Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The hydration energy of Mg^(2+) ions is larger than that of: |
|
Answer» `AL^(3+)` |
|
| 2. |
The hydration energy of Mg^(++) is larger than that of |
|
Answer» `Al^(+3)` |
|
| 3. |
The hydrated copper (II) ion is..............in colour as it absorbs................light and transmit its complementary colour. |
|
Answer» |
|
| 4. |
The hybridized state of Al^(3+) in the complex ion formed when AlCl_3 is treated with aqueous acid is |
|
Answer» `sp^3` |
|
| 5. |
The hydbridization state of carbon atoms in the product formed by the reaction of ethyl chloride with aqueous potassium hydroxide is:- |
|
Answer» SP |
|
| 6. |
The hydrated cupric chloride is strongly heated.Which of the following statements is (are) correct for this ? |
|
Answer» It is reduced to `Cu_2Cl_2` |
|
| 7. |
The hybridization states of the central atom in the complexes [Fe(CN)_(6)]^(3-),[Fe(CN)_(6)]^(4-) and [Co(NO_(2))_(6)]^(3-) are: |
|
Answer» `d^(2)sp^(3),sp^(3)d^(2) and DSP^(2)` respectively |
|
| 8. |
The hybridization of the complex [CrCl_(2)(NO_(2))_(2)(NH_(3))_(2)]^(-) is: |
|
Answer» `SP^(3)d^(2)` 
|
|
| 9. |
The hybridization states of [Ni(CO)_(4)], [Ni(CN)_(4)]^(2-) and [NiCl_(4)]^(2-) species are respectively |
|
Answer» `sp^(3), sp^(3), dsp^(2)` `[Ni(CN)_(4)]` is square planar : `dsp^(2)` hybridisation `[NiCl_(4)]^(2-)` is tetrahedral : `sp^(3)` hybridisation |
|
| 10. |
The hybridization state of carbon atoms in the product formed by the reaction of ethyl chloride with aqueous KOH is |
|
Answer» SP |
|
| 11. |
The hybridization of the central atom will change when : |
|
Answer» `NH_(3)` combines with `H^(+)` 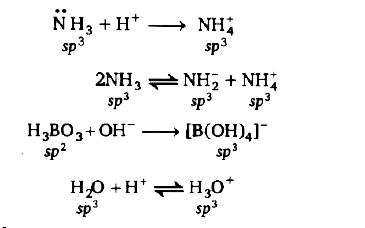
|
|
| 15. |
The hybridization of Sin SF_(4) is |
|
Answer» `SP ^(3) d ^(2)` |
|
| 16. |
The hybridization of [CoF_(6)]^(3-) and [Co(C_(2)O_(4))}^(3-) are : |
|
Answer» both `SP^(3)d^(2)` |
|
| 17. |
The hybridization of carbon attached to chlorine in benzyl chloride is : |
|
Answer» `DSP^(2)` |
|
| 18. |
The hybridization of boron in the stable borane having thelowest molecular weight is- |
|
Answer» `SP^(2) ` |
|
| 19. |
The hybridization involved in the complex [Ni(CN)_(4)]^(2-) is (At. No. of Ni = 28) |
|
Answer» `d^(2)sp^(2)`  (In PRESENCE of strong field LIGAND `CN^(-)`, electrons in d-orbitals pair up) |
|
| 20. |
The hybridization involved in the complex[Ni(CN)_4]^(2-) is (At. No. Ni = 28) |
|
Answer» `d^2sp^2` |
|
| 21. |
The hybridization involved in the complex [Ni(CN)_(4)]^(2-) is………… |
|
Answer» `SP^(3)` |
|
| 22. |
The hybridization in Icl_(7) is |
|
Answer» `sp^(3)d^(3)` |
|
| 23. |
The hybridization in interhalogen compound AX_7 is |
|
Answer» `SP^(3)d^(3)` |
|
| 24. |
The hybridization in ICI_(7) ( AX_(7)) is |
|
Answer» `SP^(3)d^(3)` |
|
| 25. |
The hybridised orbitals used by silver in the complex [Ag(NH_(3))_(2)]^(+) are of the type: |
| Answer» ANSWER :B | |
| 26. |
The hybridised orbitals are……….. And their orientation in space gives a definite……… to the complex ion. |
|
Answer» |
|
| 27. |
The hybridisation that can account for the shape of ClF_(3) molecule is : |
|
Answer» 1. `DSP^(2)` |
|
| 28. |
The hybridisation take place in [FeF_(6)]^(4-) and [Fe(H_(2)O)_(6)]^(2+) is ................. |
|
Answer» |
|
| 29. |
The hybridisation possessed by oxygen atom in OF_(2) molecule is : |
|
Answer» SP |
|
| 30. |
The hybridisation state of Mn^(7+) is permanganate ion is…… |
|
Answer» `sp_(2)` HYBRIDISATION |
|
| 31. |
The hybridisation state of carbon in fullerence is |
|
Answer» SP |
|
| 32. |
The hybridisation of Xe and Sb in product when XeF_(4) react with SbF_(5) :- |
|
Answer» `SP^(3)d, sp^(3)d^(2)` |
|
| 33. |
The hybridisation of sulphur atom in SCl_(4) and the shape of the molecule are |
|
Answer» `sp^(3)` and TETRAHEDRAL |
|
| 34. |
The hybridisation of phosphorusin phosphorus pentachlride is |
|
Answer» `dsp^(3)` |
|
| 35. |
The hybridisation of [PtCl_6]^(2-) ion is: |
|
Answer» `d^2sp^3` |
|
| 36. |
The hybridisation of P in PO_4^(3-) is same as in : |
|
Answer» I in `ICI_4^-` |
|
| 37. |
The hybridisation of orbitals of N atom in NO_(3)^(-), NO_(2)^(+) and NH_(4)^(+) are respectively : |
|
Answer» `SP, sp^(3), sp^(2)` `NO_(3)^(-)(sp^(2)), NO_(2)^(+)(sp), NO_(4)^(+)(sp^(3))` |
|
| 38. |
The hybridisation of [Ni(CN)_4]^2- ion is: |
|
Answer» `dsp^2` |
|
| 39. |
The hybridisation of [Ni(CN)_4]^-2 ion is: |
|
Answer» `dsp^2` |
|
| 40. |
The hybridisation of nickel in tetracarbonyl nickel is |
|
Answer» `dsp^2` |
|
| 41. |
The hybridisation of N in O _(2) ^(+), NO _(3) ^(-)and NH _(4) ^(+) respectively is |
|
Answer» `SP^(2), sp^(3), sp` |
|
| 43. |
The hybridisation of Iron in K_(4)[Fe(CN)_(6)] is |
|
Answer» `dsp^2` |
|
| 44. |
The hybridisation of I in IC l_(2)^(-),IC l_(4)^(-) are respectively: |
|
Answer» `SP^(3), sp^(3)` |
|
| 45. |
The hybridisation of Cu in (NH_(4))_(2)[CuCl_(4)] and Cs_(2)[CuCl_(4)] is |
|
Answer» `DSP^(2)` in both |
|
| 46. |
The hybridisation of Fe in K_(4)[Fe(CN)_(6)] is |
|
Answer» `DSP^(2)` |
|
| 47. |
The hybridisation of Fe in K_4[Fe(CN)_6] complex is: |
|
Answer» `d^2sp^2` |
|
| 48. |
The hybridisation of [CoF_(6)]^(3-) & [Co(C_(2)O_(4))_(3)]^(3-) are :- |
|
Answer» Both `SP^(3)d^(2)` `[Co(O X)_(3)]^(3-)`, hybridisation `=d^(2)sp^(3)` |
|
| 49. |
The hybridisation of central metal ion and shape of Wilkinson's catalyst is |
|
Answer» `dsp^(2)`, square planar |
|