Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which is not a nucleophilic addition reaction : |
|
Answer» `RCHO + NaHSO_3 rarr RCH(OH)SO_3Na` |
|
| 2. |
Which aldehydes are formed by the reductive ozonolysis of penta, -1, -3, - diene ? |
|
Answer» SOLUTION :Formaldehyde, ACETALDEHYDE and GLYOXAL are formed by the OZONOLYSIS of penta-1,3-diene `CH_(2) = CH- CH= CH- CH_(3) + O_(3) underset(Zn)overset(H_(2)O)rarr CH_(2)O + OHC CHO + CH_(3)CHO` |
|
| 3. |
which one is soluble in NaOH :- |
|
Answer» N-ETHYL benzensulphonamide |
|
| 4. |
Which one of the following statements is incorrect in the case of heterogeneous catalyst ? |
|
Answer» The catalyst lowers the energy of activation |
|
| 5. |
When H_2S is passed through an ammoniacal salt solution X, a dirty white precipitate is obtained. The X can be a |
|
Answer» cobalt salt |
|
| 6. |
Write the name of coordination compound [Co(NH_3)_5Cl] Cl_2. |
| Answer» SOLUTION :Chloropentamine COBALT (III) CHLORIDE. | |
| 8. |
According to kinetic theory of gases, for a datomic molecule. |
|
Answer» The root mean square VELOCITY is inversely proportional to the temperature |
|
| 9. |
Write equation for the synthesisof Buna - S . |
|
Answer» Solution :Buna - S is prepared by additionpolymerisationof buta - 1, 3- diene and STYRENE. `UNDERSET("Buta-1,3-diene")(nCH_(2))=CH- CH_(2) +underset("Styrene")underset(C_(6)H_(5))underset(|)(nCH) = CH_(2) OVERSET(Na) to underset("Buna-S")((-CH_(2) - CH =CH-CH_(2) - underset(C_(6)H_(5))underset(|)(CH) - CH_(2)-)_(n)` |
|
| 10. |
Which of the following reactions with yield 2,2-dibromopropane? |
|
Answer» `CH_(3)-C-=CH+2HBr to` 
|
|
| 11. |
Which of the following bicarbonate can not exist in solid state :- |
|
Answer» `LiHCO_(3)` |
|
| 12. |
Which of the following is an example of octahedal complex? |
|
Answer» `[FeF_(6)^(-)]^(3-)` |
|
| 13. |
Write the structures of A,B and C in the following: CH_(3)-Cl overset(KCN)to A overset(LiAlH_(4))to B overset(CHCl_(3)+alc." KOH")toC |
| Answer» Solution :`underset("Chloromethane")(CH_(3)Cl) underset("Nacleophilic substitution (-KCl)")overset(KCN)to underset("Ethanetrile (A)")(CH_(3)-CN) underset(("Reducaton"))overset(LiAlH_(4))to underset("ETHANAMINE (B)")(CH_(3)-CH_(2)NH_(2)) underset(("Carbylamine reaction"))overset(CHCl_(3)+acl." "KOH,DELTA)to underset("Ethyl carbylamine (C)")(CH_(3)CH_(2)-N overset(to)(=)C)` | |
| 14. |
Which of the following are square planar complexes ? (i) [AuCl_(4)]^(-) (ii)[PtCl_(4)]^(2-) (iii)[Mn(Br)_(4)]^(2-) (iv) [Cu (NH_(3) )_(4) ]^(2+) Select the correct answer using the codes given below: - |
|
Answer» i and II only |
|
| 15. |
Toluene di-isocyanate is used to prepare: |
|
Answer» Ployesters |
|
| 16. |
Which of the following crystal has unit cell such that aa neb ne c and alphane beta ne gamma ne 90^(@) |
| Answer» Answer :D | |
| 17. |
Which one of the following is not formed when a mixture of methyl bromide and bromobenzene is heated with sodium metal in the presence of dry ether ? |
|
Answer» ETHANE 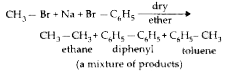
|
|
| 18. |
What happens when benzene diazonium chloride is heated with water ? |
Answer» Solution :`to` When phenol is reacted with benzene DIAZONIUM CHLORIDE, phenol is OBTAINED. 
|
|
| 19. |
Which one is a monomer of natural rubber ? |
|
Answer» `(CH_(3))_(2)-C=CH-CH_(3)` |
|
| 20. |
What type substances would make better permanent magnets, ferromagnetic or ferrimagnetic ? |
| Answer» SOLUTION :FERROMAGNETIC SUBSTANCES. | |
| 21. |
What type of substance is phenylalanine hydroxylate ? What is its importance for us? |
| Answer» SOLUTION : It is an ENZYME whose DEFICIENCY CAUSES MENTAL retardation. | |
| 22. |
The vapour pressure of pure liquid A at 300K is 577 Torr and that of pure liquid B is 390 Torr. These two compounds form ideal liquid and gaseous mixtures. Consider the equilibrium composition of a mixture in which the mole fraction of A in the vapour is 0.35. Calculate the total pressure of the vapour and the composition of the liquid mixture. |
|
Answer» <P> let mole fraction of `A` in solution `=X_(A)` HENCE, `P_("total")=P_(A^(0)X_(A)+P_(B)^(0)(1-X_(A))` also `X_(A)=` ,p,e fraction of A in the vapour `=0.35` `X_(A)=(P_(A)^(@)X_(A))/(P_(A)^(@)X_(A)+P_(B)^(@)(1-X_(A))=0.35` `=(575X_(A))/(575X_(A)+390(1-X_(A))` this GIVES `X_(A)=0.27` hence, total pressure `P_("total")=575xx0.27+390xx0.73` `=440` Torr Composition of liquid MIXTURE, `A=27` mol `%,B=73` mol `%` total pressure `=440` Torr |
|
| 23. |
The time required for 10% completion of a first order reaction at 298 K is equal to that required for its 25% completion at 308 K.If the value ofA is 4xx10^(10)S^(-1).Calculate k at 318 K and E_(a) |
|
Answer» Solution :`T_(1)`=298,time =`t_(1)` If initial concentration =`[R]_(0) mol L^(-1)` The reaction is COMPLETE 10% ,so 90% reactant will be reamain. `therefore[R]_(t)=0.9[R]_(0) mol L^(-1)` `therefore` log `k_(298)=(2.303)/(t_(1)) log [R_(0)]/(0.9 [R]_(0))=(2.303)/(t_(1))` log `(1)/(0.9)` `therefore t_(1)=(2.303)/(log k_(298))xx0.04575` `therefore t_(1)=(0.1054)/(log k_(298))` Inintial concentration =`[R]_(0) mol L^(-1),T_(2)`=308 K . The 25% reaction is complete in t time. `therefore` The concentration after completion of 75% reaction=75% of `[R]_(0)` `therefore [R]_(t)=0.75[R]_(0) mol L^(-1)` `k_(308)=(2.303)/(t_(2))xxlog ([R]_(0))/(0.75 [R]_(0))` `(2.303)/(t_(2))xxlog 1.3333=(2.303xx0.1249)/(t_(2))` `therefore t_(2)=(0.2878)/(log k_(308))`........(2) But `t_(1)=t_(2)` `therefore (0.1059)/(k_((298)))=(0.2878)/(k_((308)))` `therefore (K_((308)))/(k_((298)))=(0.2878)/(0.1059)=2.717` ..........(3) Calculation of `E_(a)` :`=4XX10^(10)s^(-1)` so, at 318 K temperature k=(?)`E_(a)` (?) `therefore (log K_(2))/(log k_(1))=(E_(a))/(2.303xx8.314)((308-298)/(308xx298))` `therefore` log 2.717=`(E_(a))/(2.303xx8.314)xx(10)/(308xx298)` `therefore E_(a)=75990.0 J mol^(-1)` logk=log A-`(E_(a))/(2.303 RT)` `=log 4xx10^(10)-(75990 mol^(-1))/(2.303xx8.314 JK^(-1) mol^(-1)xx318 K)` `therefore` k=0.013267 `s^(-1)~~0.013 s^(-1)` |
|
| 24. |
What happens to freezing point of benzene when nephthalens is added ? |
|
Answer» increases with increases in temperature. |
|
| 25. |
Which are correct statements |
|
Answer» REVERSE osmosis is used desalination of sea water |
|
| 26. |
Two electrolytic cell are attached in series and current is passing through them. First electrolytic cell X(NO_(3))_(3(aq)) and second electrolytic cell posses Y(NO_(3))_(2) and molecular mass of X and Y are 1:2 respectively. Hence what is the ratio of lierated mass ? |
|
Answer» `3:2` Molecular MASS ratio=1:2 and so EQUIVALENT mass`=(1)/(3):(2)/(2) or 1:3`. |
|
| 27. |
What type of bonding helps in stabilising the alpha-helix structure of proteins ? |
| Answer» SOLUTION :The `alpha`-helix STRUCTURE of PROTEINS is STABILIZED by intramolecular H-bonding between `C=O` of one amino acid RESIDUE and the `N-H` of the fourth amino acid residue in the chain. | |
| 28. |
Which of the followingd blockelementhas halffilledpenultimate d subshellas well as halffilledvalencesubshell ? |
| Answer» ANSWER :A | |
| 29. |
Which of the following will reduce alcohol ? |
| Answer» Solution :Red P | |
| 31. |
Which of the following will have the highest boiling point at 1 atm pressure: |
|
Answer» 0.1 M NaCl |
|
| 32. |
Which of the following statements are correct : (A) The coordination number of each type of ion in CsCl is 8. (B) A metal that crystallises in BCC structure has a coordination number 12. (C) A unit cell of an ionic crystal shares some of its ions with other unit cells (D) The length of the unit cell in NaCl is 552 pm.[r_(Na^(+))=95pm,r_(cl^(-))=181pm] |
|
Answer» SOLUTION :`r_(+) + r_(–) = a//2` (95 + 181)=` a//2` `implies a = 276 x× 2` a = 552 pm. |
|
| 33. |
When SO_2 gas is passed through acidified K_2Cr_2O_7 solution, the colour of the solution changes to |
| Answer» SOLUTION :Green | |
| 34. |
Write the role of the following : (i) NaAlF_(4) in the extraction of Aluminium (ii) CO in the refining of Ni. |
|
Answer» Solution :(i) `NaAIF_(4)` in the extraction of Aluminium `NaAlF_(4)` lowers the meelting point of `Al_(2)O_(3)` and increases CONDUCTIVITY to speed up the deposition of aluminium. (ii) CO in the refining of Ni CO forms a VOLATILE complex named NICKEL tetracarbonyl. This complex is decomposed at higher temperature to obtain pure nickel. |
|
| 35. |
Which of the following is the size of the colloidal particle? |
|
Answer» `100 MU m` DIAMETER - `1000mu m` diameter |
|
| 36. |
Which statement is true about the most stable Lewis structure for CS_(2) ? |
|
Answer» There are no LONE pairs in molecule 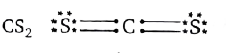 Total lone pairs =4 `"" ` Total double bond=2 All the atoms have COMPLETE octet SULPHUR is the central atom. |
|
| 37. |
Which pairs contains both ambidentate and flexidentate? |
|
Answer» `CO_(3)^(2-),CN^(-)` |
|
| 38. |
What is D in the following sequence of reactions ? |
|
Answer»

|
|
| 39. |
The substance which possesses zero resistance as 0 K: |
|
Answer» Conductor |
|
| 40. |
Which one of the following is called as Zeise's salt ? |
|
Answer» `[Pt(NH_(3))_(4)][PtCl_(4)]` |
|
| 41. |
Which of following is correct : |
|
Answer» All ALDEHYDES undergo Cannizzaro's reaction |
|
| 42. |
Zinc carbonate is precipitated from zinc sulphate solution by the addition of: |
|
Answer» `Na_(2)CO_(3)` |
|
| 43. |
Which of the following crystal systems does not have body-centred lattice- |
|
Answer» orthorhombic |
|
| 44. |
Which of the following is a semi synthetic polymer ? |
|
Answer» CELLULOSE NITRATE |
|
| 45. |
What type of detergents are used for dish washing ? |
| Answer» SOLUTION :NON ionic,i.e., POLYETHYLENE GLYCOL STEARATE or lauryl alcohol ethoxylate. | |
| 46. |
Which among the following statements is INCORRECT ? |
|
Answer» The atomic radius of a halogen atom is the smallest in its period. |
|
| 47. |
When HCOONa is heated to 390^@ C, it gives : |
|
Answer» HCHO |
|
| 49. |
Which is the correct IUPAC name for CH_(3)-underset(C_(2)H_(5))underset(|)"CH"-CH_(2)-Br? |
|
Answer» 1-Bromo-2-ethylpropane |
|
| 50. |
Which among the following equations represents the reduction reaction reaction taking place in lead accumulator at positive electrode, while it is being used as a source of electrical energy ? |
|
Answer» `PB to Pb^(2+)` `PbO_(2(s))+4H_((AQ))^(+)+SO_(4(aq))^(2-)+2e^(-) to PbSO_(4(s))+2H_(2)O_((l))` `therefore Pb^(4+)` is converted into `Pb^(2+)` |
|



