Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which compound does not decolourize bromine dissolved in carbon tetrachloride ? |
|
Answer» `C_2H_2` |
|
| 2. |
Which of the following is not a bio-polymer ? |
|
Answer» POLYSACCHARIDE. |
|
| 3. |
The unit of specific resistence is equal to ………………… . |
| Answer» Answer :A | |
| 4. |
Which of the following amines can be prepared by Gabriel synthesis. |
|
Answer» Isobutyl amine |
|
| 5. |
Write reaction between acetyl chloride and dimethyl cadmium. |
| Answer» SOLUTION :`2CH_(3)COCI+Cd[CH_3]_2overset("DRY")underset("ETHER")to2CH_(3)-underset(O)underset(||)C-CH_(3)+CdCI_(2)`. | |
| 6. |
The river 'Sone' receives wastes from |
|
Answer» Refinery |
|
| 7. |
What happens when Ethyne is hydrolysed in the presence of HgSO_4and H_2SO_4? |
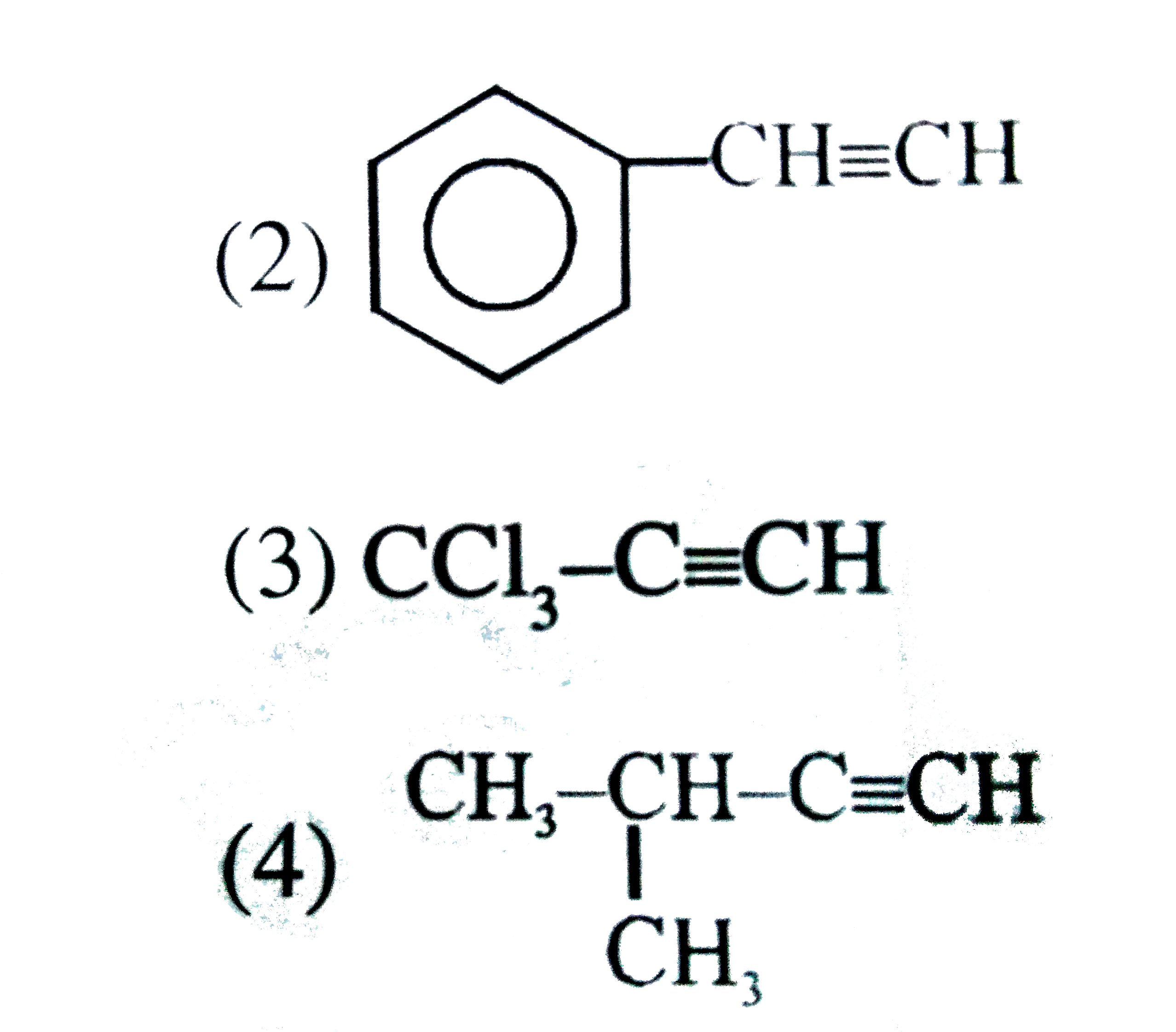
Answer» SOLUTION :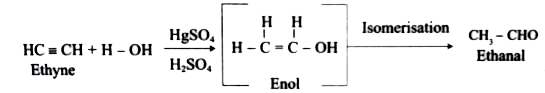
|
|
| 8. |
What is meant by 'Chelate effect' ? Give an example. |
|
Answer» Solution :When a BIDENTATE or a POLYDENTATE attaches itself to a metal ion, the complex formed is much more STABLE compared to when a monodentate attaches itself the metal ion.This is called chelate effect. Example :  Ethylenediamine which is bidentate LIGAND attaches to the metal ion from TWO points as shown and ring is formed which makes the complex stable. |
|
| 9. |
The two ions A^+ and B^- have radii 40 pm and 120 pm respectively. In the closed packed crystal of compound AB, the coordination number of A^+ would be |
|
Answer» 6 |
|
| 10. |
when 1.8 g of steam at the normal boiling point of water is converted inot water ,at the same temperature , enthalpy and entropy changes respectively will be[given , DeltaH_("vap")for water =40.8 KJ mol^(-1) ] |
|
Answer» `-8.12kJ,11 .89Jk^(-1)` ` = (-40.8)kJ XX (1.8/18) MOL =- 4.08 KJ` ` DeltaS= (DeltaH)/(T_(B))= (-4.08xx10^(3)J)/(373.15 K ) =- 10.93 JK^(-1)` |
|
| 11. |
Which of the following exhibits pink colour |
| Answer» Answer :A | |
| 12. |
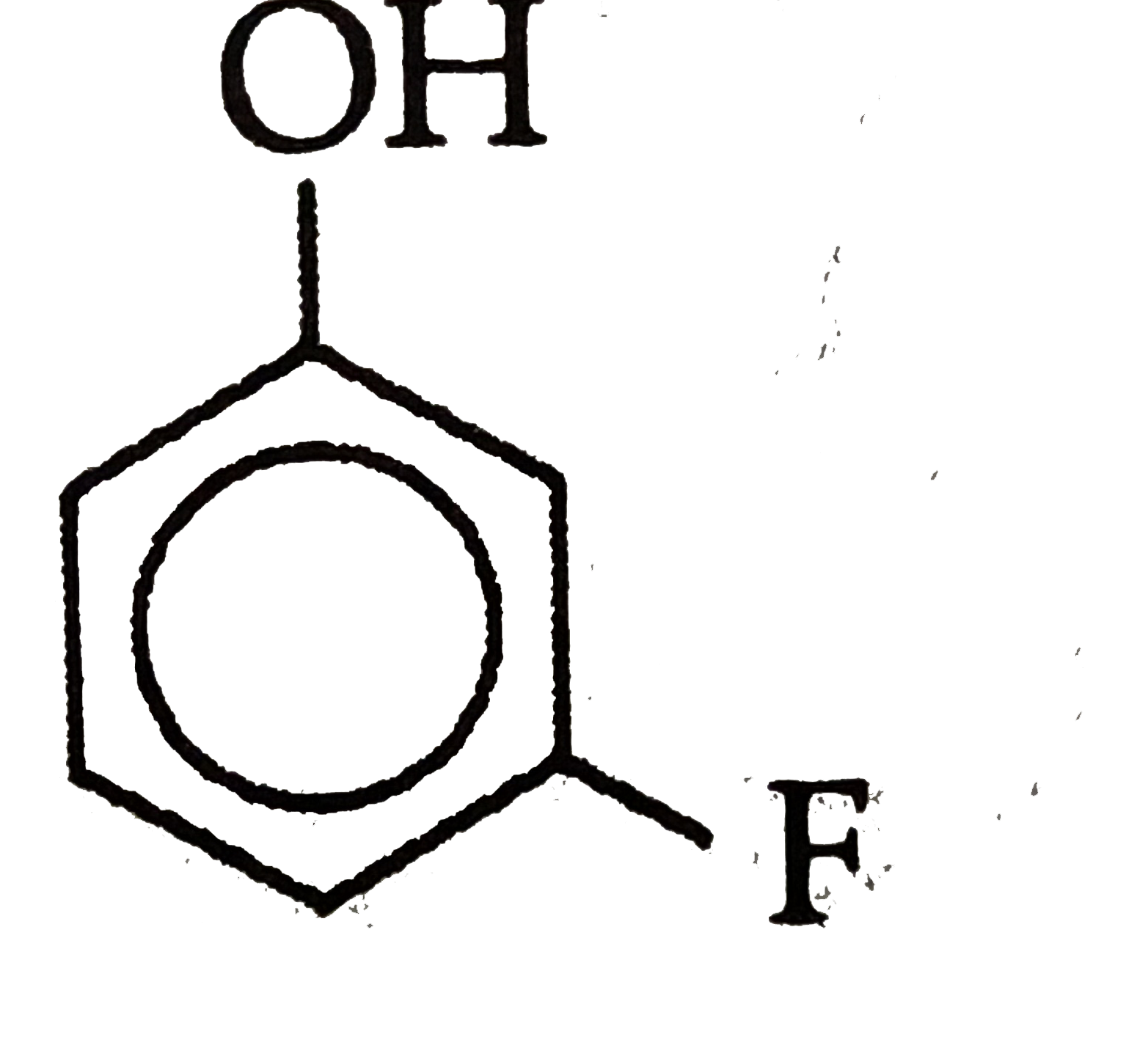
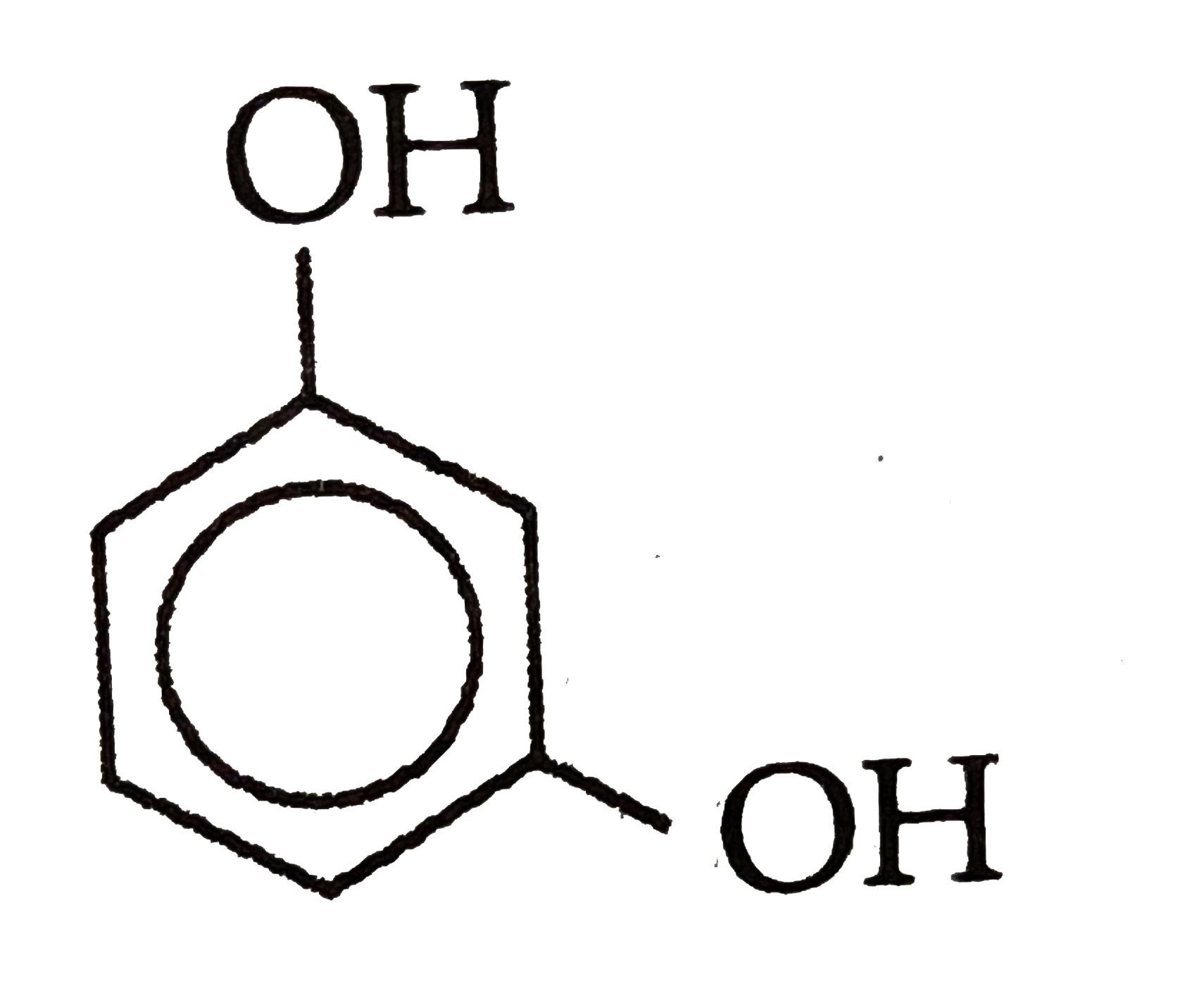
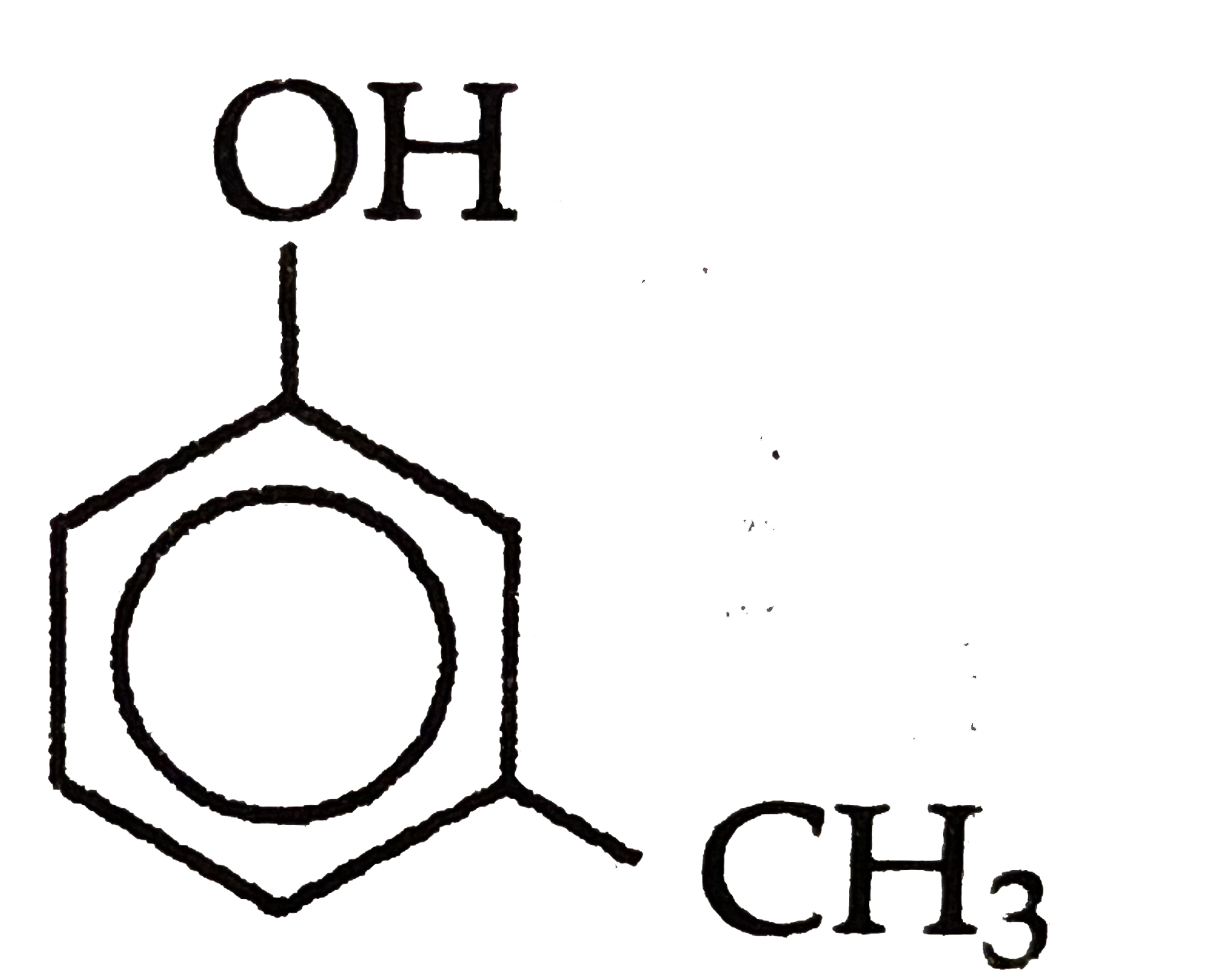
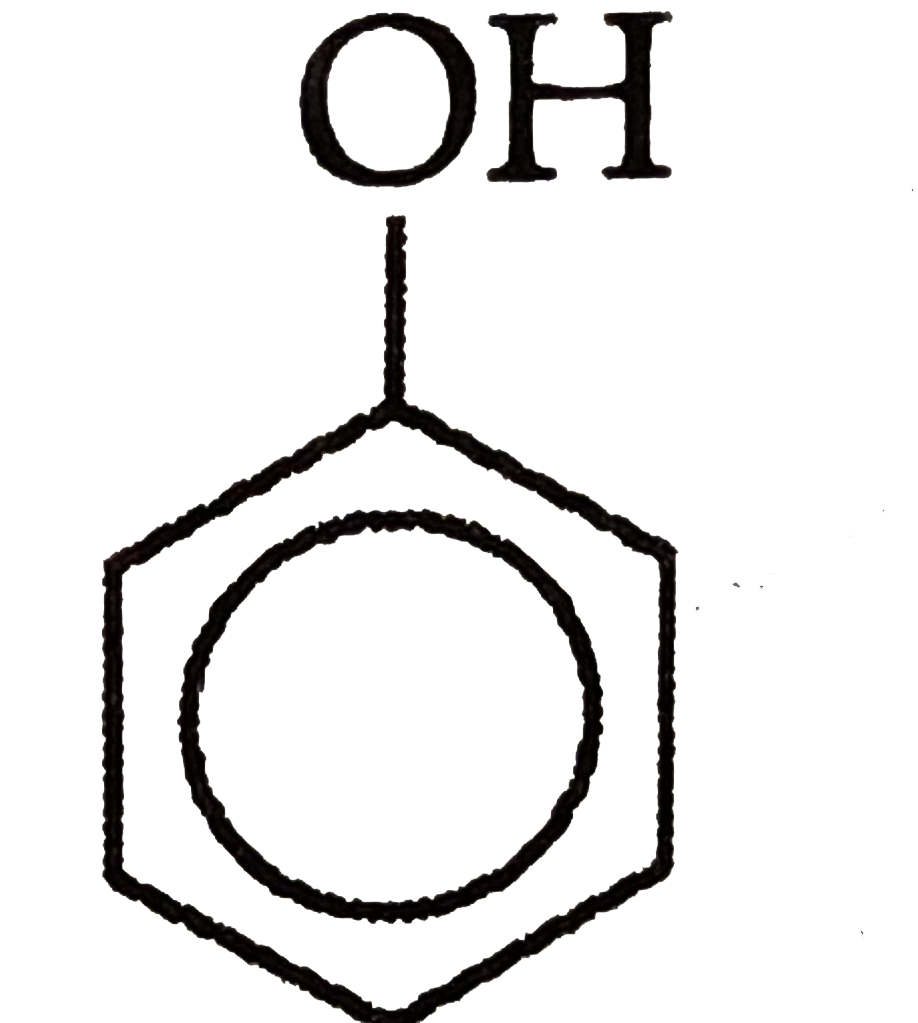
Which of the followingis most acidic ? |
|
Answer»
|
|
| 13. |
The secondary structure of a protein refers to : |
|
Answer» Hydrophobic interactions |
|
| 14. |
Comment on the nature of two S–O bonds formed in SO_(2) molecule. Are the two S–O bonds in this molecule equal ? |
| Answer» Solution :Both the S-O bonds in `SO_2` are covalent and have EQUAL strength due to RESONANCE. | |
| 15. |
Which of the following conversion can be brought about under Wolff-Kishner reduction ? |
|
Answer» BENZALDEHYDE to BENZYL alcohol |
|
| 16. |
Which one following formulae does not represent an organic compound |
|
Answer» `C_(4)H_(10)O_(4)` |
|
| 17. |
Which of the following compounds is optically acitve |
|
Answer» `(CH_(3))_(2)CHOH` |
|
| 18. |
Which of the following are examples of aldol condensation ? |
|
Answer» `2CH_(3)CHO overset("diL NAOH")to CH_(3)CHOHCH_(2)CHO` |
|
| 19. |
What happens when (i) PCl_(5) is heated ? (ii) H_(3)PO_(3) is heated ? Write the reactions involved . |
|
Answer» Solution :`(i) PCl_(5) to PCl_(3)+Cl_(2)` (ii) On HEATING, phosphorus acid disproportionates to give orthophosphoric acid and phosphire. `4H_(3)PO_(3)to3H_(3)PO_(4)+PH_(3)` |
|
| 20. |
Which of the following compounds will not liberate nitrogen gas on reaction with nitrous acid ? |
|
Answer» Diethylamine `(C_(2)H_(5))_(2)NH+HNO_(2) rarr (C_(2)H_(5))_(2)N NO`  
|
|
| 22. |
Which has largest C-O bond length in the given, complexes ? |
|
Answer» `NI(CO)_(4)` |
|
| 23. |
What are limited spectrum antibiotics? Give one example. |
| Answer» SOLUTION :Narrow spectrum antibiotics are EFFECTIVE against a single ORGANISM or disease and referred to as limited spectrum antibiotics, e.g., penicillin G. | |
| 24. |
Which of the following contain weaker Ppi-ppi back bonding? |
| Answer» Solution :`N//A` | |
| 25. |
The standar reduction potentials of Zn^(2+)|Zn,Cu^(2+)||Cu^(2+)|Cu| Zn |Zn^(2+)||Ag^(+)|AgIIICu|Cu^(2+)||Ag^(+)|Ag What is the correctorder of E_(cell)^(@)of thesecells ? |
|
Answer» `II gt IIII` `CU^(2+) rarr Cu` `Ag^(+) rarr Ag` Cell reaction of Iis `Zn + Cu^(2+) rarr Zn^(2+) +Cu` `E_(cell)^(@)=E_("oxidation" + E_("reduction")=+0.76 +0.34 =+1.10 V ` cellreactionof (III)is `Cu+Ag^(+) rarr Cu^(2+) +Ag` `E_(cell)^(@) =- 0.34 +0.8 =+0.46 V` So the correct order of `E_(Cell)^(@)` of these CELLIS `IIGT IgtIII` |
|
| 26. |
Which of the following statements is/are incorrect? |
|
Answer» The entropy of the UNIVERSE decreases and increases at a specificf rate |
|
| 27. |
Which pair of the following substances is said to be isomorphous? |
|
Answer» White VITRIOL and BLUE vitriol |
|
| 28. |
When you heat chloroform with silver powder ……… is formed. |
| Answer» Solution :`underset("Chloroform")(CHCl_(3))+6Ag+ Cl_(3)CH rarr underset("Ethyne")(CH-=CH)+6AgCl` | |
| 29. |
Vitamin B_(1) is …………………. |
|
Answer» THYMINE |
|
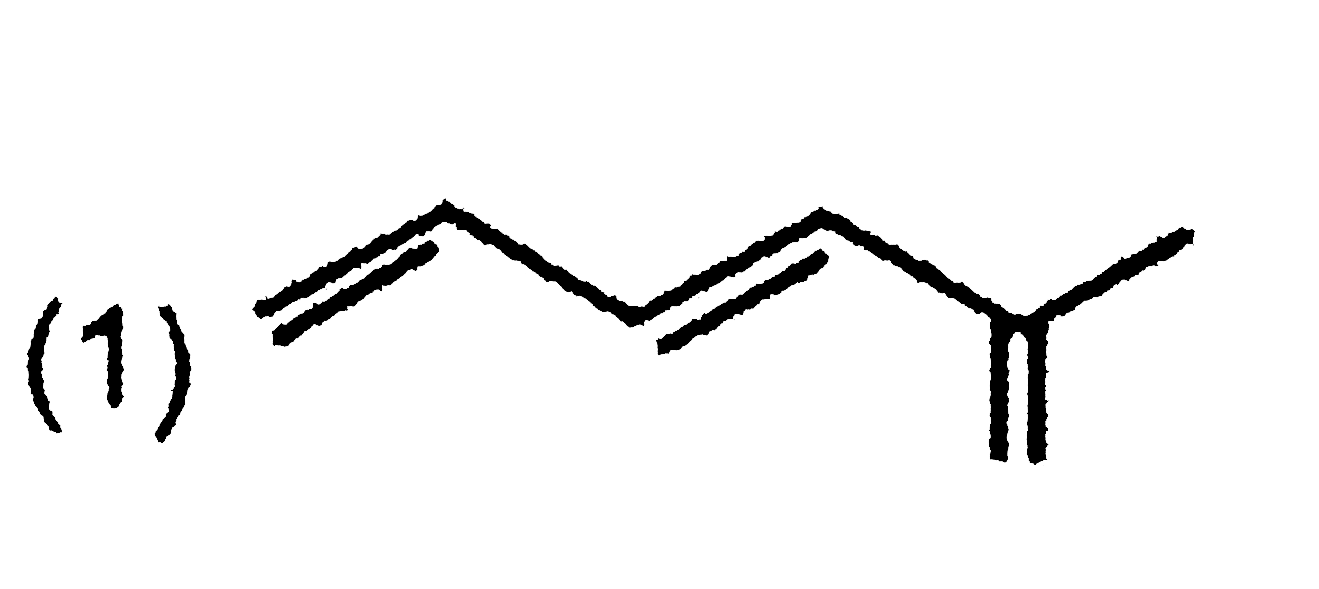
| 30. |
Which of the followingunsaturated hydrocarbons gives chloropene ( a monomer for neoprene) when treated with HCl in the presence of CuCl |
|
Answer» `CH-=C-C-=CH` |
|
| 31. |
Which isomer of cyclohexane hexachloride is a very strong insecticide |
|
Answer» `ALPHA` |
|
| 32. |
What prompted Bartlett to the discovery of noble gas compounds. |
| Answer» Solution :Since `PtF_(6)` oxidises `O_(2)` to `O_(2)^(+),` Bartlett THOUGH that `PtF_(6)` should also oxidise `XE` to `Xe^(+)` because the ionization energies of `O_(2)("1175 kJ mol"^(-1))` and `Xe ("1170 kJ mol"^(-1))` are QUITE close. | |
| 33. |
Which gives ketonic group after hydroboration oxidation : |
|
Answer» `CH_(3)-CH_(2)-C-=CH` |
|
| 34. |
Which of ther following are biopolymers ? |
|
Answer» Nucletic acids |
|
| 35. |
What are fuel cells? Discuss the working of fuel cell. |
|
Answer» |
|
| 36. |
When 1 mol of potassium succinate is electrolysed between Pt electrodes, number of moles of gaseous product obtained at cathode and anode are respectively. |
|
Answer» 1, 2 |
|
| 37. |
Which of the following are not used as food preservatives? |
|
Answer» Table salt |
|
| 38. |
Which of the following is the correct sequence of decreasing reactivity towards hydroxide ion promoted hydrolysis among the following compounds? |
|
Answer» `IgtIIgtIIIgtIV` |
|
| 39. |
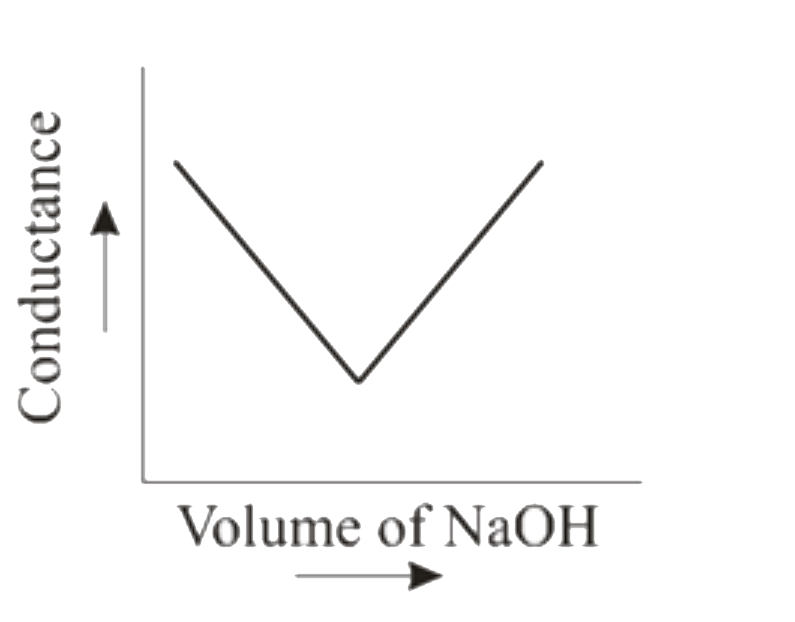
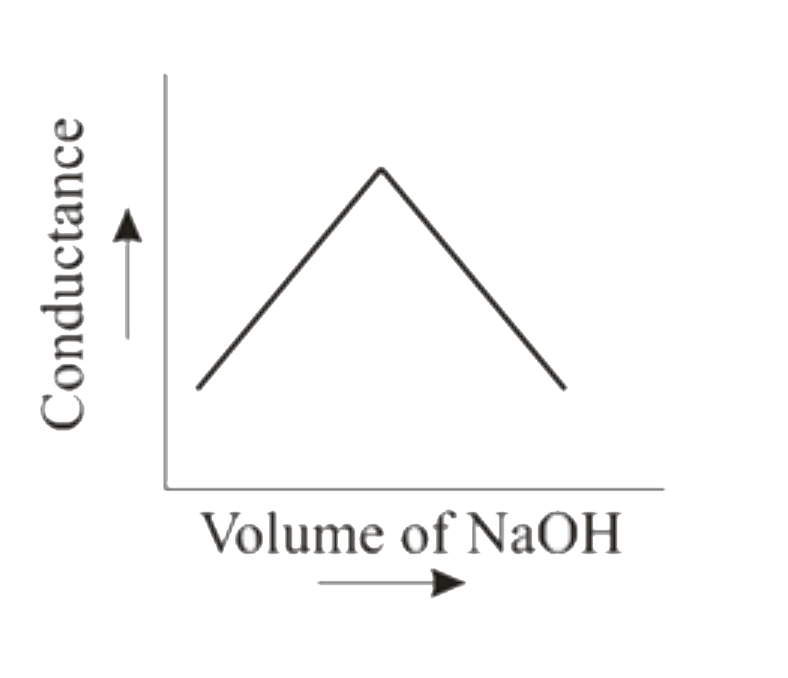
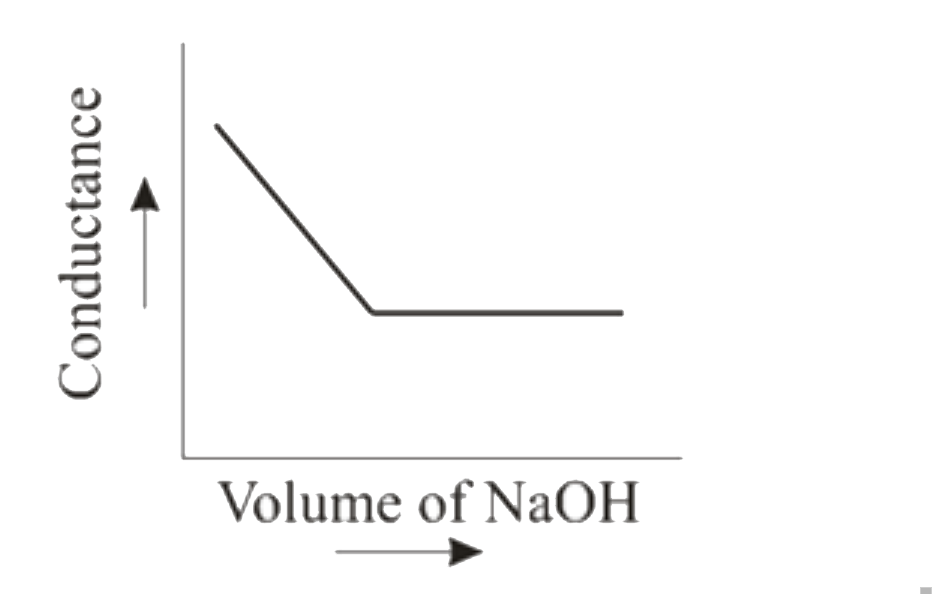
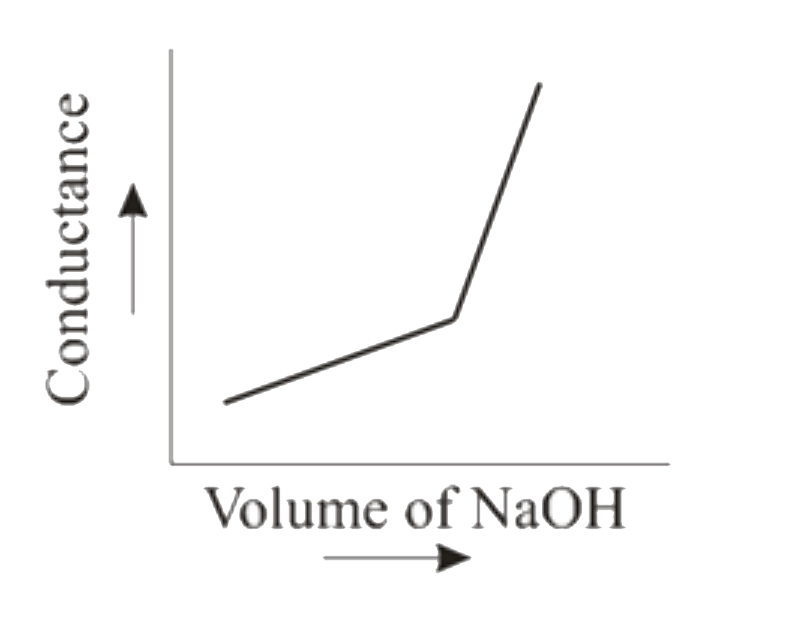
When HCl (aq) is titrated with NaOH(aq) conductometrically then the graphical representation of the titration will be |
|
Answer»
|
|
| 40. |
Which of the following ion will not form coloured aqueous solution? |
|
Answer» `CU^(2+)` |
|
| 41. |
Theunknowcompoundis takenin vessesoluble In diluteHCland small amountof NaNO_(2)is added , nitrogengasis evolvednitrogen gasin evolved . Theunknowcompoundis |
|
Answer» `1^(@)` amine |
|
| 42. |
When excess of SnCl_(2) is added to HgCl_(2), the substance formed is |
|
Answer» `HgCl_(2)` |
|
| 43. |
Which of the following complexes or molecules has/ have N-N bond length greater than that in free N_(2) molecule? |
|
Answer» `[Ru(NH_(3)_(5)(N_(2))]^(+)` |
|
| 44. |
Which gives positive haloform test and positive Fehling's solution : |
|
Answer» ACETONE |
|
| 45. |
Which one of the following can act as an insulator? |
| Answer» Answer :B | |
| 46. |
Which of the following has highest number of geometrical isomers. |
|
Answer»
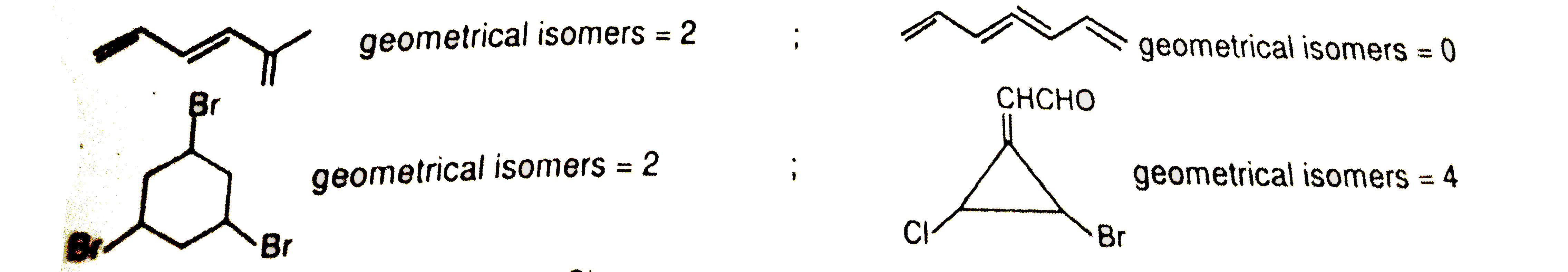
|
|
| 47. |
The sp^(3)d^(2) hybridization of central atom of molecule would lead to |
|
Answer» square PLANAR geometry 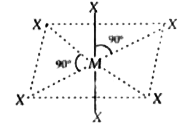
|
|
| 48. |
The total vapour pressure of a mixture of 1 mole of A (P_A = 200 mm Hg) and 3 mole of B (P_B= 360 mm Hg) is 350 mm Hg. Then |
|
Answer» There is no DEVIATION from Raoult's law |
|
| 49. |
What is coupling reaction? |
| Answer» Solution :The reaction of diazonium salts with PHENOLS in basic medium (pH9-10) or with `1^(@)` amines in acidic medium (pH 4-5) to give the corresponding AZO DYES is called to COUPLING reaction. | |