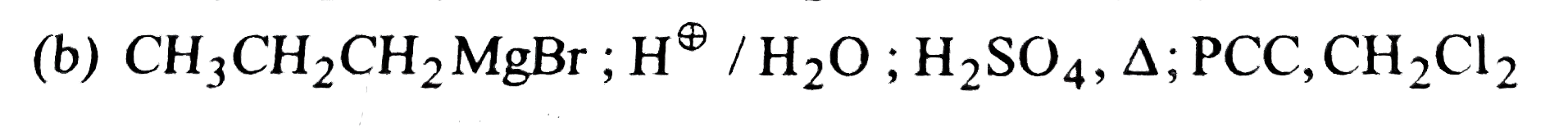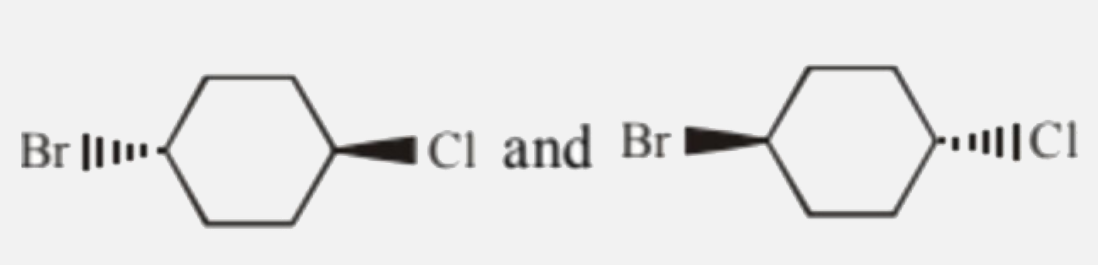Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
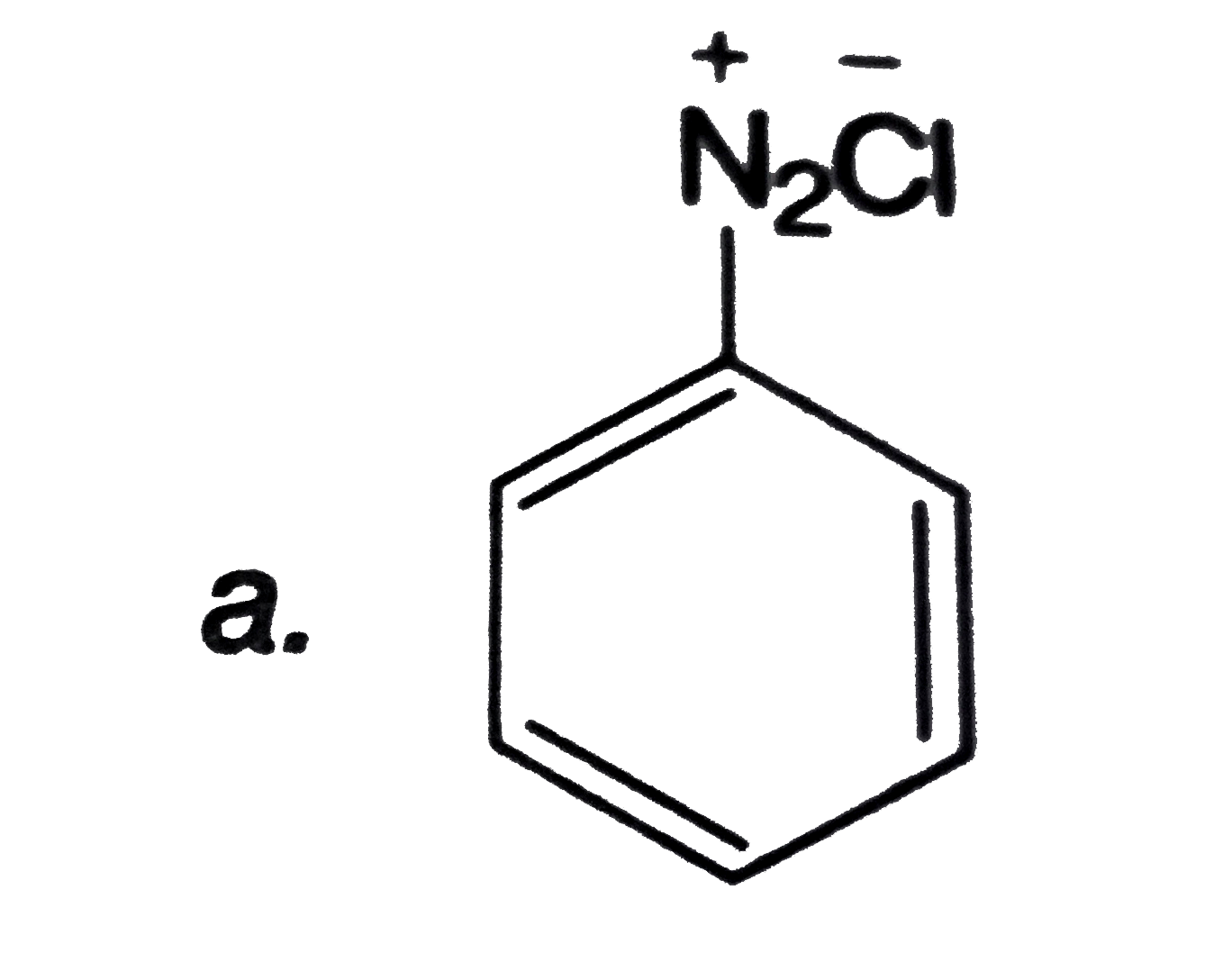
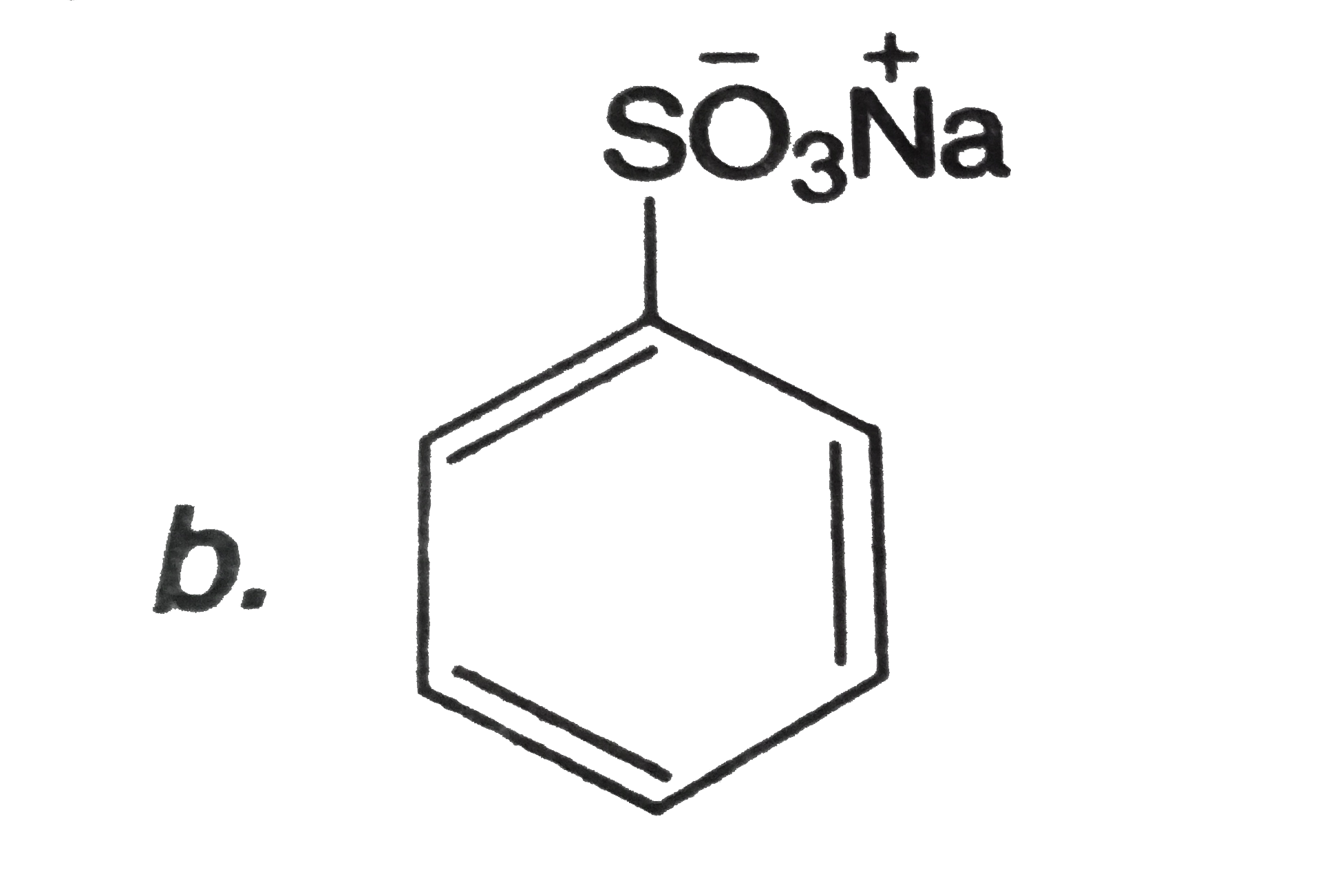
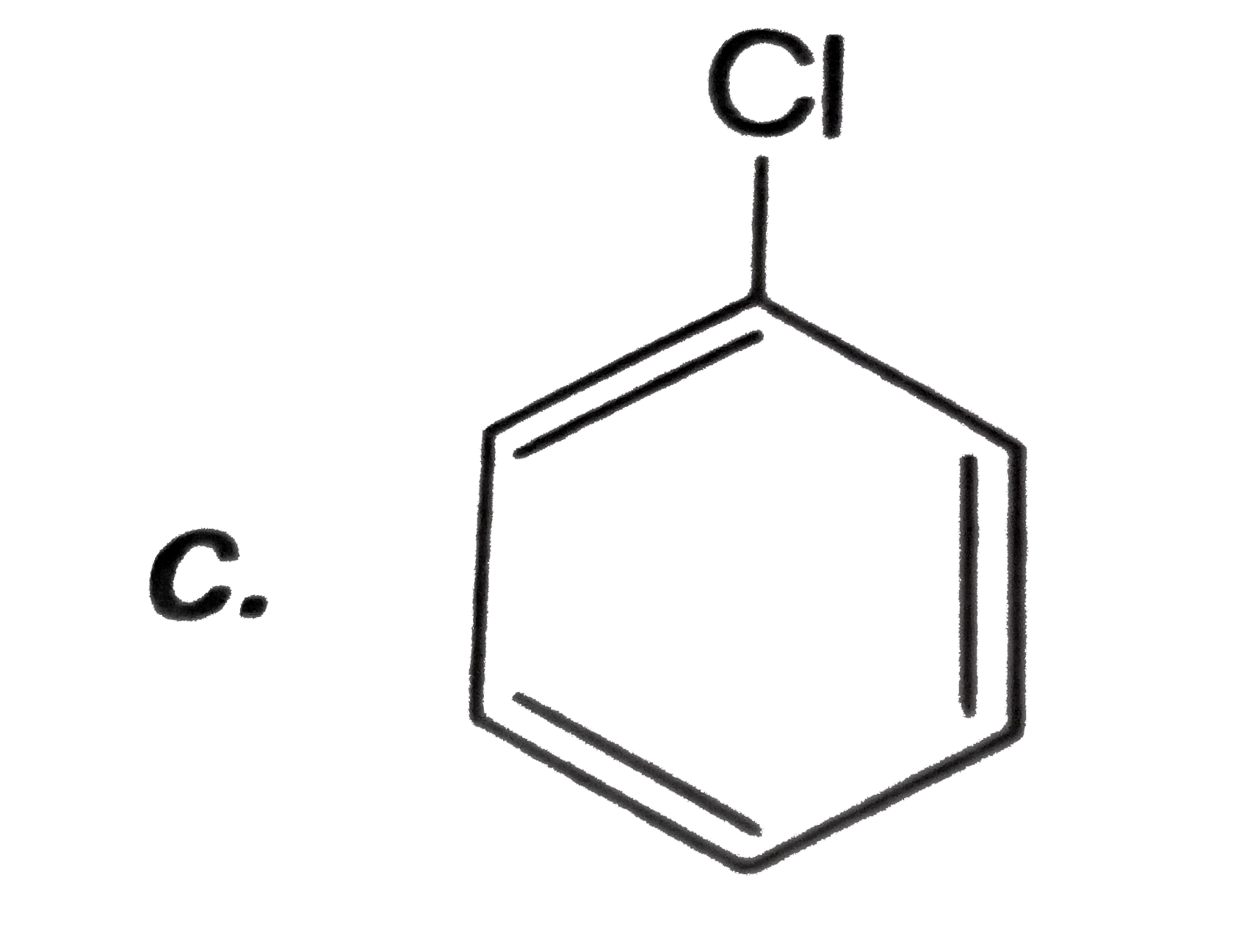
Which of the following can be used to convert into phenol in one or more steps? |
|
Answer»

|
|
| 2. |
Which lipids are present in essential oils? |
| Answer» Solution :SIMPLE LIPIDS are present in essential oils such as menthol, CAMPHOR, LIMONENE, `beta`- pinene, geraniol, etc. | |
| 3. |
How will you convert (i) Ethanal to 2-hydroxypropanoic acid. (ii) Toluene to benzoic acid. |
Answer» SOLUTION :TOLUENE to BENZOIC ACID : 
|
|
| 4. |
Which one of the following is not used for disinfection of drinking water |
|
Answer» Chlorine |
|
| 5. |
Which of the following involves ppi-ppi back bonding ? |
|
Answer» `:C Cl_2` |
|
| 6. |
Which of the following compounds will give a yellow precipitate with iodine and alkali ? |
|
Answer» 2-Hydroxy propanal |
|
| 7. |
Z_(2)undergo disproportion in an alkaline medium into a mixture of Z^(-) and ZO_(3)^(-). Equivalent mass of Z_(2)in the reaction is: [given that atomic mass of Z is 80 gm/mol] |
|
Answer» 96 `3Z_(2) rarr 5Z^(-) + ZO_(3)^(-)` ……..N FACTOR of `Z_(2)= 5//3` `therefore` Equivalent mass of `Z_(2)= (M^(@))/(5//3) = (3)/(5) [160] = 96` |
|
| 8. |
What happens when ethyl amine reacts with methyl iodide ? |
| Answer» SOLUTION :`C_2H_5NH_2 + 3CH_3I RARR C_2H_5(CH_3)_3I + 2HI` | |
| 9. |
Which of the following is the correct order of relative strength of acids ? |
|
Answer» `ClCH_(2)COOH gt BrCH_(2)COOH gt FCH_(2)COOH` |
|
| 11. |
The separation of colloidal particles from the particles having molecular dimensions by electricity is known as |
|
Answer» electrolysis |
|
| 12. |
Which of the following is coloured |
|
Answer» `ScCl_(3)`  Due to presence of unpaired electrons it will form COLOURED SALT. |
|
| 13. |
When the azimuthalquantum number has has the value of 2 , the number of orbitals possible are |
|
Answer» 3 m= MAGNETIC quantum no. l = Azimuthal quantum no . Where l=2 REPRESENTS 'd' subshell aand d subshell has five orbitals. `(d_(xy),d_(yz),d_(ZX),d_(X^(2)-y^(2)),d_(z^(2)))`. |
|
| 14. |
When a mixture, containing PCl_3 and PCl_5 is heated with ethyl alcohol , a total of 4 moles of ethyl chloride is formed . Mole ratio of PCl_3 and PCl_5 in the mixture is |
|
Answer» `3:1` |
|
| 15. |
Which of the following cartion (s) will turnblue in oxidising flame ? |
|
Answer» `CO^(2+)` |
|
| 16. |
Which type of ligands form chelates ? |
| Answer» SOLUTION :POLYDENTATE LIGANDS. | |
| 17. |
Which of the folliwng is the order of the rate of reaction of C_(6)H_(6),C_(6)D_(6), and C_(6)T_(6) towards sulphonation? |
|
Answer» Same rates of reactions of `C_(6)D_(6), C_(6)D_(6)` and `C_(6)T_(6)`. |
|
| 18. |
Which substance is not an important antitoxins ? |
|
Answer» CITRIC acid |
|
| 19. |
Which of the following species is not aromatic - |
|
Answer» BENZENE  
|
|
| 20. |
When calomel is treated with ammonium hydroxide, a black substance is formed. The black substance is….. |
|
Answer» `Hg(NH_(2))_(2) + HGO` 
|
|
| 21. |
Which of the following sets of reagents would accomplish the following conversation |
|
Answer»
|
|
| 22. |
What type of drug is chloramphenicol ? |
| Answer» SOLUTION :CHLORAMPHENICOL is a wide SPECTRUM antibiotic and is effective against a NUMBER of microorganisms. | |
| 23. |
Which of the following compound will react faster I the S_(N^(1)) reaction and why? |
| Answer» Solution :The COMPOUND (I) being a secondary alkyl halide will react faster than the compound (II) which is a primary alkyl halide in nature since the secondary carbocation formed in the SLOW step is more STABLE than the primary carbocation. | |
| 24. |
The relation between triangleG, triangleH" and "triangleS is given by |
|
Answer» `TRIANGLEG= TRIANGLEH+T TRIANGLES` |
|
| 25. |
Which of the following is used as a scavenger in metallurgy? |
|
Answer» Na |
|
| 26. |
The total internal energy change for a reversible isothermal cycles is |
|
Answer» Always 100 calories PER degree |
|
| 27. |
Which of the following is an example of a heterogeneous catalytic reaction |
|
Answer» `2SO_(2(G)) + O_(2(g)) overset(NO(g))(rarr)2SO_(3(g))` |
|
| 28. |
The specific heat of a metal is 0.16. Its approximate atomic weight would be |
|
Answer» 32 |
|
| 29. |
When the periodicity of lattice points get disturb in a certain direction it is called ......... |
| Answer» Answer :B | |
| 30. |
Which is used for ending charge on colloidal solution |
|
Answer» Electrons |
|
| 32. |
Two metals M_(1) " and " M_(2) have reduction potential values of -xV and +yV respectively. Which will liberate H_(2) " and " H_(2)SO_(4)? |
| Answer» Solution :Metals having higher oxidation potential will liberate `H_(2) " from " H_(2)SO_(4)`. Hence, the metal `M_(1)` having +XV, oxidation potential will liberate `H_(2)` from `H_(2)SO_(4)`. | |
| 33. |
Which sugar is called invert sugar ? Why it is called so ? |
| Answer» Solution :Sucrose is called invert sugar. Sugar is obtained as DEXTROROTATORY NATURALLY. When HYDROLYSED sucrose give D-GLUCOSE and D-fructose in equimolar MIXTURE. Glucose is dextrorotatory `(+52.5^(@))` and fructose is laevorotatory `(-92.4^(@))` and as a result, the net resultant mixture is laevorotatory. This product changes from dextro (+) to laevo (-) after hydrolysis is called invert sugar. | |
| 34. |
Which one of the following will have largest number of atoms ? (i) 1 g Au(s) (ii) 1 g Na (s) (iii) 1 g Li (s) (iv) 1 g of Cl_(2)(g) (Atomic masses : Au = 197, Na = 23, Li = 7, Cl = 35.5 amu) |
|
Answer» Solution :(i) `"1 G Au"=(1)/(197)" MOL "=(1)/(197)xx6.02xx10^(23)" atosm"` (ii)`"1 g Na"=(1)/(23)" mol "=(1)/(23)xx6.02xx10^(23)" atosm"` (III) `"1 g LI"=(1)/(7)" mol "=(1)/(7)xx6.02xx10^(23)" atosm"` (iv)`"1 g Cl"_(2)=(1)/(71)" mol "=(1)/(71)xx6.02xx10^(23)" molecules"=(2)/(71)xx6.02xx10^(23)" atoms"` |
|
| 35. |
Why are reactions of higher order less in number ? |
| Answer» Solution :A reaction takes PLACE because the molecules collide. The chances for a LARGE number of molecules or IONS to collide simultaneously are less. Hence, the reactions of higher ORDER are less. | |
| 36. |
What is collision frequency ? |
| Answer» Solution :It is the NUMBER of collisions PER SECOND per UNIT VOLUME of the reaction mixture. | |
| 37. |
Which of the following haloakanes reacts with aqueous KOH most easily ? Explain giving reason.(i) 1-Bromobutane(ii) 2-Bromobutane(iii) 2-Bromo-2-methylpropane(iv) 2-Chlorobutane |
Answer» Solution :2-Bromo-2-methylpropane will react most EASILY with aqueous KOH because being `3^(@)`- alkyl HALIDE it follows `S_(N)1` path which results in FORMATION of a STABLE `3^(@)` - carbocation intermedate.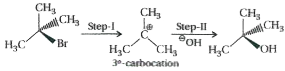
|
|
| 38. |
Which of the following is INCORRECT according to valence bond theory (VBT) ? |
|
Answer» The bond FORMED between metal atom/ion is purely ionic. |
|
| 39. |
the subshell that arlses after fsubshell is called g subshell then the correct statement (s)regarding g subshell is/are (a) it contains 16electrons and 8orbitals, (b) corresponds to l= 4 and first of course in 5th energy level, (c) a g orbital can have maximum of two electrons (d) 5f subshellhas higher energy than 5g subshell |
|
Answer» Only (a) |
|
| 40. |
Which of the following was previously known as muriatic acid |
|
Answer» `Cl_2` |
|
| 42. |
Which one of the following reaction is Swart reaction ? |
|
Answer» `C_2 H_5Cl +Ag f toC_2 H_5 F+KCL` <BR>`C_2 H_5Cl+NaBr toC_2 H_5 Br +NaCl` |
|
| 43. |
Which of the following cannot be accelerated |
| Answer» Answer :A | |
| 44. |
Total number of structural isomeric amines having molecular formula C_(4)H_(11)N are |
|
Answer» |
|
| 45. |
Which one is unimolecularreaction : |
|
Answer» `NH_4NO_2rarrN_2+2H_2O` |
|
| 46. |
Which element is used because for making filaments of electricbulbs and why? |
| Answer» SOLUTION :TUNGSTEN is usedin making FILAMENTS of electric bulbs because if has very high MELTING point. | |
| 48. |
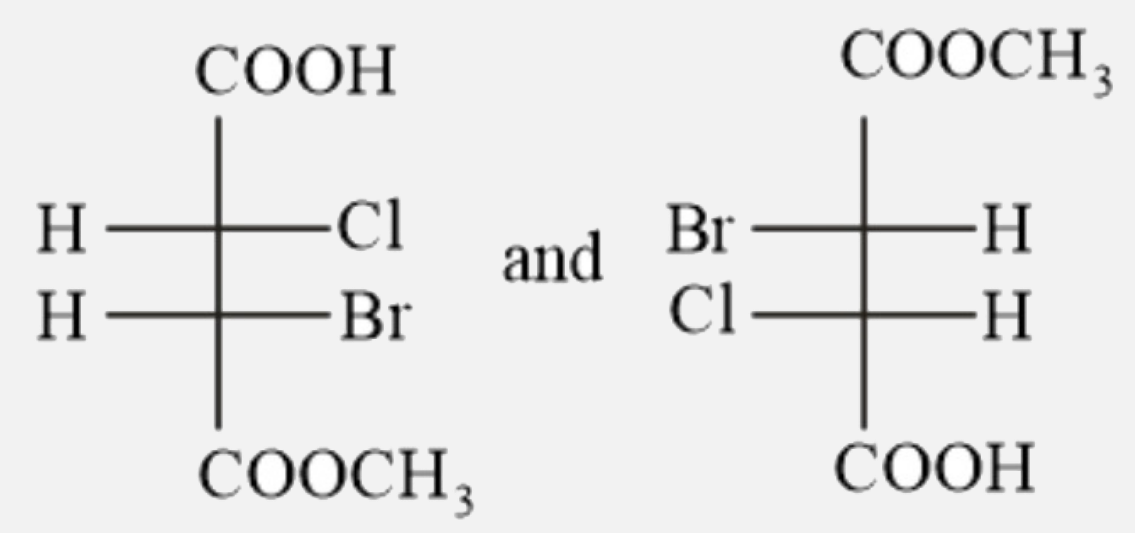
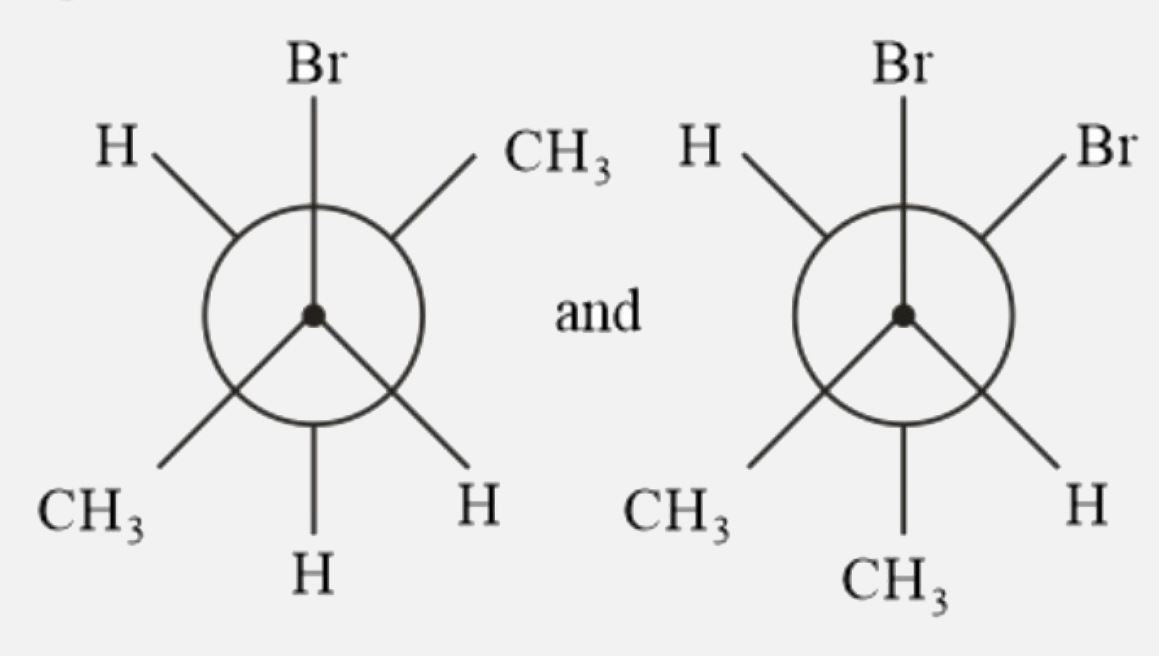
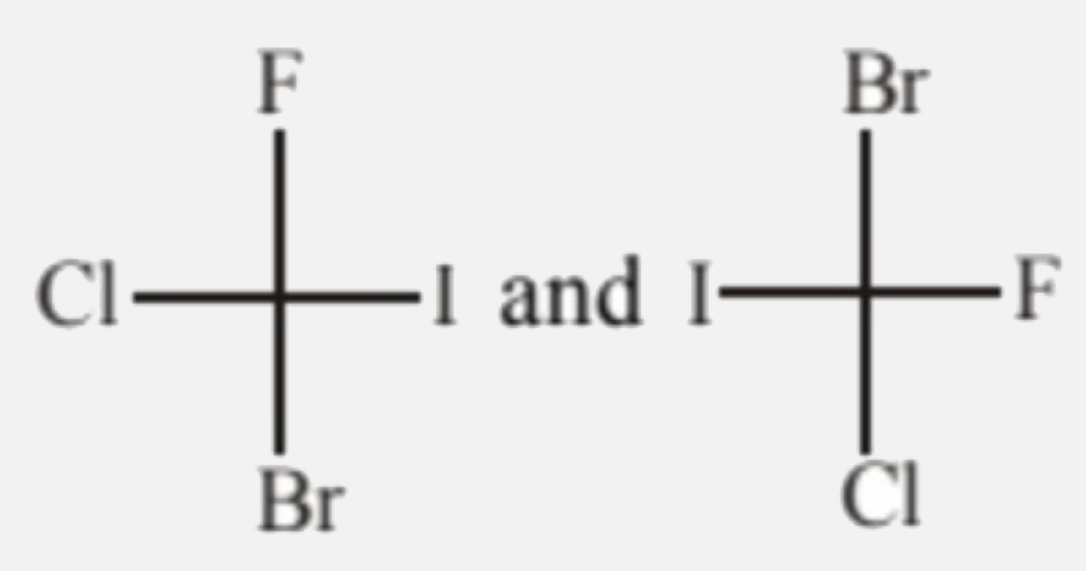
Which of the following pairs are enantiomers? |
|
Answer»
|
|
| 49. |
Which one of the following is a reduction reaction |
|
Answer» `2Mg(s)+O_(2)rarr2Mg(s)` |
|