and silicates are metal derivatives of silicic acid. While carborundum are obtained by heating mixture of sand, carbon and common salt.Q(SİH_(3))_(3)is a weaker base than (CH_(3))_(3)N, because |
|
Answer» In `(SiH_(3))_(3)N` lone PAIR of electrons on nitrogen is involved in `dpi-ppi` bonding |
|
| 19. |
Read the paragraph carefully and answer the following questions:Silicon is the second most abundant element occurring in earth crust. It is always found in combined state. It is obtained by reduction of silica and exists in two allotropic forms, i.e. amorphous and crystalline. Amorphous form is obtained by heating dry powdered silica with magnesium. Crystalline form is obtained by heating sand or quartz in a furnace. Silicon is used in manufacturing of certain alloys and polymers such as ferrosilicon, silicon bronze etc. The compounds of silicon are carborundum, silicone, silicates, glass etc. Silicones are organosilicon polymer and silicates are metal derivatives of silicic acid. While carborundum are obtained by heating mixture of sand, carbon and common salt.QWhen two structural units of silicates join along a corner containing oxygen atom is called |
|
Answer» orthosilicate |
|
| 20. |
Read the paragraph carefully and answer the following questions:Silicon is the second most abundant element occurring in earth crust. It is always found in combined state. It is obtained by reduction of silica and exists in two allotropic forms, i.e. amorphous and crystalline. Amorphous form is obtained by heating dry powdered silica with magnesium. Crystalline form is obtained by heating sand or quartz in a furnace. Silicon is used in manufacturing of certain alloys and polymers such as ferrosilicon, silicon bronze etc. The compounds of silicon are carborundum, silicone, silicates, glass etc. Silicones are organosilicon polymer and silicates are metal derivatives of silicic acid. While carborundum are obtained by heating mixture of sand, carbon and common salt.QThe general formulae for a glass obtained from silica is(where R = Na or Kand M = Ca, Ba, Zn or Pb) |
|
Answer» `RO.MO:4SIO_(2)` |
|
| 21. |
Read the paragraph carefully and answer the following questions:Silicon is the second most abundant element occurring in earth crust. It is always found in combined state. It is obtained by reduction of silica and exists in two allotropic forms, i.e. amorphous and crystalline. Amorphous form is obtained by heating dry powdered silica with magnesium. Crystalline form is obtained by heating sand or quartz in a furnace. Silicon is used in manufacturing of certain alloys and polymers such as ferrosilicon, silicon bronze etc. The compounds of silicon are carborundum, silicone, silicates, glass etc. Silicones are organosilicon polymer and silicates are metal derivatives of silicic acid. While carborundum are obtained by heating mixture of sand, carbon and common salt.Q The structural unit of silicates is |
|
Answer» `SiO_(2)` |
|
| 22. |
Read the paragraph carefully and answer the following questions:Silicon is the second most abundant element occurring in earth crust. It is always found in combined state. It is obtained by reduction of silica and exists in two allotropic forms, i.e. amorphous and crystalline. Amorphous form is obtained by heating dry powdered silica with magnesium. Crystalline form is obtained by heating sand or quartz in a furnace. Silicon is used in manufacturing of certain alloys and polymers such as ferrosilicon, silicon bronze etc. The compounds of silicon are carborundum, silicone, silicates, glass etc. Silicones are organosilicon polymer and silicates are metal derivatives of silicic acid. While carborundum are obtained by heating mixture of sand, carbon and common salt.QWhich type of linkage is present in silicones? |
|
Answer»
|
|
| 23. |
Read the paragraph carefully and answer the following questions: Real gases deviate from ideal behaviour because of the following two faulty assumptions of kinetic theory. (I) The actual volume occupied by molecules is negligible as compared to the total volume of the gas. (II) The forces of attraction and repulsion between molecules of the gas are negligible. The extent of deviation of a real gas from ideal behaviour is expressed in terms of compressibility factor (z). Hence, suitable corrections were applied to the ideal gas equation so that it can also explain the behaviour of real gases. The equation obtained by applying the two corrections to the usual gas equation is known as van der Waal equation. A Volume correction Corrected (ideal) volume = (V - b) Where b is the effective volume of the molecules. B. Pressure correction (intermolecular attraction corection). Corrected (ideal) pressure = P + P However, P=(a)/(V^(2)) therefore The van der Waal's equation becomes (P+(an^(2))/(V^(2)))(V-nb)=nRT van der Waal equation is obeyed by the real gases |
|
Answer» over a WIDE RANGE of TEMPERATURE and pressure |
|
| 24. |
Read the paragraph carefully and answer the following questions: Real gases deviate from ideal behaviour because of the following two faulty assumptions of kinetic theory. (I) The actual volume occupied by molecules is negligible as compared to the total volume of the gas. (II) The forces of attraction and repulsion between molecules of the gas are negligible. The extent of deviation of a real gas from ideal behaviour is expressed in terms of compressibility factor (z). Hence, suitable corrections were applied to the ideal gas equation so that it can also explain the behaviour of real gases. The equation obtained by applying the two corrections to the usual gas equation is known as van der Waal equation. A Volume correction Corrected (ideal) volume = (V - b) Where b is the effective volume of the molecules. B. Pressure correction (intermolecular attraction corection). Corrected (ideal) pressure = P + P However, P=(a)/(V^(2)) therefore The van der Waal's equation becomes (P+(an^(2))/(V^(2)))(V-nb)=nRT The values of van der Waal's constant 'a' ("atm"^(2) L^(2)" mol"^(2)) for the gases N_(2), H_(2) NH_(3) and SO_(2) are given 0.25, 3.5, 4,9 and 6.9 respectively. Which will have the highest ease of liquefaction? |
| Answer» Answer :A | |
| 25. |
Read the paragraph carefully and answer the following questions: Real gases deviate from ideal behaviour because of the following two faulty assumptions of kinetic theory. (I) The actual volume occupied by molecules is negligible as compared to the total volume of the gas. (II) The forces of attraction and repulsion between molecules of the gas are negligible. The extent of deviation of a real gas from ideal behaviour is expressed in terms of compressibility factor (z). Hence, suitable corrections were applied to the ideal gas equation so that it can also explain the behaviour of real gases. The equation obtained by applying the two corrections to the usual gas equation is known as van der Waal equation. A Volume correction Corrected (ideal) volume = (V - b) Where b is the effective volume of the molecules. B. Pressure correction (intermolecular attraction corection). Corrected (ideal) pressure = P + P However, P=(a)/(V^(2)) therefore The van der Waal's equation becomes (P+(an^(2))/(V^(2)))(V-nb)=nRT The pressure correction used on the ideal gas behaviour is generally indicated by P +P what does this P' stand for |
|
Answer» the pressure not involved in STRIKING the wall |
|
| 26. |
Read the paragraph carefully and answer the following questions: Real gases deviate from ideal behaviour because of the following two faulty assumptions of kinetic theory. (I) The actual volume occupied by molecules is negligible as compared to the total volume of the gas. (II) The forces of attraction and repulsion between molecules of the gas are negligible. The extent of deviation of a real gas from ideal behaviour is expressed in terms of compressibility factor (z). Hence, suitable corrections were applied to the ideal gas equation so that it can also explain the behaviour of real gases. The equation obtained by applying the two corrections to the usual gas equation is known as van der Waal equation. A Volume correction Corrected (ideal) volume = (V - b) Where b is the effective volume of the molecules. B. Pressure correction (intermolecular attraction corection). Corrected (ideal) pressure = P + P However, P=(a)/(V^(2)) therefore The van der Waal's equation becomes (P+(an^(2))/(V^(2)))(V-nb)=nRT At low pressure, the van der waal equation is |
|
Answer» `P=RT+(a)/(V)` |
|
| 27. |
Read the paragraph carefully and answer the following questions: On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV=(1)/(3) mnU^(2) where, P = Pressure of the gas, V = Volume of the gas, m = Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = Root mean square speed For one mole of gas, PV=RT and n=N_(A) (1)/(3) mN_(A)u^(2)=RT (2)/(3) cdot (1)/(2) mN_(A) u^(2)=RT ((1)/(2)mN_(A)u^(2)=" K. E. per mol") (2)/(3) K.E.=RT rArr K.E. =(3)/(2) RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. Thus, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule =("Average K. E. per mole")/(N) =(3)/(2) (RT)/(N) rArr (3)/(2) kT where k is the Boltzmann constant. The average K.E. of an inert gas in calories per mole is approximately equal to |
|
Answer» THREE TIMES the ABSOLUTE TEMPERATURE |
|
| 28. |
Read the paragraph carefully and answer the following questions: Real gases deviate from ideal behaviour because of the following two faulty assumptions of kinetic theory. (I) The actual volume occupied by molecules is negligible as compared to the total volume of the gas. (II) The forces of attraction and repulsion between molecules of the gas are negligible. The extent of deviation of a real gas from ideal behaviour is expressed in terms of compressibility factor (z). Hence, suitable corrections were applied to the ideal gas equation so that it can also explain the behaviour of real gases. The equation obtained by applying the two corrections to the usual gas equation is known as van der Waal equation. A Volume correction Corrected (ideal) volume = (V - b) Where b is the effective volume of the molecules. B. Pressure correction (intermolecular attraction corection). Corrected (ideal) pressure = P + P However, P=(a)/(V^(2)) therefore The van der Waal's equation becomes (P+(an^(2))/(V^(2)))(V-nb)=nRT 0.5 value of compressibility factor (z) indicates that the gas |
|
Answer» shows positive DEVIATION from the IDEAL gas |
|
| 29. |
Read the paragraph carefully and answer the following questions: On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV=(1)/(3) mnU^(2) where, P = Pressure of the gas, V = Volume of the gas, m = Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = Root mean square speed For one mole of gas, PV=RT and n=N_(A) (1)/(3) mN_(A)u^(2)=RT (2)/(3) cdot (1)/(2) mN_(A) u^(2)=RT ((1)/(2)mN_(A)u^(2)=" K. E. per mol") (2)/(3) K.E.=RT rArr K.E. =(3)/(2) RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. Thus, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule =("Average K. E. per mole")/(N) =(3)/(2) (RT)/(N) rArr (3)/(2) kT where k is the Boltzmann constant. Which of the following is valid at absolute zero? |
|
Answer» KINETIC ENERGY of the gas becomes zero but the molecular motion does not become zero. |
|
| 30. |
Read the paragraph carefully and answer the following questions: On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV=(1)/(3) mnU^(2) where, P = Pressure of the gas, V = Volume of the gas, m = Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = Root mean square speed For one mole of gas, PV=RT and n=N_(A) (1)/(3) mN_(A)u^(2)=RT (2)/(3) cdot (1)/(2) mN_(A) u^(2)=RT ((1)/(2)mN_(A)u^(2)=" K. E. per mol") (2)/(3) K.E.=RT rArr K.E. =(3)/(2) RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. Thus, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule =("Average K. E. per mole")/(N) =(3)/(2) (RT)/(N) rArr (3)/(2) kT where k is the Boltzmann constant. Calculate the average kinetic energy (in joule) of the molecules in 8 g methane at 27^(@)C. |
|
Answer» `62.14 XX 10^(-22) J` |
|
| 31. |
Read the paragraph carefully and answer the following questions: On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV=(1)/(3) mnU^(2) where, P = Pressure of the gas, V = Volume of the gas, m = Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = Root mean square speed For one mole of gas, PV=RT and n=N_(A) (1)/(3) mN_(A)u^(2)=RT (2)/(3) cdot (1)/(2) mN_(A) u^(2)=RT ((1)/(2)mN_(A)u^(2)=" K. E. per mol") (2)/(3) K.E.=RT rArr K.E. =(3)/(2) RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. Thus, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule =("Average K. E. per mole")/(N) =(3)/(2) (RT)/(N) rArr (3)/(2) kT where k is the Boltzmann constant. Which of the following expressions correctly represents the relationship between the average molar kinetic energies of CO and N_(2) molecules at the same temperature? |
|
Answer» `K.E. (CO) =K.E.(N_(2))` |
|
| 32. |
Read the paragraph carefully and answer the following questions: On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV=(1)/(3) mnU^(2) where, P = Pressure of the gas, V = Volume of the gas, m = Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = Root mean square speed For one mole of gas, PV=RT and n=N_(A) (1)/(3) mN_(A)u^(2)=RT (2)/(3) cdot (1)/(2) mN_(A) u^(2)=RT ((1)/(2)mN_(A)u^(2)=" K. E. per mol") (2)/(3) K.E.=RT rArr K.E. =(3)/(2) RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. Thus, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule =("Average K. E. per mole")/(N) =(3)/(2) (RT)/(N) rArr (3)/(2) kT where k is the Boltzmann constant. In deriving the kinetic gas equation, the use of the root mean square speed of the molecule is done, because it is |
|
Answer» average SPEED of the molecules |
|
| 33. |
Read the paragraph carefully and answer the following questions: Mole fraction of acetic acid in benzene given a solution having freezing point 277.4K. Freezing point of benezene is 278.4K and heat of fusion of benzene is 10.042 kJ/mol. If molarity of solution is equal to molality, and acetic acid is found in equilibrium with it dimmer than answer the following based upon above data Calculate the equilibrium constant for dimerisation of acetic acid |
|
Answer» 3.39 |
|
| 34. |
Read the paragraph carefully and answer the following questions: During gas analysis, the eudiometer tube filled with mercury is inverted over a trough containing. mercury. A known volume of the gas. or gaseous mixture to be studied is next introduced, which displaces an equivalent amount of mercury. Next a known amount, of oxygen in excess is introduced and the electric spark is passed, whereby the combustible material gets oxidized. The volume of carbon dioxide, water vapour or other gaseous products of combustíon are next determined by absorbing them in suitable reagents. The various reagents used for absorbing different gases areO_(3) rarr turpentine oil O_(2) rarr alkalino pyragallol CO_(2), SO_(2) rarr alkali solution [NaOH,KOH,Ca(OH)_(2)] In a solid 'AB' having a NaCl structure, A atoms occupy the coners of the cubic unit cell. If all the face centred, atoms alonig one of the axes are removed, then the resultant stoichiometry of the solid is |
|
Answer» `AB_(2)` |
|
| 35. |
Read the paragraph carefully and answer the following questions: Mole fraction of acetic acid in benzene given a solution having freezing point 277.4K. Freezing point of benezene is 278.4K and heat of fusion of benzene is 10.042 kJ/mol. If molarity of solution is equal to molality, and acetic acid is found in equilibrium with it dimmer than answer the following based upon above data K_(1) for benzene is |
|
Answer» `3Kxx` MOLALITY |
|
| 36. |
Read the paragraph carefully and answer the following questions: Mole fraction of acetic acid in benzene given a solution having freezing point 277.4K. Freezing point of benezene is 278.4K and heat of fusion of benzene is 10.042 kJ/mol. If molarity of solution is equal to molality, and acetic acid is found in equilibrium with it dimmer than answer the following based upon above data The degree of association according to above data acetic acid should be |
|
Answer» `0.60` |
|
| 37. |
Read the paragraph carefully and answer the following questions: During gas analysis, the eudiometer tube filled with mercury is inverted over a trough containing. mercury. A known volume of the gas. or gaseous mixture to be studied is next introduced, which displaces an equivalent amount of mercury. Next a known amount, of oxygen in excess is introduced and the electric spark is passed, whereby the combustible material gets oxidized. The volume of carbon dioxide, water vapour or other gaseous products of combustíon are next determined by absorbing them in suitable reagents. The various reagents used for absorbing different gases areO_(3) rarr turpentine oil O_(2) rarr alkalino pyragallol CO_(2), SO_(2) rarr alkali solution [NaOH,KOH,Ca(OH)_(2)] 16 ml of a gaseous aliphatic compound C_(3)H_(3n)O_(n) was mixed with 60 ml CO_(2) and sparked. The gas mixture on cooling occupied 44 ml. After treatment with KOH Sol. the volume of gas remaining was 12 ml. Deduce the fomula of compound |
| Answer» Answer :A | |
| 38. |
Read the paragraph carefully and answer the following questions: Mole fraction of acetic acid in benzene given a solution having freezing point 277.4K. Freezing point of benezene is 278.4K and heat of fusion of benzene is 10.042 kJ/mol. If molarity of solution is equal to molality, and acetic acid is found in equilibrium with it dimmer than answer the following based upon above data What should be the molality of the solution formed |
|
Answer» 0.462 m |
|
| 39. |
Read the paragraph carefully and answer the following questions: During gas analysis, the eudiometer tube filled with mercury is inverted over a trough containing. mercury. A known volume of the gas. or gaseous mixture to be studied is next introduced, which displaces an equivalent amount of mercury. Next a known amount, of oxygen in excess is introduced and the electric spark is passed, whereby the combustible material gets oxidized. The volume of carbon dioxide, water vapour or other gaseous products of combustíon are next determined by absorbing them in suitable reagents. The various reagents used for absorbing different gases areO_(3) rarr turpentine oil O_(2) rarr alkalino pyragallol CO_(2), SO_(2) rarr alkali solution [NaOH,KOH,Ca(OH)_(2)] The percentage by volume of C_(3)H_(8) in a mixture of C_(3)H_(8), CH_(4) and CO is 36.5. Calculate the volume of CO_(2) produced when 100 ml of mixture is burnt in excess of O_(2). |
| Answer» Answer :C | |
| 40. |
Read the paragraph carefully and answer the following questions: During gas analysis, the eudiometer tube filled with mercury is inverted over a trough containing. mercury. A known volume of the gas. or gaseous mixture to be studied is next introduced, which displaces an equivalent amount of mercury. Next a known amount, of oxygen in excess is introduced and the electric spark is passed, whereby the combustible material gets oxidized. The volume of carbon dioxide, water vapour or other gaseous products of combustíon are next determined by absorbing them in suitable reagents. The various reagents used for absorbing different gases areO_(3) rarr turpentine oil O_(2) rarr alkalino pyragallol CO_(2), SO_(2) rarr alkali solution [NaOH,KOH,Ca(OH)_(2)] A gaseous hydrocarbon requires 6 times its. owm volume of O_(2) for complete oxidation and produces 4 times its volume of CO_(2). What is its formula? |
|
Answer» `C_(4)H_(10)` |
|
| 41. |
Read the givenpassage ansanswer question number1 to5 that follow : Amino acids are classified as acidic,baisc orneutral dependingunpon the relative numberofmakes it neutral , morenumberof amino than carboxyl groups makes it basic and more carboxyl groups are compared to amino groups makes it acidic . Theamino acids ,which can be synthesised in the body and mustbe obtained through diet, areknown as essential amino acids. Amino acids areusually colourless, crystalline solids. Theseare water-solute, high melting solids and behavelikesalts rather than simple amines or carboxylicacids . This behaviouris dueto the presence ofboth acidic (carboxyl group) and basic (amino group) in the samemlecule . In aqueous solution , the carboxyl group can losea proton and amino group can accepta proton , giving rise to a dipolarion known as zwitter ion. This is neutral but contains both positiveandnegative charges. In zwitter ionic , form amino acids show amphoteric behaviouras they reactboth with acids and bases.What are essintal amino acids |
| Answer» Solution :AMINO acidswhichare not SYNTHESISED by HUMAN BODY and must be provided through DIET are calledessenital amino acids . | |
| 42. |
Read the paragraph carefully and answer the following questions: During gas analysis, the eudiometer tube filled with mercury is inverted over a trough containing. mercury. A known volume of the gas. or gaseous mixture to be studied is next introduced, which displaces an equivalent amount of mercury. Next a known amount, of oxygen in excess is introduced and the electric spark is passed, whereby the combustible material gets oxidized. The volume of carbon dioxide, water vapour or other gaseous products of combustíon are next determined by absorbing them in suitable reagents. The various reagents used for absorbing different gases areO_(3) rarr turpentine oil O_(2) rarr alkalino pyragallol CO_(2), SO_(2) rarr alkali solution [NaOH,KOH,Ca(OH)_(2)] 0.2 mole sample of hydrocarbon C_(x)H_(y) yields after complete combustion with excess O_(2) gas, 0.8 mole of CO_(2), 1.1 mole of H_(2)O. Hence,, hydrocarbon is |
| Answer» Answer :A | |
| 43. |
Read the givenpassage ansanswer question number1 to5 that follow : Amino acids are classified as acidic,baisc orneutral dependingunpon the relative numberofmakes it neutral , morenumberof amino than carboxyl groups makes it basic and more carboxyl groups are compared to amino groups makes it acidic . Theamino acids ,which can be synthesised in the body and mustbe obtained through diet, areknown as essential amino acids. Amino acids areusually colourless, crystalline solids. Theseare water-solute, high melting solids and behavelikesalts rather than simple amines or carboxylicacids . This behaviouris dueto the presence ofboth acidic (carboxyl group) and basic (amino group) in the samemlecule . In aqueous solution , the carboxyl group can losea proton and amino group can accepta proton , giving rise to a dipolarion known as zwitter ion. This is neutral but contains both positiveandnegative charges. In zwitter ionic , form amino acids show amphoteric behaviouras they reactboth with acids and bases. What is zwitter ion? |
| Answer» SOLUTION :In AQUEOUS solution , amino acids are presentas dipolarion. Thisis KNOWN as ZWITTERION . | |
| 44. |
Read the givenpassage ansanswer question number1 to5 that follow : Amino acids are classified as acidic,baisc orneutral dependingunpon the relative numberofmakes it neutral , morenumberof amino than carboxyl groups makes it basic and more carboxyl groups are compared to amino groups makes it acidic . Theamino acids ,which can be synthesised in the body and mustbe obtained through diet, areknown as essential amino acids. Amino acids areusually colourless, crystalline solids. Theseare water-solute, high melting solids and behavelikesalts rather than simple amines or carboxylicacids . This behaviouris dueto the presence ofboth acidic (carboxyl group) and basic (amino group) in the samemlecule . In aqueous solution , the carboxyl group can losea proton and amino group can accepta proton , giving rise to a dipolarion known as zwitter ion. This is neutral but contains both positiveandnegative charges. In zwitter ionic , form amino acids show amphoteric behaviouras they reactboth with acids and bases. Whatis meant byamphoteric character ? |
| Answer» SOLUTION :Substancethatreact withbothacidsand BASES are calledamphoteric. | |
| 45. |
Read the givenpassage ansanswer question number1 to5 that follow : Amino acids are classified as acidic,baisc orneutral dependingunpon the relative numberofmakes it neutral , morenumberof amino than carboxyl groups makes it basic and more carboxyl groups are compared to amino groups makes it acidic . Theamino acids ,which can be synthesised in the body and mustbe obtained through diet, areknown as essential amino acids. Amino acids areusually colourless, crystalline solids. Theseare water-solute, high melting solids and behavelikesalts ratherthan simple amines or carboxylicacids . This behaviouris dueto the presence ofboth acidic (carboxyl group) and basic (amino group) in the samemlecule . In aqueous solution , the carboxyl group can losea proton and amino group can accepta proton , giving rise to a dipolarion known as zwitter ion. This is neutral but contains both positiveandnegative charges. In zwitter ionic , form amino acids show amphoteric behaviouras they reactboth with acids and bases.An amino acid containstwo amino and onecarboxylicgroup. In whichclass of amino acids would you place it ? |
| Answer» SOLUTION :It MUST be basicamino ACIDS. | |
| 46. |
Read the given passage and answer the questions: Zero order reactions are relatively uncommon but they occur under special conditions. Some enzyme catalysed reactions and reactions which occurs on metal surfaces are a few examples of zero order reactions. The decompositions which occurs on metal surfaces are a few examples of zero order reactions. The decomposition of gaseous ammonia on a hot platinum surface is a zero order reaction at high pressure. 2NH_(3)(g) underset("Pt catalyst") overset(1130K)to N_(2)(g)+3H_(2)(g) Rate=k[NH_(3)]^(0)=k In this reaction, Pt acts as a catalyst. At high pressure, the metal surface gets saturated with gas molecules. So a further change in reaction conditions is unable to alter the amount of ammonia on the surface of the catalyst making the rate of the reaction independent of its concentration. Q. What are zero order reactions? |
| Answer» Solution :Those reaction in which the RATE is not DEPENDENT on any concentration TERM are CALLED zeroorder reaction. | |
| 47. |
Read the givenpassage ansanswer question number1 to 5 that follow : Amino acids are classified as acidic,baisc orneutral dependingunpon the relative numberofmakes it neutral , morenumberof amino than carboxyl groups makes it basic and more carboxyl groups are compared to amino groups makes it acidic . Theamino acids ,which can be synthesised in the body and mustbe obtained through diet, areknown as essential amino acids. Amino acids areusually colourless, crystalline solids. Theseare water-solute, high melting solids and behavelikesalts rather than simple amines or carboxylicacids . This behaviouris dueto the presence ofboth acidic (carboxyl group) and basic (amino group) in the samemlecule . In aqueous solution , the carboxyl group can losea proton and amino group can accepta proton , giving rise to a dipolarion known as zwitter ion. This is neutral but contains both positiveandnegative charges. In zwitter ionic , form amino acids show amphoteric behaviouras they reactboth with acids and bases. Whatare the different classes ofamino acids ? |
| Answer» SOLUTION :ACIDIC, basicand neturalamino ACIDS. | |
| 48. |
Read the given passage and answer the questions: Zero order reactions are relatively uncommon but they occur under special conditions. Some enzyme catalysed reactions and reactions which occurs on metal surfaces are a few examples of zero order reactions. The decompositions which occurs on metal surfaces are a few examples of zero order reactions. The decomposition of gaseous ammonia on a hot platinum surface is a zero order reaction at high pressure. 2NH_(3)(g) underset("Pt catalyst") overset(1130K)to N_(2)(g)+3H_(2)(g) Rate=k[NH_(3)]^(0)=k In this reaction, Pt acts as a catalyst. At high pressure, the metal surface gets saturated with gas molecules. So a further change in reaction conditions is unable to alter the amount of ammonia on the surface of the catalyst making the rate of the reaction independent of its concentration. Q. Why is the decomposition of NH_(3) on Pt surface a zero order reaction? |
| Answer» Solution :In this reaction, Pt acts as a catalyst. At high temperature, the METAL surface gets SATURATED with gas molecules. So, a further change in reaction conditions is unable to alter the AMOUNT of AMMONIA on the surface of platinum catalyst. Thus the reaction remains independent of its concentration. | |
| 49. |
Read the given passage and answer the questions: Zero order reactions are relatively uncommon but they occur under special conditions. Some enzyme catalysed reactions and reactions which occurs on metal surfaces are a few examples of zero order reactions. The decompositions which occurs on metal surfaces are a few examples of zero order reactions. The decomposition of gaseous ammonia on a hot platinum surface is a zero order reaction at high pressure. 2NH_(3)(g) underset("Pt catalyst") overset(1130K)to N_(2)(g)+3H_(2)(g) Rate=k[NH_(3)]^(0)=k In this reaction, Pt acts as a catalyst. At high pressure, the metal surface gets saturated with gas molecules. So a further change in reaction conditions is unable to alter the amount of ammonia on the surface of the catalyst making the rate of the reaction independent of its concentration. Q. If [R]_(0) and [R] are the concentrations of the reactant initially and after time t, give the equationrelating k. |
| Answer» SOLUTION :`K=([R]_(0)-[R])/(t)` | |
| 50. |
Read the given passage and answer the questions: Zero order reactions are relatively uncommon but they occur under special conditions. Some enzyme catalysed reactions and reactions which occurs on metal surfaces are a few examples of zero order reactions. The decompositions which occurs on metal surfaces are a few examples of zero order reactions. The decomposition of gaseous ammonia on a hot platinum surface is a zero order reaction at high pressure. 2NH_(3)(g) underset("Pt catalyst") overset(1130K)to N_(2)(g)+3H_(2)(g) Rate=k[NH_(3)]^(0)=k In this reaction, Pt acts as a catalyst. At high pressure, the metal surface gets saturated with gas molecules. So a further change in reaction conditions is unable to alter the amount of ammonia on the surface of the catalyst making the rate of the reaction independent of its concentration. Q. Give the shape of the graph between concentration of reaction vs time in a zero order reaction. |
Answer» SOLUTION :
|
|