Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which reagent can be used to distinguish O_(3)and H_2O_2 ? |
|
Answer» Solution :`G_(2)O_(2)`reduces `KMnO_4` `H_2O_2` ACTS as both oxidizing and reducing agent while `O_(3)`acts only as oxidizing agent. |
|
| 3. |
There is no effect of temparature on the mole fraction of a solution . |
| Answer» | |
| 4. |
Which one of the following sets of vitamins is fat soluble? |
|
Answer» `C,D,B_6,B_(12)` |
|
| 5. |
When H_(2) reacts with Na, it acts as |
|
Answer» OXIDISING agent |
|
| 6. |
Which of the following has highest octane number? |
|
Answer» 2,2,4-Trimethylpentane |
|
| 7. |
Which of the following is also known as ''Fools gold'' |
|
Answer» Wurtzite |
|
| 8. |
Which among the followingis NOT CORRECT order of boiling point ? |
|
Answer» Ethyl ALCOHOL ` gt` ethylamine ` gt` ETHANE |
|
| 9. |
Which of the following is called mixed anhydride ? |
| Answer» Answer :A | |
| 12. |
Which of the following is isoelectronic pair ? |
|
Answer» `ICl_(2), ClO_(2)` |
|
| 13. |
Which of the following parameters of a chemical readtion are increased when a catalyst is used? |
|
Answer» RATE & ACTIVATION energy |
|
| 14. |
Vitamin D is called |
|
Answer» ASCORBIC acid |
|
| 15. |
The volume of 10 N and 4 N HCl required to make 1 L of 7 N HCl are |
|
Answer» 0.50 L of 10 N HCl and 0.50 L of 4 N HCI volume of 4N required = 1 - x `N_1V_1 + N_2V_2 = N_3V_3` `10 xx x +4 xx (1 - x) = 7 xx 1` ` 10 x + 4 - 4X = 7` `6x = 3` ` x = 3//6 = 0.5 L` volume of 10N HCl required = 0.5 L volume of 4N HCl required = 1 - 0.5 = 0.5L |
|
| 16. |
What is correct about P_(10). |
|
Answer» It can not GIVE Bromine WATER test 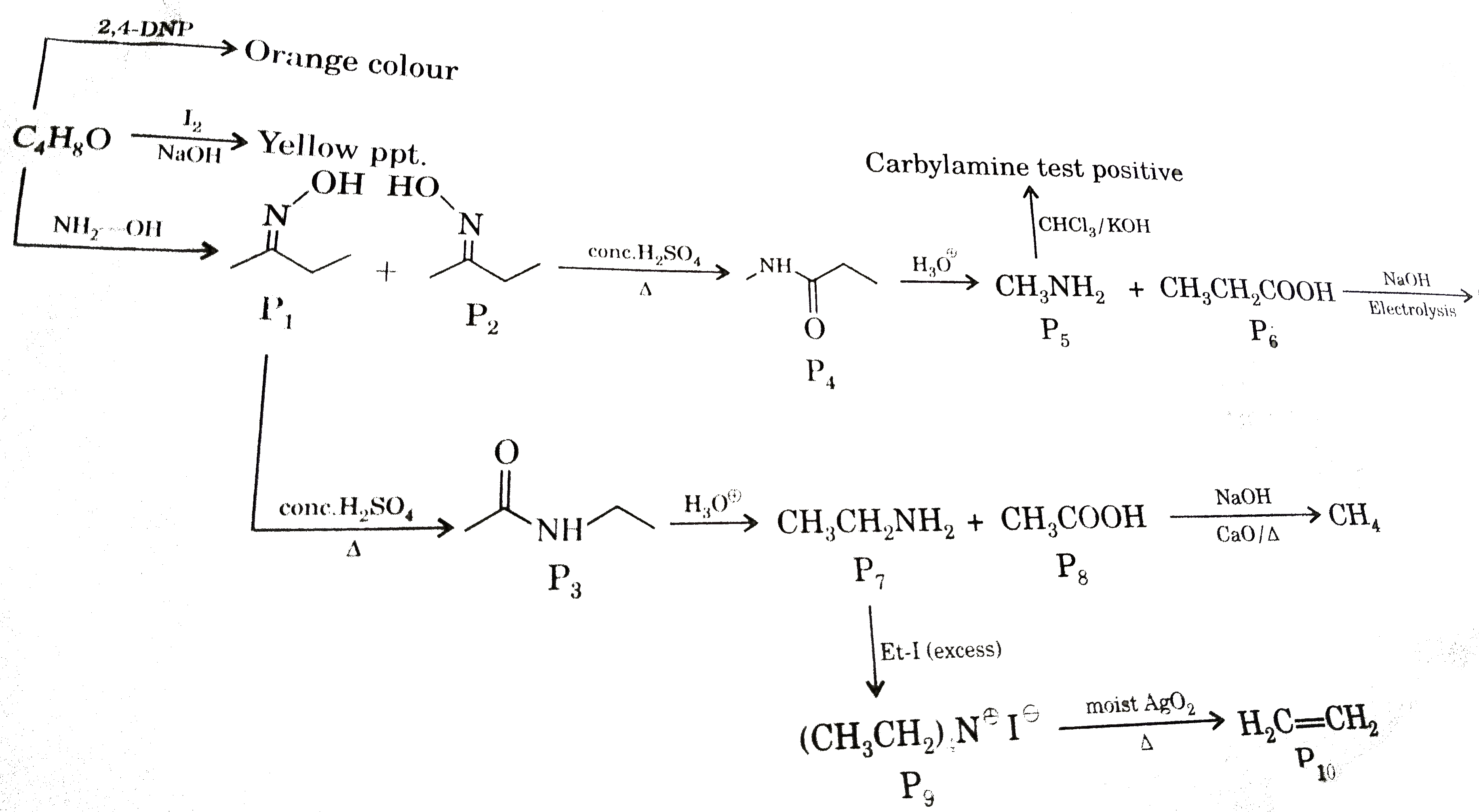
|
|
| 17. |
Who proposed the concept, that solute particles in solution behaves like gaseous molecules |
|
Answer» Boyle |
|
| 18. |
White phosphorus molecule (P_(4)) doesn't have |
|
Answer» Six P-P SINGLE BONDS |
|
| 19. |
Which of the statements regarding formation of compound A is correct ? |
|
Answer» EPOXIDE ring opening is by TRIMOLECULAR reaction |
|
| 20. |
Which of the following is not considered to be apollutant? |
|
Answer» `NO_2` |
|
| 21. |
What are the common types of defects in solids ? |
| Answer» SOLUTION :Solids are not PERFECT in structure. There are different types of imperfections or defects in them. POINT defects ANDLINE defects are COMMON types of defects. | |
| 22. |
The water is electrolysed in a cell, hydrogen is liberated at one electrode and oxygen is simultaneously liberated at the other. In a particular experiment hydrogen and oxygen so produced were collected together and the total volume measured 16.8 mL at NTP. How many coulombs were passed through the cell in the experiment ? |
| Answer» SOLUTION :96.5 COULOMBS | |
| 23. |
Which of the following can be obtained from napth? |
|
Answer» Parffin wax |
|
| 24. |
Which one of the following complex ions has geometrical isomers ? |
|
Answer» `[Co(en)_(3)]^(3+)` 
|
|
| 25. |
Which defines the standard reduction electrode potential of Zn^(2+) ions? |
|
Answer» `Zn_((aq))^(2+) + 2e RARR Zn_((s)) , [Zn^(2+)]=1M` |
|
| 26. |
Which of the following has high vapour pressure at temperature below its melting point? |
|
Answer» Citric ACID |
|
| 27. |
What will happen during the electrolysis of aqueous solution of CuSO_4 in the presence of Cu electrodes ? |
|
Answer» COPPER will deposit at CATHODE. |
|
| 28. |
Whichofthe followingin unstable |
|
Answer» `C_(6)H_(5) -N_(2)Br` |
|
| 29. |
When dilute ferrous (Fe^(2+)) salt solution is added to an aqeous solution containing nitrate (NO_(3)^(-)) ion followed by the addition of conc. H_(2)SO_(4) forms a brown colored ring. What is te chemical composition of the complex that is responsible for this brown colored ring. |
|
Answer» `Fe(NO_(3))_(2)` |
|
| 30. |
Which of the following crystals has unit cell such that a ne b ne c and alpha ne beta ne gamma ne 90^@ ? |
|
Answer» `K_2Cr_2O_7` |
|
| 31. |
You are given 10^(-5)MNaCl solution and 10^(-8)MAgNO_(3) solution.They are mixed in 1:1 volume ratio predict whether AgCl will be precipitated or not ,if solubility product (Ksp) of AgCl=10^(-10) |
|
Answer» SOLUTION :Upon mixing equal volumes ,volume of solution will get doubled.so CONCENTREATION of each ion will get HALVED from original VALUE. ionic product `K_(lp)=[Ag^(+)][Cl^(-)]_("upon mixing")=(10^(-5))/(2)xx(10^(-5))/(2)=25xx10^(-15)( ltK_(sp))` Hence ,no precipitaion will take place. |
|
| 32. |
Which of the following compounds will behave as a reducing sugar in an aqueous KOH solution? |
|
Answer»

|
|
| 34. |
What may be expected to happen when phosphine gas is mixed with chlorine gas? |
|
Answer» The mixure only COOLS down |
|
| 35. |
Which of the following order is wrong ? |
|
Answer» `H_2OgtH_2SgtH_2SegtH_2Te` (Thermal STABILITY) |
|
| 36. |
The synthetic steroid ethynylestradiol (1) is a compound used in the birth control pill. A. How many sp^(3) hybridised carbon atoms are present in compound (1) ? (a) 8(b) 9(c ) 10 (d) 11(e ) 12 B. Hiow many sp^(2) hybridised carbon atoms are present in compound (1) ? (a) 4 (b) 5 (c ) 6 (d) 7(e ) 8 C. How many sp hybridised carbon atoms are present in compound (1) ? (a) 2(b) 4 (c ) 6 (d) 8 (e ) 10 D. Which of the following functional group is contained in compound (1) ? (a) A ketone(b) An alcohol(c ) A carboxylic acid(d) An ester E. How many asymmetric (stereogenic) centres are present in compound (1) ? (a) 2(b) 3 (c ) 4 (d) 5 |
|
Answer» |
|
| 37. |
The relation between osmotic pressure, concentration and temperature of a solution is given by: |
|
Answer» `PI = CRT` |
|
| 38. |
The rms velocity of CO gas molecules at 27^(@)C is approximately 1000 m/s. For N_(2) molecules at 600 K the rms velocity is approximately |
|
Answer» 2000 m/s `(C_(CO))/(C_(N_(2)))=sqrt((3RT_(CO))/(M_(CO))xx(M_(N_(2)))/(3RTN_(2)))` `1000/(C_(N_(2)))=sqrt(300/28xx28/600)impliesC_(N_(2))=1000xxsqrt(2)` `C_(N_(2))=1000xx1.414=1414m//s` |
|
| 39. |
Which of the following complexes are tetrahedral but spin free ? |
|
Answer» `[NiCl_(4)]^(2-)` |
|
| 40. |
Which pair (s) has some bond angle ? (a)BF_(3),BCl_(3) (b) PO_(4)^(-3),SO_(4)^(-2) ( c) BF_(3),PF_(3) (d) NO_(2)^(+),N_(2)O (e) N_(3)^(-),NO_(2) correct option are - |
| Answer» Answer :A | |
| 41. |
What are micelles ? Give an example of a micelles system. |
|
Answer» SOLUTION :The aggregate of colloidal particle which have both hydrophobic and hydrophilic parts are CALLED micelles. These are formed above a particular temperature called Kraft.s temperature `(T_k)` and above certain concentrations. called Critcal Micelles Concentration (CMC). These molecules are arranged radially with the HYDROCARBON or non-polar part towards the centre and the polar part towards the PERIPHERY, e.i., soap solution in WATER is an example of micelles system. |
|
| 42. |
The stabilization of the dispersed phase in a lyophobic sol is due to: |
|
Answer» The VISCOSITY of the medium 
|
|
| 43. |
Which of the following is the weakest Bronsted base ? |
|
Answer»
|
|
| 44. |
Which would not give a precipitate with dilute AgNO_3 solution ? |
|
Answer» `HNO_3` |
|
| 45. |
Thermodynamically graphite is more stable than diamond but diamond does not transform into graphite on its own. Why? |
| Answer» SOLUTION :This conversion is not favoured by KINETIC FACTORS (the activation ENERGY for this is very HIGH). | |
| 46. |
You are given 6 identical balls . The maximum number of square voids and triangular voids (in separate arrangements ) that can be created respectively are |
| Answer» ANSWER :A | |
| 47. |
Which among the following reaction involves nucleophilic addition followed by elimination of water ? |
|
Answer» `CH_3COCH_3+HCN OVERSET(""^(-)OH)(to)` 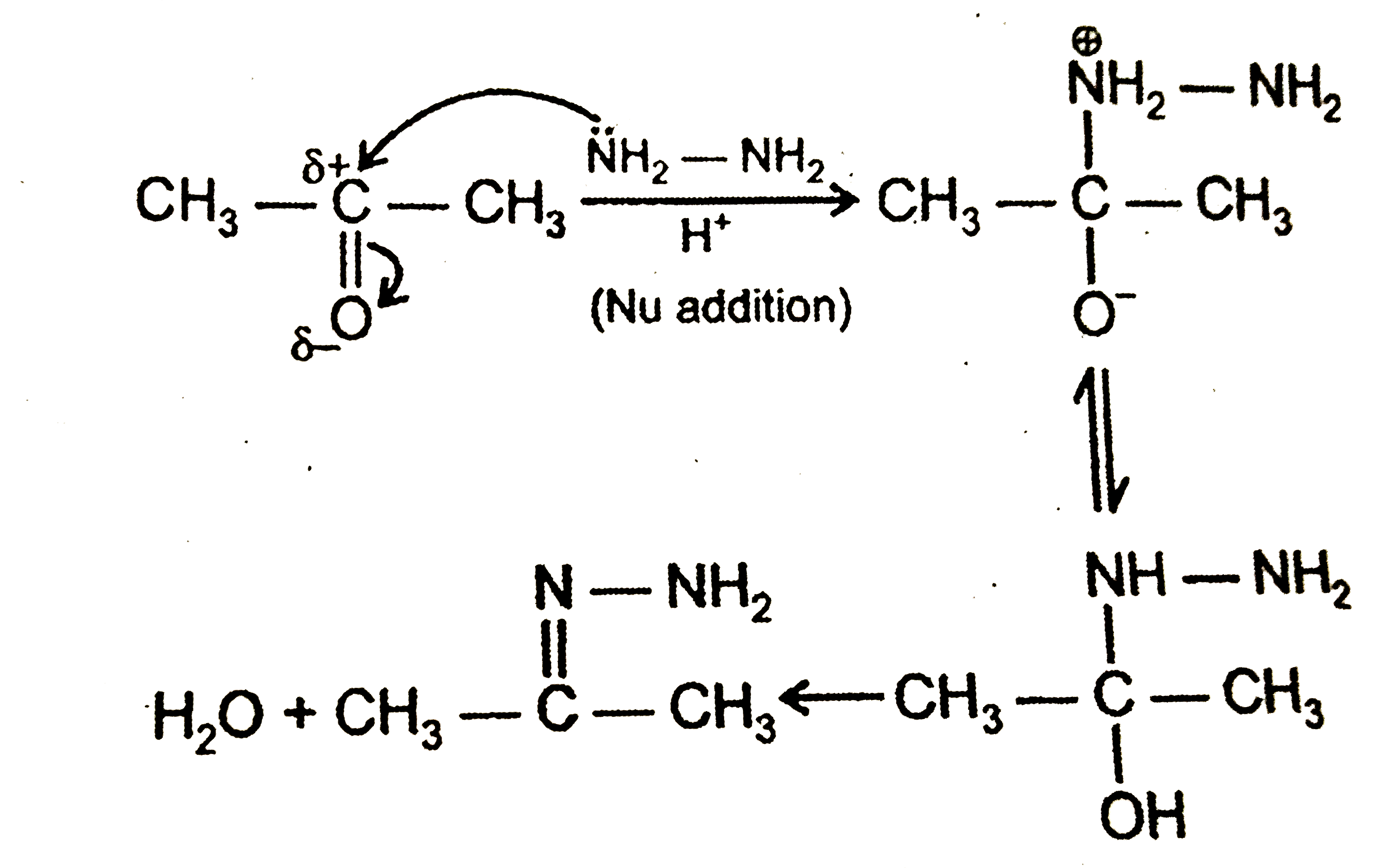
|
|
| 48. |
Which of the following compounds when heated with CO at 150^@C and 500 atm pressurein presence of BF_3 formsethyl propionate ? |
|
Answer» `C_2H_5OH` `C_2H_5-O-C_2H_5+CO underset"500 atm"OVERSET(BF_3,150^@C)to underset"ethyl propionate"(C_2H_5COOC_2H_5)` |
|
| 49. |
Thermosetting polymer , bukelite is formed by the reaction of phenol with: |
|
Answer» `CH_3CHO` |
|