Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Red and white Phosphorous will differ but not in |
|
Answer» smell |
|
| 2. |
Rectified spirit contains : |
|
Answer» `75.0%` ALCOHOL |
|
| 3. |
Rectified spirit is denatured by adding |
|
Answer» methyl alcohol and formic acid |
|
| 4. |
Rectified spirit is made unsuitable for drinking by adding |
| Answer» Answer :A | |
| 5. |
Rectified spirit is a mixture of: |
|
Answer» 95% ETHYL ALCOHOL + 5% water |
|
| 6. |
Rectified spirit contains 95% ethanol by mass. The mole fraction of ethanol will be |
|
Answer» 0.881 `X_(2)=n^(2)/(n_(2) + n_(1))=(W_(2)//M_(2))/((W_(2)//M_(2))+(W_(1)//M_(1)))` `x_(2)=(95//46)/(95//46 + 5//18)=2.065/(2.065+0.278)=2.065/2.343=0.881` |
|
| 7. |
Rectifiedspirit can be dried with : |
|
Answer» conc. `H_(2)SO_(4)` |
|
| 8. |
Rectified spirit can be converted into absolute alcohol by distilling with |
|
Answer» `Na_2CO_3` |
|
| 9. |
Recharging of lead storage battery involves |
|
Answer» ANODE is REDUCED to pb |
|
| 10. |
Receptors of hormones are generally |
|
Answer» CARBOHYDRATES |
|
| 12. |
Recent X-ray work, IR and other spectroscopic methods have proved that Turnbull's blue is identical to Prussian blue. Q. Intense blue colour arises as a result of: |
|
Answer» ELECTRON TRANSFER between `FE(II) and Fe(I)` |
|
| 13. |
Receptors are |
|
Answer» Proteins |
|
| 14. |
Recently discovered allotropes of carbon is ……………….. |
|
Answer» GRAPHITE |
|
| 15. |
Recent X-ray work, IR and other spectroscopic methods have proved that Turnbull's blue is identical to Prussian blue. Q. What is the common formula of Turnbull's blue and Prussian blue? |
|
Answer» `Fe_(3)[FE(CN)_(6)]_(2)` |
|
| 16. |
Recent method for the charaterisation of an organic compound is : |
|
Answer» VACUUM distillation |
|
| 17. |
Recemic mixture is obtained due to the halogenation of |
|
Answer» isopentane  . .
|
|
| 18. |
Reason of lanthanoid contraction is….. |
|
Answer» NEGLIGIBLE SCREENING EFFECT of f-orbitals |
|
| 19. |
Reason of lanthanoid contraction is |
|
Answer» Decreasing SCREENING effect |
|
| 20. |
Reason of lanthanoid contraction is - |
|
Answer» negligible screening effect of F - orbitals |
|
| 21. |
Reason of lanthanodi contraction is |
|
Answer» NEGLIGIBLE screening effect of 'F' orbitals |
|
| 22. |
Reason for geometrical isomerism shown by 2- butene is |
|
Answer» Chiral carbon |
|
| 23. |
Reason for excessive solubility of alcohol in water is due to |
|
Answer» COVALENT BONDING |
|
| 24. |
Rearrange the following in an increasing order of their basic strengths : ""C_(6)H_(5)NH_(2), C_(6)H_(5)N(CH_(3))_(2), (C_(6)H_(5))_(2)NH" and "CH_(3)NH_(2) |
|
Answer» `""(C_(6)H_(5))_(2)NH lt C_(6)H_(5)N(CH_(3))_(2) ltC_(6)H_(5)NH_(2) lt CH_(3)NH_(2)` |
|
| 25. |
Reason: Complex anion [Re_(2)Cl_(8)]^(2-) has one delta-bond, one sigma and one pi-bond. Reason: d_(z^(2)) orbital can never form delta-bond. |
|
Answer» If ASSERTION is true but the REASON is false 
|
|
| 26. |
Rearrange the following in an increasing order of their basi strengths: C_(6)H_(5)NH_(2),C_(6)H_(5)N(CH_(3))_(2),(C_(6)H_(5))_(2)NH and CH_(3)NH_(2). |
|
Answer» Solution :(i) Due to delocalization of lone pair of electrons of the N-atom over the BENZENE ring, all aromatic amines are less basic than alkylamines, i.e., `CH_(3)NH_(2)`. (ii) PRESENCE of electron donating grous (i.e., `CH_(3)`) on the N-atom increases the basicity of substituted aniline w.r.t. `C_(6)H_(5)NH_(2)`. (iii) In `(C_(6)H_(5))_(2)NH`, th lone pair of electrons on the N-atom is delocalized over two benzene rings instead of one in `C_(6)H_(5)NH_(2)`, therefore, `(C_(6)H_(5))_(2)NH` is much less basic than `C_(6)H_(5)NH_(2)`. Combining all the three trends together, the basicitiy of the four amines increases in the ORDER: `(C_(6)H_(5))_(2)NH lt C_(6)H_(5)NH_(2) lt C_(6)H_(5)N(CH_(3))_(2) lt CH_(3)NH_(2)`. |
|
| 27. |
Rearrange the following compounds in the increasing order of their boiling points : CH_(3)-CHO, CH_(3)-CH_(2)-OH, CH_(3)-CH_(2)-CH_(3) |
|
Answer» Solution :The compounds MAY be REARRANGED in increasing ORDER of theirboiling points as under : `CH_(3)CH_(2)CH_(3) tl CH_(3)CHO LT CH_(3)CH_(2)OH` |
|
| 28. |
Rearrange the following compounds in increasing order of their boilig ponits: CH_(3)-CHO,CH_(3)CH_(2)-OH,CH_(3)-CH_(2)-CH_(3). |
|
Answer» Solution :Due to EXTENSIVE intermolecular H-bonding, the b.p. of `CH_(3)CH_(2)OH` is the highest. Now `CH_(3)CHO` is polar while `CH_(3)CH_(2)CH_(3)` is non-polar. THEREFORE, due to dipole-dipole interactions, the b.p. of `CH_(3)CHO` is higher than that of `CH_(3)CH_(2)CH_(3)`. thus, the b.ps. of the three compounds INCREASE in the order: `CH_(3)CH_(2)CH_(3) LT CH_(3)CHO lt CH_(3)CH_(2)OH`. |
|
| 29. |
Rearrange the following compounds in an increasing order of their basic strengths : ""Aniline, p-nitroaniline, p-toluidine |
|
Answer» |
|
| 30. |
Rearrange the compounds of each of the following sets in order of reactivity towards S_(N_(3)^(2)) displacement : (i) 2- Bromo-2-methylbutane, 1-Bromopentane, 2- Bromopentaure. (ii) 1- Bromo-3-methylbutance, 2-Bromo-2-methylbutance,3-Bromo-2- methybutane. (iii)1- Bromobutane, 1- Bromo-2, 2-dimethylbutane 1-Bromo -2- methylbutane. |
|
Answer» Solution :(i) 2-Bromo-2-methybutane, 1-Bromopentane , 2Bromopentane: The reactivity is `SN^(2)` reaction depents uponsteric hindance, more thesteric hindence slower the reaction.  Sincedue to stericreasionthe order of reactivityin `SN^(2)`reactionfollowsthe order: `1^(@) gt2^(@) GT3^(@)`, therefore, order of reactivityofthe givenalklyl BROMIDES is : 1-Bromopentane `gt 2`- Brompebtane `gt 2-` Bromo -2 Methylbutane. 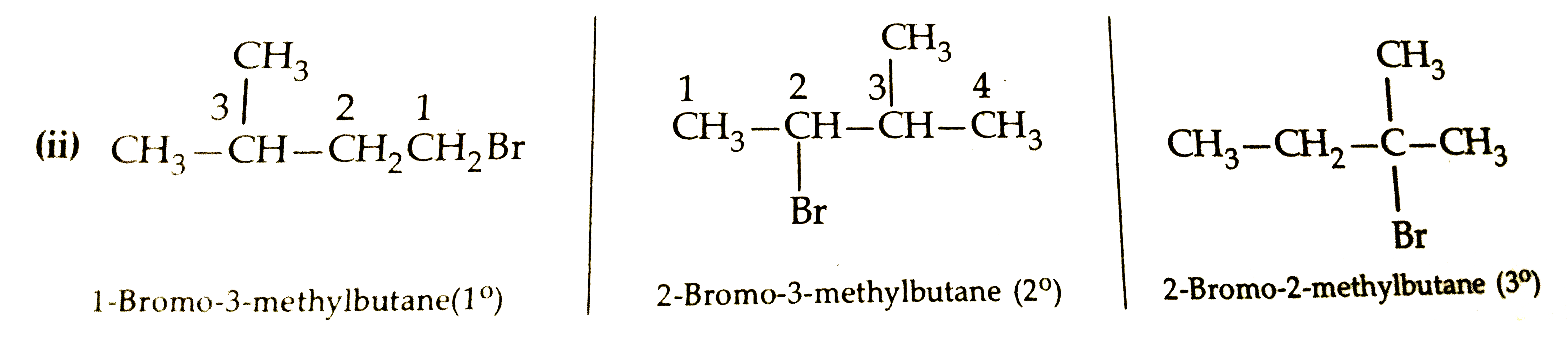 Since due to steric reason, the order of reactivity of alkyl halides in `SN^(2)` reactionfollows the order `1^(@) gt 2^(@) gt 3^(@)` therefore, the order ofreactivity of thegivenalkyl bromies is . 1-Bromo-3-Methylbutane`gt2`- Bromo -3-Methylbutane `gt 2`-Bromo -2-Methylbutane. 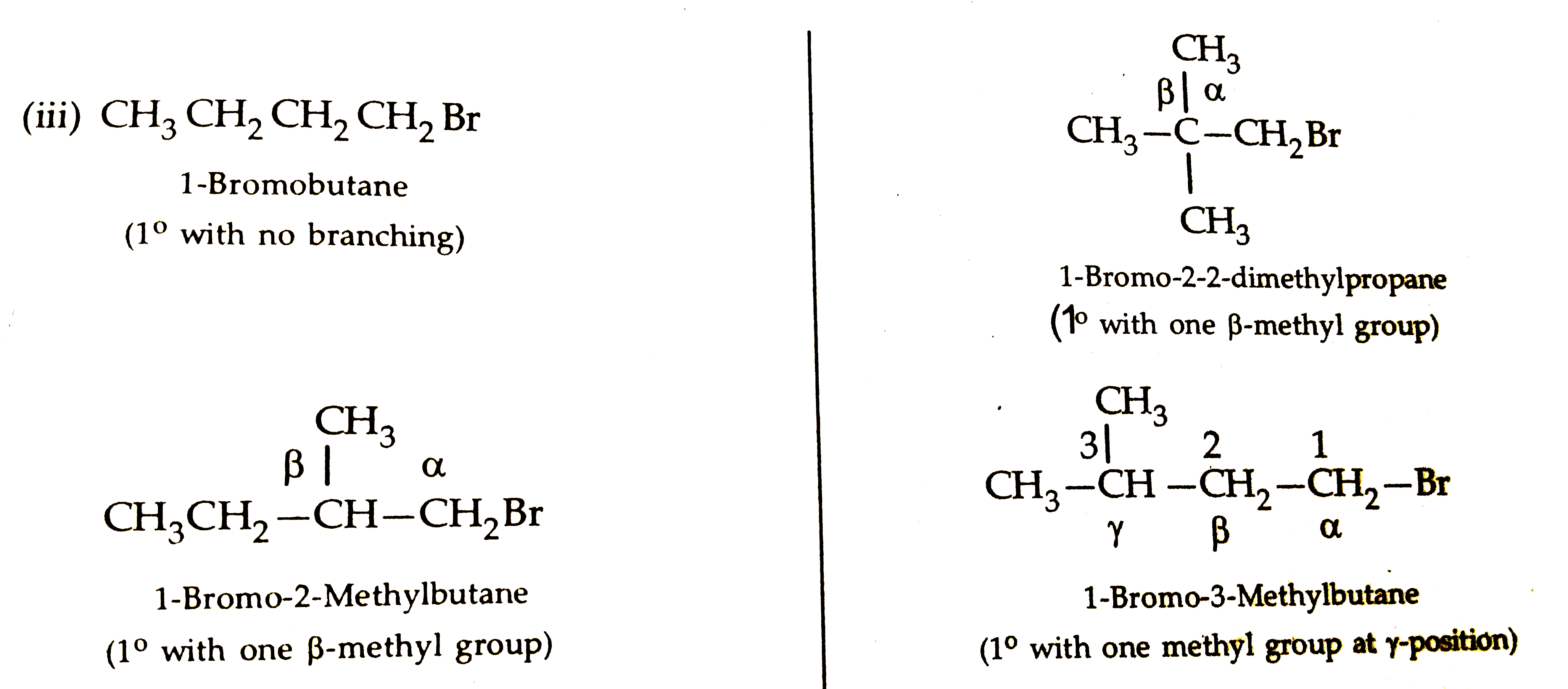 Since in CASE of `1^(@)` alkyl halides,steric hindrancein theorder : n-alkyl halides, alkyl halide substituentsatthe `beta`-position, therefore , the reactivitydecrease in the sameorder- Thu, the reactivityof the givenalkyl bromides decrease in the order. 1-Bromobutance `gt 1`- Bromo - 3-Methybutane `gt 1`- Bromo-2- Methylbutane `GT1`- Bromo-2-Methylbutane`gt1`- Bromo-2-dimethylpropane. |
|
| 31. |
Rearrange the compounds of each of the following sets in order of reactivity towards S_(N)2displacement : (i) 2-Bromo-2-methylbutane, 1-Bromopentane, 2 Bromopentane (ii) 1-Bromo-3-methylbutane, 2-Bromo-2 methylbutane, 3-Bromo-2-methylbutane (iii)1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane |
|
Answer» Solution :(i) `UNDERSET("TERTIARY halogencompound")(CH_(3) 0 CH_(2) - underset(CH_(3))underset(|)overset(Br)overset(|)C - CH_(3)) "" underset("Primary halogen compound ")(CH_(3)CH_(2)CH_(2)CH_(2)CH_(2)Br) "" underset("Secondary halogen compound")(CH_(3)CH_(2)CH_(2)overset(Br)overset(|)CHCH_(3))` In `S_(N)2` reaction , the order of reactivity is: Primary ` gt` Secondary ` gt` Tertiary . Therefore, the order of reactivity for the given COMPOUNDS will be 1-Bromopentane `gt`2-Bromopentane `gt`2-Bromo-2-methylbutane (ii) `underset("Primary halide")(CH_(3) - overset(CH_(3))overset(|)(CH) - cH_(2) - CH_(2)Br ) "" underset("Tertiary halide")(CH_(3) - CH_(2) - underset(CH_(3))underset(|)overset(Br)overset(|)C - CH_(3)) "" underset("Secondary halide")(CH_(3) - overset(Br)overset(|)CH - underset(CH_(3))underset(|)CH - CH_(3))` Therefore , the order of reactivitywill be 1-Bromo-3-methylbutane `gt`3-Bromo-2-methylbutane `gt ` 2-Bromo-2-methylbutane (iii) `underset("Primary halide (I)")(CH_(3) - CH_(2) - CH_(2) - CH_(2)Br) "" underset(II) (CH_(3) - underset(CH_(3))underset(|)overset(CH_(3))overset(|)C - CH_(2)Br) "" underset(III)(CH_(3) - CH_(2) - overset(CH_(3))overset(|)(CH) - CH_(2)Br)` All the three are primary halide. But the NUCLEOPHILE will face maximum hinderance with II followed by III followed by I. Hence, the order of reactivity towards `S_(N)2`is: 1-Bromobutane `gt`1-Bromo-2-methylbutane `gt` 1-Bromo-2,2-dimethylpropane. |
|
| 32. |
Real ggas show same behaviour as that of an ideal gas at |
|
Answer» LOW TEMPERATURE and HIGH pressure |
|
| 33. |
Real gases show an ideal-like behaviour at |
|
Answer» HIGH PRESSURE and LOW temperatures |
|
| 34. |
Reagents which cannot be used to distinguish Allylbromide from n - propyl bromide are |
|
Answer» `Br_2//"CCl"_4` b) Shaking with `AgNO_3` - Both can react with` AgNO_3`and can form AgBr an yellow ppt c) Boiling with alcoholic KOH, followed by acidification with `HNO_3`and addition of `AgNO_3` gives same product of yellow ppt of AgBr d) Fusion with sodium metal followed by acidification with dil `HNO_3`and addition of `AgNO_3`gives an yellow ppt of AgBr |
|
| 35. |
Reagents which cannot be used to distinguish allyl bromide from n-propyl bromide are |
|
Answer» `Br_(2)//C Cl_(4`<BR>by SHAKING with an aqueous solution of `AgNO_(3)` |
|
| 36. |
Reagents used in hydroboration oxidation reaction |
|
Answer» `B_2H_6+CrO_3 + NAOH` |
|
| 37. |
Reagent which cannot be used to distinguish allyl bromide from n-propyl bromide are: |
|
Answer» `Br_(2)//C Cl_(4)` |
|
| 39. |
Reagent used tp detect sulphate ion is ……………… . |
|
Answer» `BaCl_(2)` |
|
| 40. |
Reagent used to distinguish H_2O_2 and O_3 is: |
|
Answer» `PBS` |
|
| 41. |
Reagent used to carry out following conversion from alkyne to alkene is |
|
Answer» `Pd-C//H_(2)` |
|
| 42. |
Reagent used in Reimer -Tiemann reaction are |
|
Answer» `CH_3Cl` and aq.NaOH |
|
| 43. |
Reagent used in Finkestein reaction is |
|
Answer» NAI in DRY acetone |
|
| 44. |
Reagent used for detecting CHCI_3 is |
|
Answer» `AQ. AgNO_3` |
|
| 45. |
Reagent that candetect any of Fe^(3+),Co^(2+) and Cu^(2+) is (a) ____. |
|
Answer» |
|
| 46. |
Reagent required to convert B to A is |
|
Answer» `K_(2) Cr_(2) O_7 //H^(+)` |
|
| 47. |
Reagent (s) used for the conversion of propanol into propane is |
|
Answer» `I_2//P` |
|
| 48. |
Reagent A used in this change is |
|
Answer» `B_(2)H_(6)` |
|