Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Nuclides |
|
Answer» Have specific ATOMIC NUMBER |
|
| 2. |
Nuclic acids are called acids mainly because of the presence of |
|
Answer» `-COOH` group |
|
| 3. |
Nucleus of an element contains 9 protons Its valency would be : |
|
Answer» 1 |
|
| 4. |
Nucleus of deuterium contains: |
|
Answer» ONE PROTON and One neutron |
|
| 5. |
Nucleus model of the atom was proposed by: |
|
Answer» Thomson |
|
| 6. |
Nucleosides are joined together by phosphodiester linkage between 5' and 3' carbon atoms of pentose sugar. |
|
Answer» |
|
| 7. |
Nucleotides and nucleosides mainly differ from each other in : |
|
Answer» PRESENCE of PHOSPHATE units |
|
| 8. |
Nucleoside on hydrolysis gives |
|
Answer» PENTOSE SUGAR and PURINE base |
|
| 9. |
Nucleoside on hyd rolysis gives |
|
Answer» an ALDOPENTOSE and HETEROCYCLIC BASE |
|
| 10. |
Nucleoside of DNA contain |
|
Answer» `BETA`-D-ribose SUGAR and PHOSPHORIC ACID |
|
| 11. |
Nucleoside is composed of……………………. |
|
Answer» SUGAR+ BASE + PHOSPHATE |
|
| 12. |
Nucleoside involvesthecombination of |
|
Answer» SUGAR + BASE +`H_3PO_4` |
|
| 13. |
Nucleoside consist of |
|
Answer» SUGAR and `H_3PO_4` |
|
| 14. |
Nucleoprotein is a _____ type protein. |
|
Answer» |
|
| 15. |
Nucleophilicity order is correctly represented by |
|
Answer» `CH_(3)^(-) lt NH_(2)^(-) lt HO^(-) lt F^(-)` |
|
| 16. |
Nucleophilic substitution of primary alkyl chlorides with sodium acetate is catalysed by sodium iodide. Discuss |
|
Answer» Solution :Primary lakyl chlorides `(R-CH_(2)-Cl)` are less reactive towards nucleophilic substitution as compared to primary alkyl IODIDES `(R-CH_(2)-I)` because C-I bond can cleave moreasily that `C-Cl` bond o accout of less bond dissociation enthalpy. Therefore, the catalyst sodium iodide CONVERTS alkyl chloride to alkyl iodide by finkelstin REACTION. the latter react with sodium acetate to form ester. `UNDERSET("ETHYL chloride")(CH_(3)-CH_(2)-)Cl+NaItounderset("Ethyl iodide")(CH_(3)-CH_(2)-)I+NaCl` `underset("Ethyl iodide")(CH_(3)-CH_(2)I+CH_(3)COONa)tounderset("Ethyl acetate")(CH_(3)COOC_(2)H_(5)+NaI` |
|
| 17. |
Nucleophilic substitution reaction of optically active halide, PIC is accompanied by |
|
Answer» INVERSION of configuration |
|
| 18. |
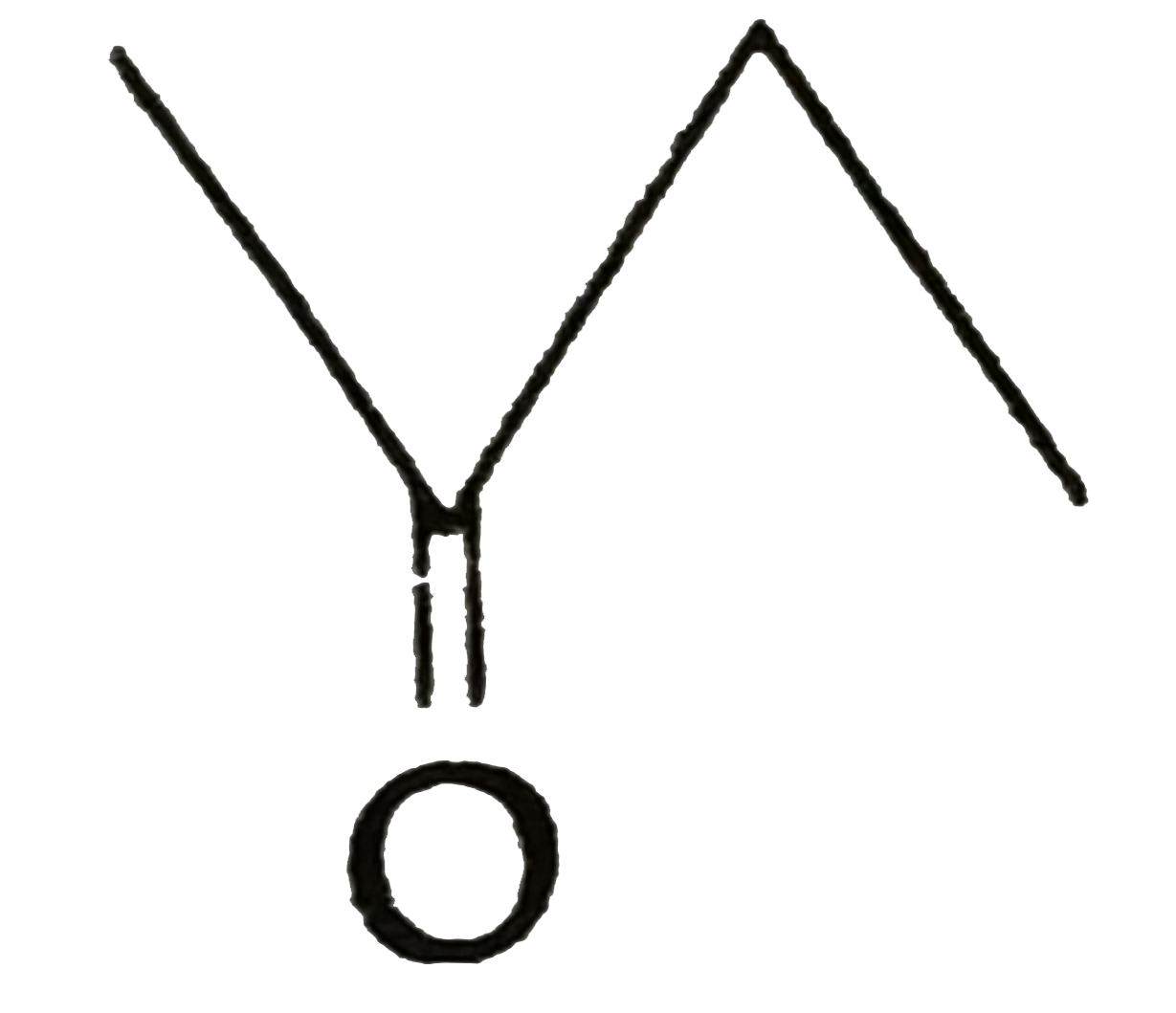
Nucleophilic attack on carbonyl carbon changed its hydridisation from |
|
Answer» SP to `sp^(2)` |
|
| 19. |
Nucleophilic substitution in aryl halides is facilitated by electron withdrawing groups while electrophilic substitution is facilitated by electron releasing groups. Why? |
| Answer» Solution :Nucleophilic substitution in aryl halides takes place VIA carbanion as intermediate, which are stabilised by electron WITHDRAWING groups. Electrophilic substitution takes place through carbocations as intermediates, which are stabilised by electron releasing groups. The rate of the REACTION INCREASES if the intermediate INVOLVED in this reaction is stabilised. | |
| 20. |
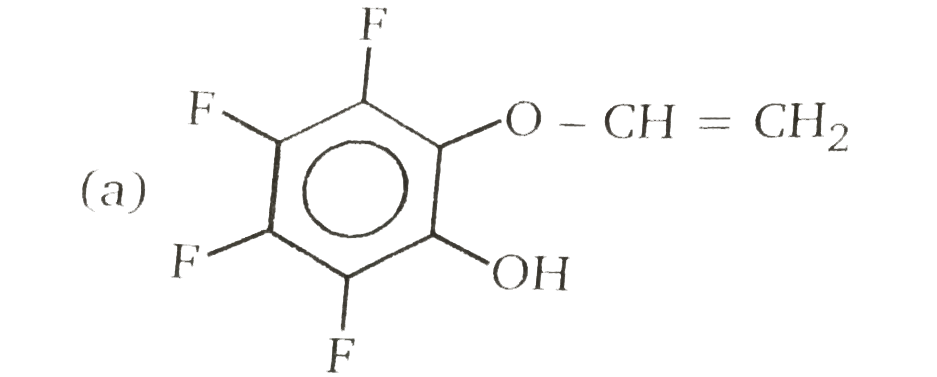
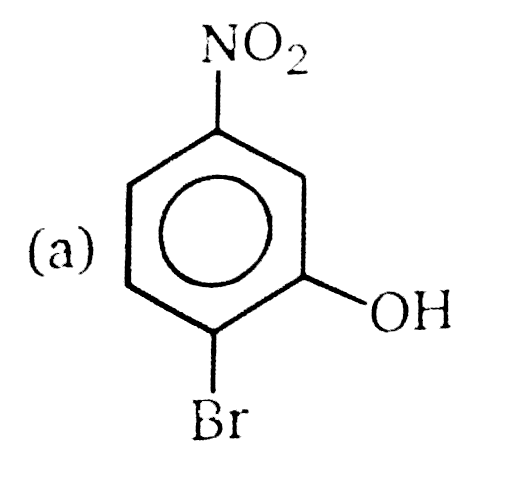
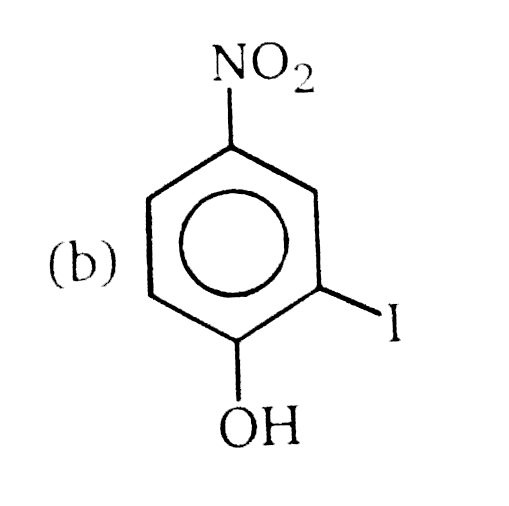
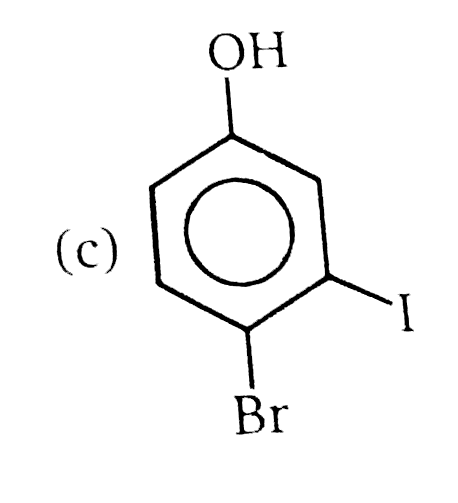
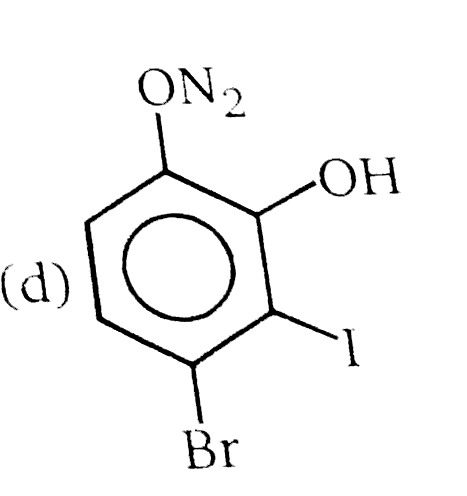
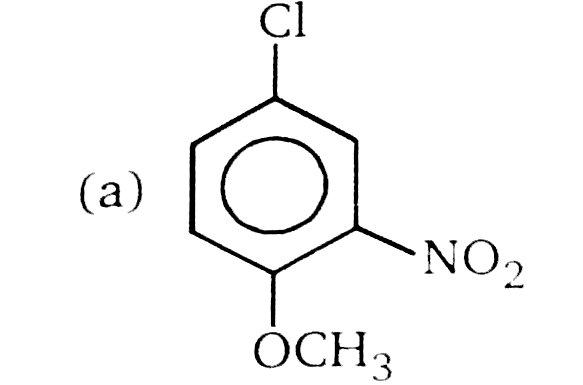
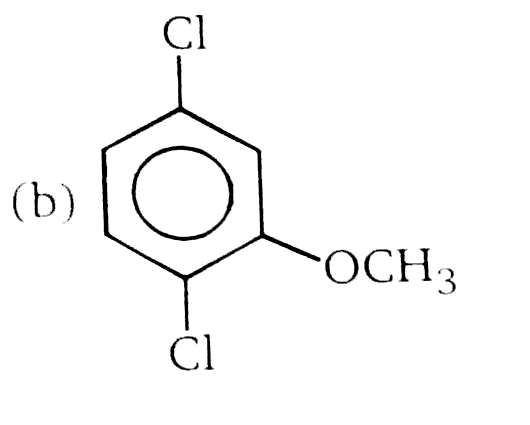
Nucleophilic Aromatic substitution (SN_(Ar)): A substituted benzene derivative containing - NO_(2) and Cl group at p-position is subjected to Nu- substitution The cumulative effect of their fluorine active the rings of penta and hexa fluorobenzene toward nucleophilic aromatic substitution. What is compound X in the following synthesis ? |
|
Answer»
|
|
| 21. |
Nucleophilic Aromatic substitution (SN_(Ar)): A substituted benzene derivative containing - NO_(2) and Cl group at p-position is subjected to Nu- substitution Which is the best route for the synthesis of Strating from benzen of ? |
|
Answer» `underset(FeBr_(3))overset(Br_(2))rarr underset(H_(2)SO_(4))overset(HNO_(3))rarr underset(H_(2)SO_(4))overset(HNO_(3))rarr underset(CH_(3)OH)overset(NaOCH_(3))rarr`<BR>`underset(H_(2)SO_(4))overset(HNO_(3))rarr underset(H_(2)SO_(4))overset(HNO_(3))rarr underset(FeBr_(3))overset(Br_(2))rarr underset(CH_(3)OH)overset(NaOCH_(3))rarr` |
|
| 22. |
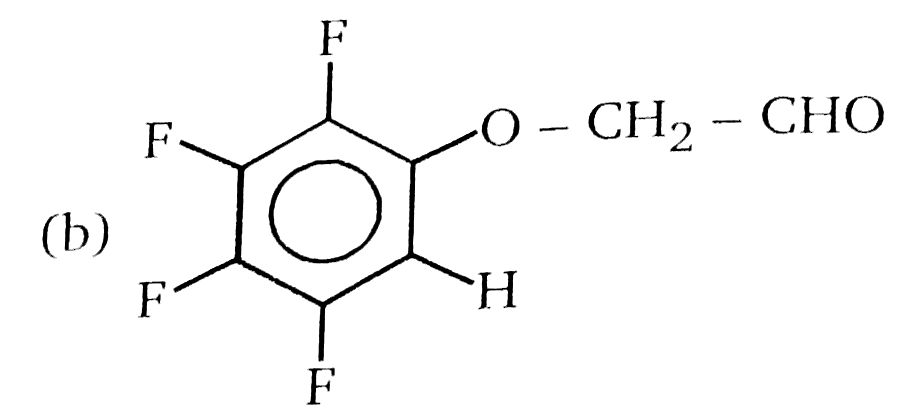
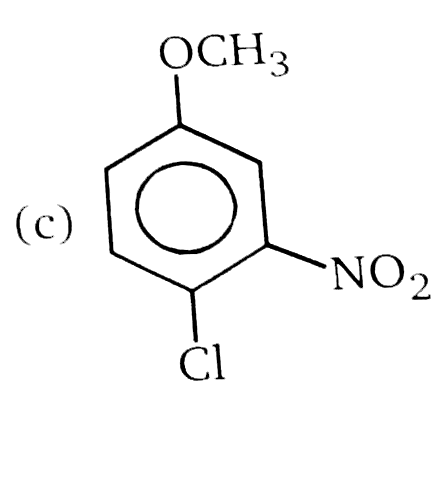
Nucleophilic Aromatic substitution (SN_(Ar)): A substituted benzene derivative containing - NO_(2) and Cl group at p-position is subjected to Nu- substitution , Product (A) is: |
|
Answer» <P> |
|
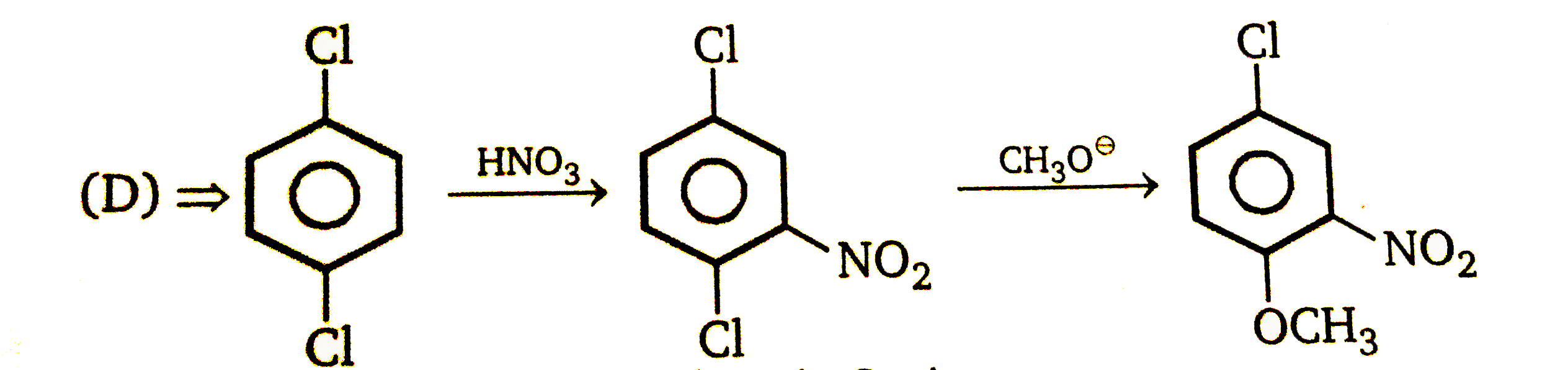
| 23. |
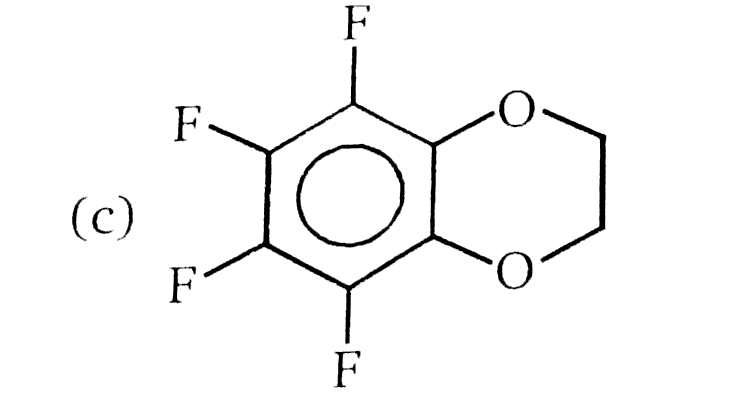
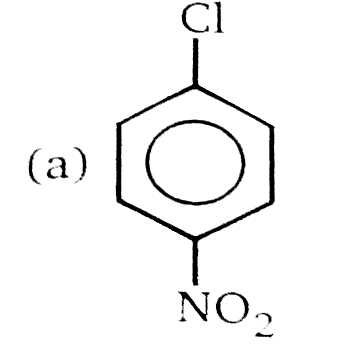
Nucleophilic Aromatic substitution (SN_(Ar)): A substituted benzene derivative containing - NO_(2) and Cl group at p-position is subjected to Nu- substitution , Product (B) is: |
|
Answer»
|
|
| 24. |
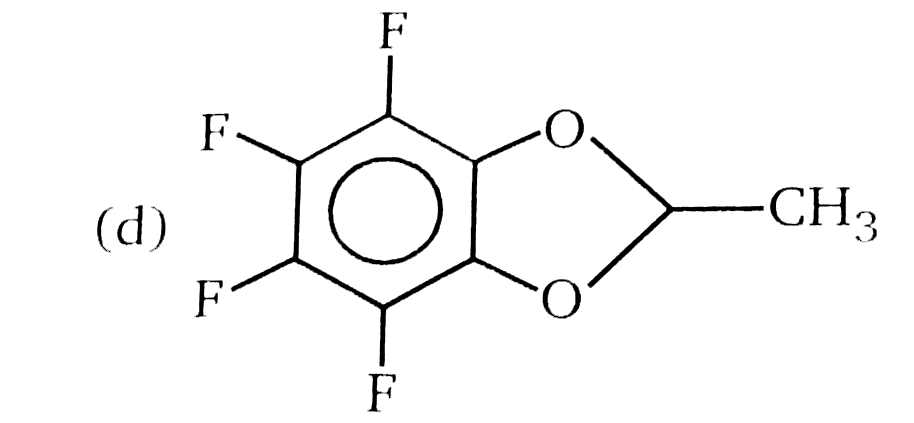
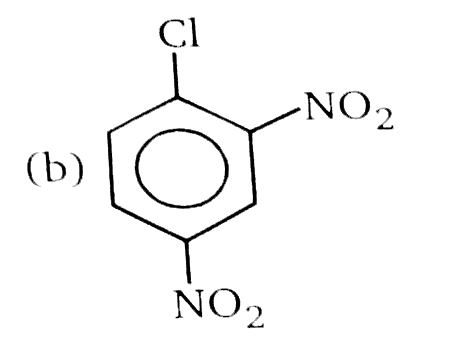
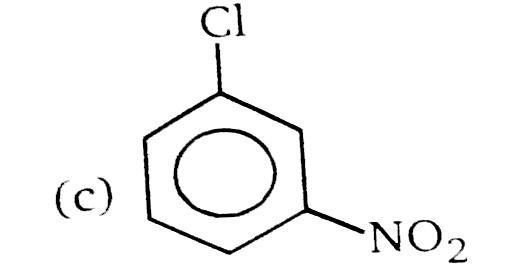
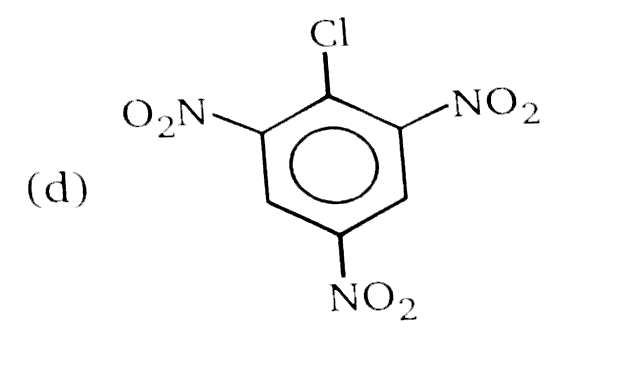
Nucleophilic Aromatic substitution (SN_(Ar)): A substituted benzene derivative containing - NO_(2) and Cl group at p-position is subjected to Nu- substitution Which of the following is most reactive towards SN_(Ar) |
|
Answer» <P> |
|
| 25. |
Nucleophilic Aromatic substitution (SN_(Ar)): A substituted benzene derivative containing - NO_(2) and Cl group at p-position is subjected to Nu- substitution Match the column I and II {:(,"Column (I)",,"Column (II)",),(,X = "halogen",,"relative reactivity toward" (SN_(Ar)),),((a),-F,(p),312,),((b),-Cl,(q),1,),((c),-Br,(r),0.8,),((d),-I,(s),0.6,):} |
|
Answer» |
|
| 26. |
Nucleophilic Aromatic substitution (SN_(Ar)): A substituted benzene derivative containing - NO_(2) and Cl group at p-position is subjected to Nu- substitution If step -2 were rate determining step, which halogen of aryl halide is most reactive towards SN_(Ar). |
|
Answer» Fluoride |
|
| 27. |
Nucleophilic addition of Grignard reagent to aldehydes/ketones take place in the presence of ……………………. followed by acid hydrolysis gives …………………... |
| Answer» SOLUTION :DRY ETHER, ALCOHOL | |
| 28. |
Nucleophilic addition reaction is mostfavored in |
|
Answer»
|
|
| 30. |
Nucleophiles are |
|
Answer» `H_(2)` O |
|
| 31. |
Nucleic acids were first discovered by |
| Answer» Solution :NUCLEIC ACIDS were FIRST discovered by meischer. | |
| 33. |
Nucleic acids are the polymers of ............ |
|
Answer» Nucleotides |
|
| 34. |
Nucleic acids contain : |
|
Answer» 4 purine bases |
|
| 35. |
Nucleic acids are the polymers of |
|
Answer» nucleosides |
|
| 36. |
Nucleic acids are the polymers of ……….. |
|
Answer» NUCLEOSIDES |
|
| 37. |
Nucleic acid is made up of _______. |
|
Answer» an ORGANIC BASE |
|
| 38. |
Nucleic acid is a polymer of…………. . |
|
Answer» NUCLEOSIDES |
|
| 39. |
Nuclei of isotopes differ in |
|
Answer» The NUMBER of protons |
|
| 40. |
Nuclei of a radioactive element 'A' are being produced at a constant rate alpha the element has a decay constant lambda At time t=0 there are N_(0) nuclei of the element If alpha = 4N_(0) lambda the number of nuclei of A when A has reached steady state: |
|
Answer» 0 |
|
| 41. |
Nuclei of a radioactive element 'A' are being produced at a constant rate alpha the element has a decay constant lambda At time t=0 there are N_(0) nuclei of the element If alpha=2N_(0)lambdathe number of nuclei of A afterundergone radioactivity decay till time t=(l n2) / lambda: |
|
Answer» `0.5N_(0)` |
|
| 42. |
Nuclei of a radioactive element 'A' are being produced at a constant rate alpha the element has a decay constant lambda At time t=0 there are N_(0) nuclei of the element The number of nuclei of A at time 't' is |
|
Answer» `(ALPHA)/(lambda)(1-e^(-lambdat))` |
|
| 43. |
Nuclei of a radioactive element 'A' are being produced at a constant rate alpha the element has a decay constant lambda At time t=0 there are N_(0) nuclei of the element If alpha=2N_(0)lambdathe number of nuclei of A after t=(l n2)/lambda will become: |
|
Answer» ZERO |
|
| 45. |
Nuclear theory of the atom was put forward by: |
|
Answer» RUTHERFORD |
|
| 46. |
Nuclear theory of the atom was put forward by |
|
Answer» RUTHERFORD |
|
| 47. |
Nuclear reactivity of Na and Na^(+) is same because both have |
|
Answer» Same ELECTRON and proton |
|
| 48. |
Nuclear reactions accompanied with emission of neutron (s) are |
|
Answer» `._(13)^(27)Al + ._(2)He^(4) rarr ._(15)^(30)P` (b) `._(96)Am^(241) + ._(2)He^(4) rarr ._(97)BK^(244) + ._(1)e^(0) + ._(0)n^(1)` |
|
| 49. |
Nuclear reactions accompared with emission of neutron are : |
|
Answer» `""_(13)^(27) Al + ""_(2)^(4) He to ""_(15)^(30) P` |
|
| 50. |
Nuclear reactions accompained with emission of neutron(s) are |
|
Answer» `""_(13)^(27)Al + ""_(2)^(4)He to ""_(15)^(30)P` `""_(96)^(241)Cm + ""_(2)^(4)He to ""_(97)^(244)Bk + ""_(+1)^(0)e + ""_(0)^(1)n`. |
|