Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is correct name according to IUPACnomenclature ? |
|
Answer» 2-Hydroxy-5-methyl CYCLOHEXANOL |
|
| 2. |
Which compound is formed when excess KCN is added to an aqueous solution of copper sulphate? |
|
Answer» `CU(CN)_(2)` |
|
| 3. |
Which gas has the same rate of diffusion as that of CO_2 at same P and T ? |
|
Answer» `N_2O` |
|
| 4. |
Which of the following acids is the strongest ? |
|
Answer» `HCN(K_(a)=4xx10^(-10))` |
|
| 5. |
Which is not correct |
|
Answer» Phenol is more acidic than ACETIC acid Ethanol has higher boiling point due to hydrogen bonding. Ethyne has linear shape due to sp-hybridisation . `K_(a)underset(RCOOH)(10^(-5))gt underset(H_(2)CO_(3))(10^(-7))gt underset(C_(6)H_(5)OH)(10^(-10))gt underset(HOH)(10^(-14))gt underset(ROH)(10^(-18))` |
|
| 6. |
Which one is not synthesized by Grignard reagent ? |
|
Answer» PRIMARY ALCOHOL |
|
| 7. |
What is the [OH^-] in an aqueous solution which has a pH =11.70 ? |
|
Answer» `7.1xx10^(-2) M` |
|
| 8. |
To prepare 2- propanol from CH_3Mgl, the other chemical required is : |
|
Answer» HCHO |
|
| 9. |
Which of the following molecule is capable of forming zwitter ion ? |
|
Answer» `CH_(3)-CHOH-NH_(2)` |
|
| 10. |
Which shows reducing properties |
|
Answer» `SO_3` |
|
| 11. |
Which is increasing order of the reducing power of the following metals on the basis of standard electrode potential ? Ag^(+)//Ag=0.80 V " "Mg^(2+)//Mg=-2.37V Hg^(2+)//Hg=0.79V" "Cr^(3+)//Cr=-0.74V |
|
Answer» `Hg LT Ag lt MG lt CR` |
|
| 13. |
Which of the following is the strongest reducing agent ? |
|
Answer» `PH_(3)` |
|
| 14. |
Which of the given statements is incorrect? |
|
Answer» Peroxide EFFECT proceeds via free radical chain mechanism |
|
| 15. |
Write the balanced equations in the manufacture of potassium dichromate from chromite ore. |
|
Answer» Solution :1. `4FeOCr_(2)O_(3) + 8Na_(2)CO_(3) + 7O_(2) rarr 8Na_(2)CrO_(4) + 2Fe_(2)O_(3) + 8CO_(2) uarr` 2. `2Na_(2)CrO_(4) + 2H^(+) rarr Na_(2)Cr_(2)O_(7) + 2Na^(+) + H_(2)O` or `2Na_(2)CrO_(4) + H_(2)SO_(4) rarr Na_(2)Cr_(2)O_(7) + Na_(2)SO_(4) + H_(2)O` 3. `Na_(2)Cr_(2)O_(7) + 2KCl rarr K_(2)Cr_(2)O_(7) + 2NaCl` equation for manufacture of potassium dichromate from chromite ORE : (i) Conversion of chromite into sodium CHROMATE : `underset("(Chromite ore)")(4FeO.Cr_(2)O_(3)) + 8Na_(2)CO_(3) + 7O_(2) rarr underset("(Sodium chromate)")(8Na_(2)CrO_(4)) + 2Fe_(2)O_(3) + 9CO_(2)` (ii) Conversion of sodium chromate into sodium dichromate : `underset("(Sodium chromate)")(2Na_(2)CrO_(4))+H_(2)SO_(4) rarr underset("(Sodium dichromate)")(Na_(2)Cr_(2)O_(7)) + Na_(2)SO_(4)+H_(2)O` (iii) Conversion of sodium dichromate into potassium dichromate : `Na_(2)cr_(2)O_(7) + 2KCl rarr underset("(Potassium dichromate)")(K_(2)Cr_(2)O_(7)) + 2NaCl` |
|
| 17. |
When compared to DeltaG^(@) for the formation of Al_(2)O_(3), the DeltaG^(@) for the formation of Cr_(2)O_(3) is |
|
Answer» same `Cr_(2)O_(3)+2Alto2Cr+Al_(2)O_(3)+`heat, THEREFORE `DeltaG^(o)` for the formation of `Al_(2)O_(3)` is lower in comparison to `DeltaG^(o)` for the formation of `Cr_(2)O_(3)`. |
|
| 18. |
what is IUPAC name of compound when imino group is attached to ethyl and n-propyl group ? |
|
Answer» N-Ethylisopropyl AMINE |
|
| 19. |
The solubility of PbCL_2 is given by, |
|
Answer» `sqrtK_(SP)` |
|
| 20. |
When reaction is at standard state at equilibrium , then: |
|
Answer» `DELTAH^(@) =0` |
|
| 21. |
What is the activation energy for a reaction if the rate is doubled when the temperature is raised from 20^(@) C to 35^(@)C (R=8.314 J mol^(-1)K^(-1)) |
|
Answer» 15.1 kJ `MOL^(-1)` `T_(2)=35 + 273=308`K `k=8.314 JK^(-1)mol^(-1)` `log k_(2)/k_(1) =E_(a)/(2.303 ) [1/T_(1)-1/T_(2)]` `log2= (E_(a))/(2.303 xx 8.314(JK^(-1)mol^(-1))[1/(293K)-1/(308K)]` `0.3010= (E_(a) xx 15)/(2.303 xx 8.314 (Jmol^(-1)) xx 293 xx 308)` `E_(a) = (0.3010 xx 2.303 xx 8.314 x 293 xx 308)/(15) (Jmol^(-1))` `=34.673 J mol^(-1) = 34.673 kJ mol^(-1)` |
|
| 22. |
What is meant by 'limiting molar conductivity ? |
| Answer» SOLUTION :It is MOLAR CONDUCTIVITY at INFINITE DILUTION. | |
| 23. |
Which of the following statement is not correct for nitrogen ? |
|
Answer» Its electronegativity is very high `N(7)= 1s^(2) , 2s^(2)2p^(3)` 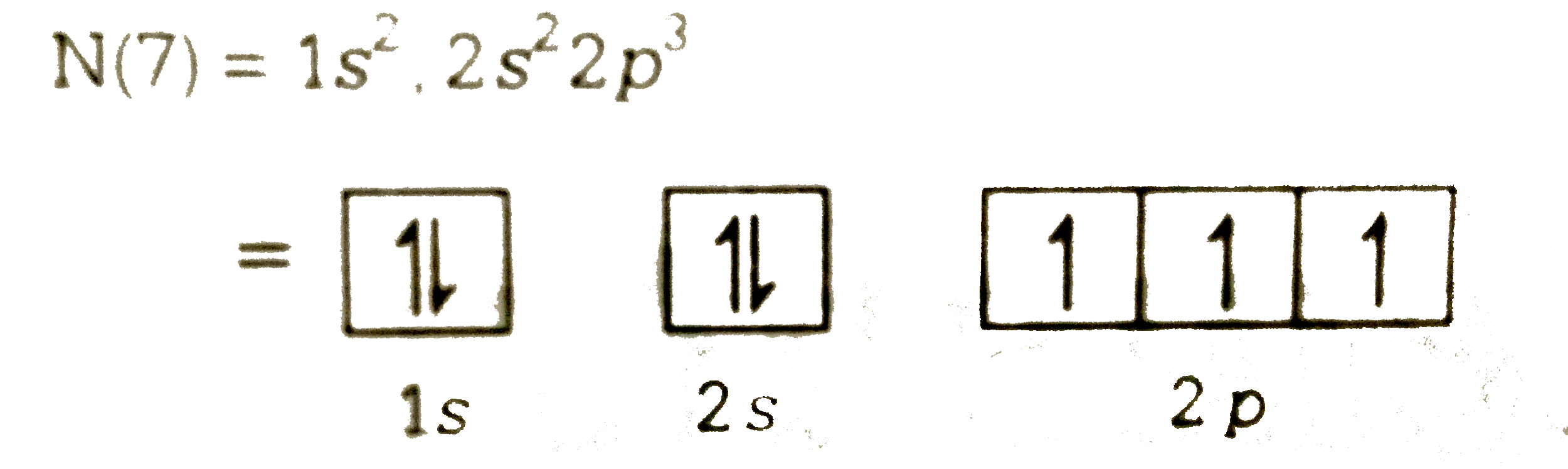
|
|
| 24. |
Which of the following biomolecules is insoluble in water |
|
Answer» `alpha`-KERATIN |
|
| 25. |
What is the primary structural feature necessary for a molecule to make it useful in a condensation polymerisation reaction ? |
Answer» Solution :The primary feature necessary for MONOMER UNITS to undergo condenation polymerization is that each monomer unit must have two or THREE functional groups. For example , the MONOMERS of polyester are : terephthatic acid and ethylene glycol has two `-OH` groups.  In contrast , glypal polymer can be obtained by condensation of phthalic acid or its anhydride with either ethylene glycol which has two `-OH` groups or GLYCEROL which as three `-OH` groups. |
|
| 26. |
Which is used for electrical purposes: |
|
Answer» GERMAN silver |
|
| 27. |
What are inner-transition elements ? Decide which of the following atomic numbers are the numbers of the inner-transition elements : 29, 59, 74, 95, 102, 104. |
| Answer» Solution :The f-block elements, i.e., in which the last electron enters into f-subshell are CALLED inner-transistion elements. These INCLUDE lanthanoids (58 - 71) and actinoids (90 - 103). THUS, elements with ATOMIC numbers 59, 95 and 102 are inner - transition elements. | |
| 28. |
Whichof the following does not condensewithaldehydes or ketones. |
|
Answer» `CH_(3)- NO_(2)` |
|
| 29. |
Which one of the following compounds gives a positive iodoform test? |
|
Answer» Pentanal |
|
| 30. |
Which of the following shows greatest mert pair effect ? |
|
Answer» `F_2` |
|
| 31. |
Which of the following molecules is covalent and shows expanded octet in its formation ? |
|
Answer» HF |
|
| 32. |
Whichone of the followingstatementsrelated tolanthanons isincorrect ? |
|
Answer» Europiumshow + 2oxidation STATE. |
|
| 33. |
Which of the following compounds are gem-dihalides? |
|
Answer» Ethylidene chloride |
|
| 34. |
Which of the following does not exist in free form? |
|
Answer» `BF_(3)` |
|
| 35. |
Which of the following specie does not exist: |
|
Answer» `HEH^(+)` |
|
| 36. |
Whichprocess in utilizedin the synthesis of carbohydrates in green plants ? |
|
Answer» OXIDATION |
|
| 37. |
Which of the following is not characteristic of alcohols? |
|
Answer» Lower ALCOHOLS are stronger and have bitter taste |
|
| 38. |
Which of the following is correct about actinides ? |
|
Answer» They form stable oxidation state +2 |
|
| 39. |
Which instrument is used potential difference ? |
|
Answer» Ammeter |
|
| 40. |
Which of the following is not an amino acid?Glycine,Aniline,Histidine,Lysine. |
|
Answer» Glycine |
|
| 41. |
TiCl has structure similar to CsCl, the co- ordination number of Ti^(+)is- |
|
Answer» 4 |
|
| 42. |
When boron is fused with potassium hydroxide which pair of species are formed ? |
|
Answer» `K_2O+B_2O_3` |
|
| 43. |
Which out of the following is chiral in nature? |
|
Answer» `CH_(3)CH(OH)CH_(3)` |
|
| 44. |
Tritium undergoes radioactive decay giving |
|
Answer» `alpha-`particle |
|
| 45. |
Which of the following metal Fe, Zn, Pb, Ag and Pt do not give a metal nitrate on treatment with concentrated HNO_3? |
| Answer» Answer :D | |
| 46. |
Which of the following complex will have three isomeric forms ? |
|
Answer» CIS-`[Co(en)_(2)Cl_(2)]` 
|
|
| 47. |
The structure of butylated hydroxy toluene is_______ |
|
Answer»
|
|
| 48. |
Threonine is (2S,3R) -2-amino-3-hydroxybutanoic acid. Which of the following is threonine ? |
|
Answer»
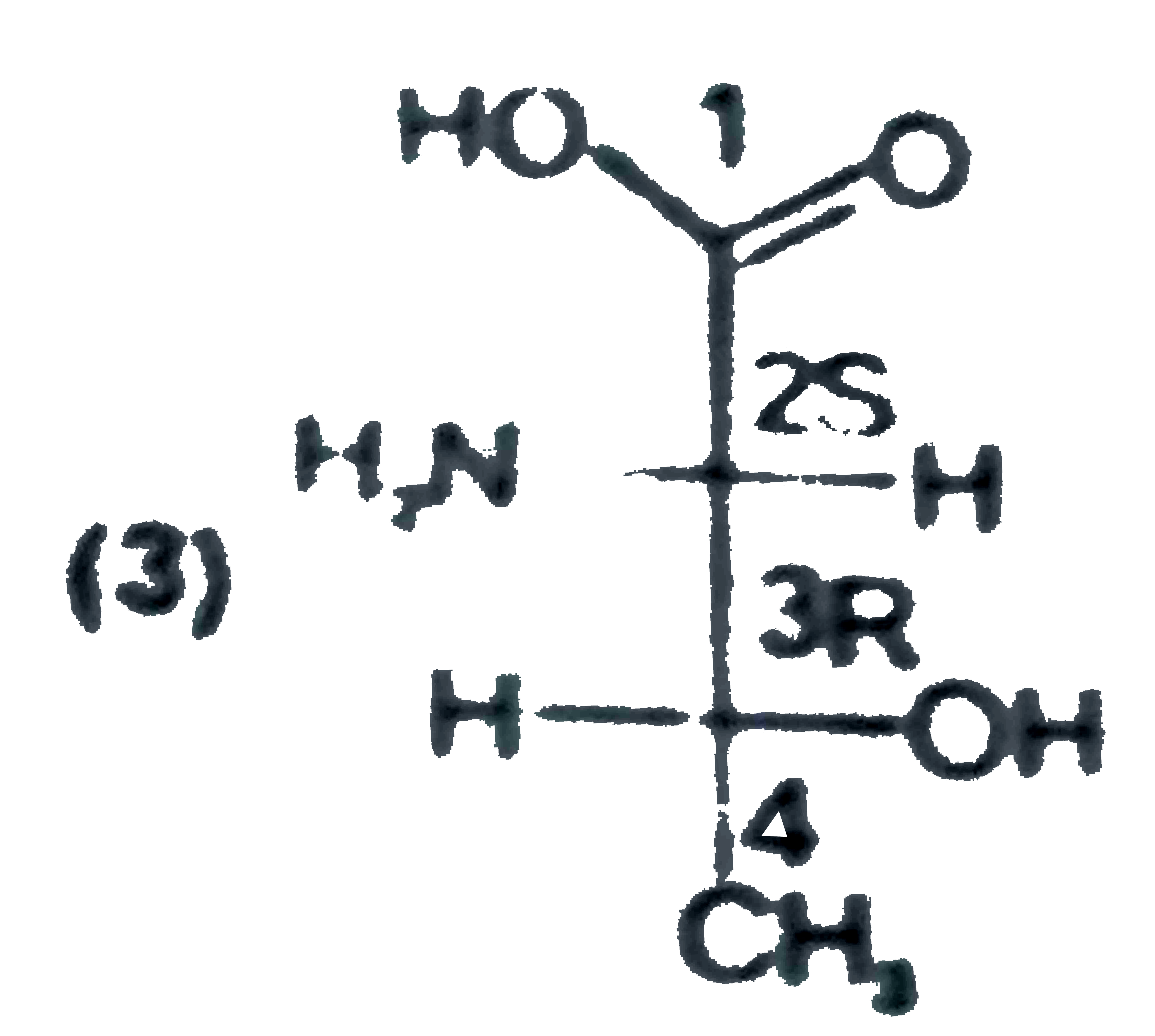
|
|
| 49. |
Which of the following is correct decreasing order of r.ms.s velocity at same temperature for H_2, N_2, CO_2 and O_2 |
|
Answer» `O_2 GT CO_2 gtN_2 gt H_2` |
|











