Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The amount of energy required to remove an electron from the last orbit of an isolated (free) atom in gaseous state is known as ionization potential or first ionization potential of the element. Similarly, the energy required for the removal of the electron from the unipositive ion (produced above) is referred to as second ionization potential and thus the third, fourth etc. lonization potentials may be defined in general as the number of ionization potentials of an element may be as much as its number of electrons. Ionization energy of nitrogen is more than that of oxygen which can be explained by |
|
Answer» high effective NUCLEAR charge |
|
| 2. |
The amount of energy required to remove an electron from the last orbit of an isolated (free) atom in gaseous state is known as ionization potential or first ionization potential of the element. Similarly, the energy required for the removal of the electron from the unipositive ion (produced above) is referred to as second ionization potential and thus the third, fourth etc. lonization potentials may be defined in general as the number of ionization potentials of an element may be as much as its number of electrons. The realationship between IE _(3) and IE_(1) of element is |
|
Answer» `IE _(3) GT IE _(1)` |
|
| 3. |
The amount of energy required to remove an electron from the last orbit of an isolated (free) atom in gaseous state is known as ionization potential or first ionization potential of the element. Similarly, the energy required for the removal of the electron from the unipositive ion (produced above) is referred to as second ionization potential and thus the third, fourth etc. lonization potentials may be defined in general as the number of ionization potentials of an element may be as much as its number of electrons. Ionization energy increases on moving down a group due to |
|
Answer» increase in the number of ELECTRONS |
|
| 4. |
The amount of energy released when 20 mL of 0.5 M NH_4OH are mixed with 100 mL of 0.1 M HCl is x kJ. The heat of dissociation of NH_4OH will be (heat of neutralization of NaOH & HCl is y kJ/mol). |
|
Answer» `-100 x` `:.` For 1 mole `implies 100 xx kJ` heat is released `:. Delta H = -100 xx kJ//MOL` For 1 mole `NAOH + 1"mole" HCl, Delta H = y kJ //mol "":. ""` Heat of DISSOCIATION of `NH_(4)OH = -100x - y`. |
|
| 5. |
the amount of energy released when 10^(6) atoms of iodine in vapour state are converted to I^(-) ions is 4.8 xx 10^(-13) J. What is the electron affinity of iodine in ev/atom. |
|
Answer» `2.0` `THEREFORE` Energy released for 1 ATOM `=(4.9xx10^(-13))/(10^(6))J` `=(4.9xx10^(-13)xx10^(-6))/(1.6xx10^(-19))` `=3.06eV`. |
|
| 6. |
The amount of energy expanded during the passage of one ampere current for 100 second under a potential of 115 V is: |
|
Answer» `20kJ` |
|
| 7. |
The amount of electrolytes required to coagulate a given amount of AgI colloidal solutio n (-ve charge) will be in the order |
|
Answer» `NaNO_(3) gt AL (NO_3)_3 gt Ba(NO_3)_2` Quantity of electrolyte ` PROP (1)/("charge on the cation/anion")` To coagulate AgI, `I^(-)` (-ve charge)is to be coagulated. `Al (NO_3)_3 rarr 2 Al^(3+) +3NO_(3)^(-)` `Ba(NO_3)_2 rarr Ba^(2) +2NO_(3)^(-)` `NaNO_(3) rarr NA^(+) + NO_(3)^(-)` Charge on the cation decreases in the order : `Al^(3+) gt Ba^(2+) gt Na^(-)` Thus, the amountof electrolytes required will be in the order. `NaNO_(3) gt Ba(NO_3)_2 gt Al (NO_3)_3` |
|
| 8. |
The amountof electrolytes required to coagulate a give amountof Agl colloidal solution (-ve charge) will be in the order . |
|
Answer» `NaNO_(3) gtAl(NO_(3))_(3) GT Ba(NO_(3))_(2)` To coagulate `AgI,I^(-) (-ve "charge")` is to be coagulated . `Al(NO_(3))_(3) to 2Al^(3+) + 2NO_(3)^(-)` `Ba(NO_(3))_(2) to Ba^(2+) +2NO_(3)^(-)` `NaCO_(4) to Na^(+) + NO_(3)^(-)` Chargeon thecationdecrease in the order . `Al^(3+) gt Ba^(2+) gt Na^(+)` Thus , the AMOUNTOF electrolytes REQUIRED will bethe order `NaNO_(3) gt Ba(NO_(3))_(2) gt Al(NO_(3))_(3)` |
|
| 9. |
The amount of current in Faraday is required for the reduction of 1 mol of Cr_2 O_7^(2-)ions to Cr^(3+) is , |
|
Answer» 1F |
|
| 10. |
The amount of CuSO_(4). 5H_(2) O required to prepare 500 c.c. of 0.5 N solution is - ( Mol. wt. of CuSO_(4). 5H_(2)O = 249.5) |
|
Answer» 249.5 |
|
| 11. |
The amount of copper deposited by the passage of 96500 coulomb of electricity through copper sulphate solution is : |
|
Answer» `2.0 `MOLE |
|
| 12. |
The amount of copper deposited by one Faraday current will be maximum in an acidic solution of one litre of |
|
Answer» 1M `Cu_(2)Cl_(2)` |
|
| 13. |
The amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to |
|
Answer» CURRENT only. |
|
| 14. |
The amount of chlorine evolved when 2 amperes of current is passed for 30 minutes in an aqueoussolution of NaCl |
| Answer» Answer :B | |
| 15. |
The amount of benzene that wil separate out (in grams) if a solution containing 7.32 g of triphenylmethane in 1000g of benzene is cooled to a temperature which is 0.2^(@)C below the freezing point of benzene? (K_(f)=5.12K-Kg//"mol") |
|
Answer» |
|
| 16. |
The amount of anhydrous Na_(2)CO_(3) present in 250 ml of 0.25 M solution is |
|
Answer» 6.225 G |
|
| 17. |
The amount of an ion discharged during electrolysis is directly proportional to : |
|
Answer» RESISTANCE time |
|
| 18. |
The amount of an ion descharged during electrolyses is not dependent of : |
|
Answer» RESISTANCE of solution |
|
| 19. |
The amount of ._(6)^(14)C isotope in a piece of wood is found to one fourth (1/4) of that present in a fresh piece of wood. Calculate the age of the piece of wood (t_(12) "of "_(6)^(14)C=5770 "years") |
|
Answer» 7999 year |
|
| 20. |
The amount of ._(53)I^(128) (t_(1//2) = 25 minutes) left after 50 minutes will be |
|
Answer» One-half = 25 minutes `= (100)/(25) = (1)/(4)` |
|
| 21. |
The amount of ""_(6)^(14)C isotope in a place of wood is found to be one fifth of that present in a fresh piece of wood. Calculate the age of the piece of wood (half-life of ""_(6)^(14)C=5577 year). |
|
Answer» SOLUTION :`t=(2.303)/(lambda)LOG.(N_(0))/(N)` `impliest=(2.303xxt_(1//2))/(0.693)log.(N_(0))/(N)` `=(2.303xx5577)/(0.693)log.(N_(0))/(N_(0)//5)` `(2.303xx5577)/(0.693)xx0.6989=12.953` YEARS |
|
| 22. |
The amorphous solid among the following is |
| Answer» Solution :Glass | |
| 23. |
The ammonia prepared by treating ammoniumsuphate with calcium hydroxideis completelyused by NiCl_(2)* 6H_(2) Oto formstablecoordination compound . Assumethat boththe reactionsare 100%complete.If1584g ofammonium sulphate and 952 g of NiCl_(2) * 6H_(2) O are usedin thepreparation , the combined weight(in gram) of gypsumand thenickel ammoniacoordination compoundthus produced is(H = 1, N = 14, O = 16, s = 32, C1 = 35 . 5, Can= 40, Ni = 59) |
|
Answer» As weightin GRAM = no. of mol `xx` Mol . wt. Wt. of `CaSO_(4)*2H_(2)O("gypusm") + [Ni(NH_(3))_(6)]Cl_(2)` `= (12 xx 172 + 4 xx 232 g)` |
|
| 24. |
The ammonia evolved from the treatment of 0.30g of an organic compound for the estimation of nitrogen was passed in 100ml of 0.1 M H_(2)SO_(4). The excess acid required 20ml of 0.5M NaOH solution for complete neutralisation . The organic compound is: |
|
Answer» Thiourea `underset("acid")(N_(1)V_(1))=underset("base")(N_(2)V_(2))` `0.2xxV_(1)=0.5xx20` `V_(1)=(0.5xx20)/(0.2)=50mL` Volume of `H_(2)SO_(4)` used `=100-50=50mL` `%` of `N` in the COMPOUND `=(1.4xxVxxN)/(W)` `=(1.4xx50xx0.2)/(0.30)=46.6%` The PERCENTAGE of N is same as in urea `(NH_(2)CONH_(2))` Percentage of `N=("MASS of "N)/("Mass of urea")xx100` `=(14xx2)/(60)xx100=46.6%` |
|
| 25. |
The ammonia evolved from the treatment of 0.30 g of an organic compound for the estimation of nitrogen was passed |
|
Answer» Urea `= (100 XX 0.1 xx 2)/(1000) - (20 xx 0.5)/(1000)= 1/100` percent of nitrogen in the unknown ORGANIC compound `= 1/100 xx 14/0.3 xx 100 = 46.6%` percent of nitrogen in urea`(NH_(2))_(2) CO` `= (14 xx 2)/(60) xx 100 = 46.6%` `:.` The compound must be urea. |
|
| 26. |
The ammonia evolved from 0.5g of the organic compound in Kjeldahl's estimation of nitrogen neutralizes 10 ml of 1 M H_2SO_4. Identify the correct statement(s) out of the following: |
|
Answer» Percentage of NITROGEN in the organic compound is 56% |
|
| 27. |
The ammonia evolved from0.5 g of the organic compound in Kjeldahl's estimation of nitrogen neutralizes 10 ml of 1M H_(2)SO_(4), identify the correct statement(s) out of the following: |
|
Answer» Percentage of nitrogen in the organic compound is `56%` |
|
| 28. |
The amino group when attached to the ring makes it highly activating towards electrophilic substitution. Consequently, the electrophilic substitution readily takes place in aniline at all the three ortho and para positions. However on acetylation with acetyl chloride the activation of the ring to amino group is sufficiently reduced and the electrophilic substitution in benzene ring takes place preferably at the para position which is less hindered than the ortho position. Which of the following reagent is used to distinguish between |
|
Answer» Dilute `HCl` |
|
| 29. |
The amino group when attached to the ring makes it highly activating towards electrophilic substitution. Consequently, the electrophilic substitution readily takes place in aniline at all the three ortho and para positions. However on acetylation with acetyl chloride the activation of the ring to amino group is sufficiently reduced and the electrophilic substitution in benzene ring takes place preferably at the para position which is less hindered than the ortho position. 3, 5 dibromotolyene can be best synthesised by |
|
Answer»

|
|
| 30. |
The amino group when attached to the ring makes it highly activating towards electrophilic substitution. Consequently, the electrophilic substitution readily takes place in aniline at all the three ortho and para positions. However on acetylation with acetyl chloride the activation of the ring to amino group is sufficiently reduced and the electrophilic substitution in benzene ring takes place preferably at the para position which is less hindered than the ortho position. Towards electrophilic substitution, most reactive will be |
|
Answer» Nitrobenzene |
|
| 31. |
The amino acids which cannot be synthsised in the body but must be supplied through diet are |
|
Answer» ESSENTIAL AMINO acids |
|
| 32. |
The amino acids, which build up proteins, have both the COOH and NH_2 groups. These amino acids are |
|
Answer» `alpha`-AMINO ACIDS |
|
| 33. |
The amino acid with primary alcohol group is |
|
Answer» CYSTEINE |
|
| 35. |
The amino acid without chiral carbon is |
|
Answer» GLYCINE |
|
| 36. |
The amino acid which is not optically active is |
|
Answer» lactic acid  Except glycine, all `ALPHA`-amino acids are OPTICALLY active and have an asymmetric CARBON atom. Hence each of these amino acids can exist in two optical isomeric forms.  It also possesses an asymmetric carbon atom.Therefore it is also optically active. |
|
| 37. |
The amino acid which has a nonpolar side chain is |
|
Answer» isoleusine |
|
| 38. |
The amino acid which contain a hydroxy group in the side chain |
|
Answer» CYSTEINE |
|
| 39. |
The amino acid in neutral solution exist as………………….. |
| Answer» Solution :Zwitter ion | |
| 40. |
The amino acid chain with a mass of 10000 u is called |
|
Answer» POLYPEPTIDE |
|
| 41. |
The amines that will give off N_(2) upon treatment with NaNO_(2) and dil. H_(2)SO_(4) at 0 to 5^(0)C is(are) |
|
Answer»
|
|
| 42. |
The amine whichformco-ordinatebondwith Lewis acidis /are . |
|
Answer» `RNH_(2)` |
|
| 43. |
The amine which will not liberate nitrogen on reaction with nitrous acid is |
|
Answer» tri-methylamine 
|
|
| 44. |
The amine which reacts with p-toluenesulphonyl chloride to gives a clear solution which on treating with alkali gives insoluble compound is |
|
Answer» `C_(2)H_(5)NH_(2)` 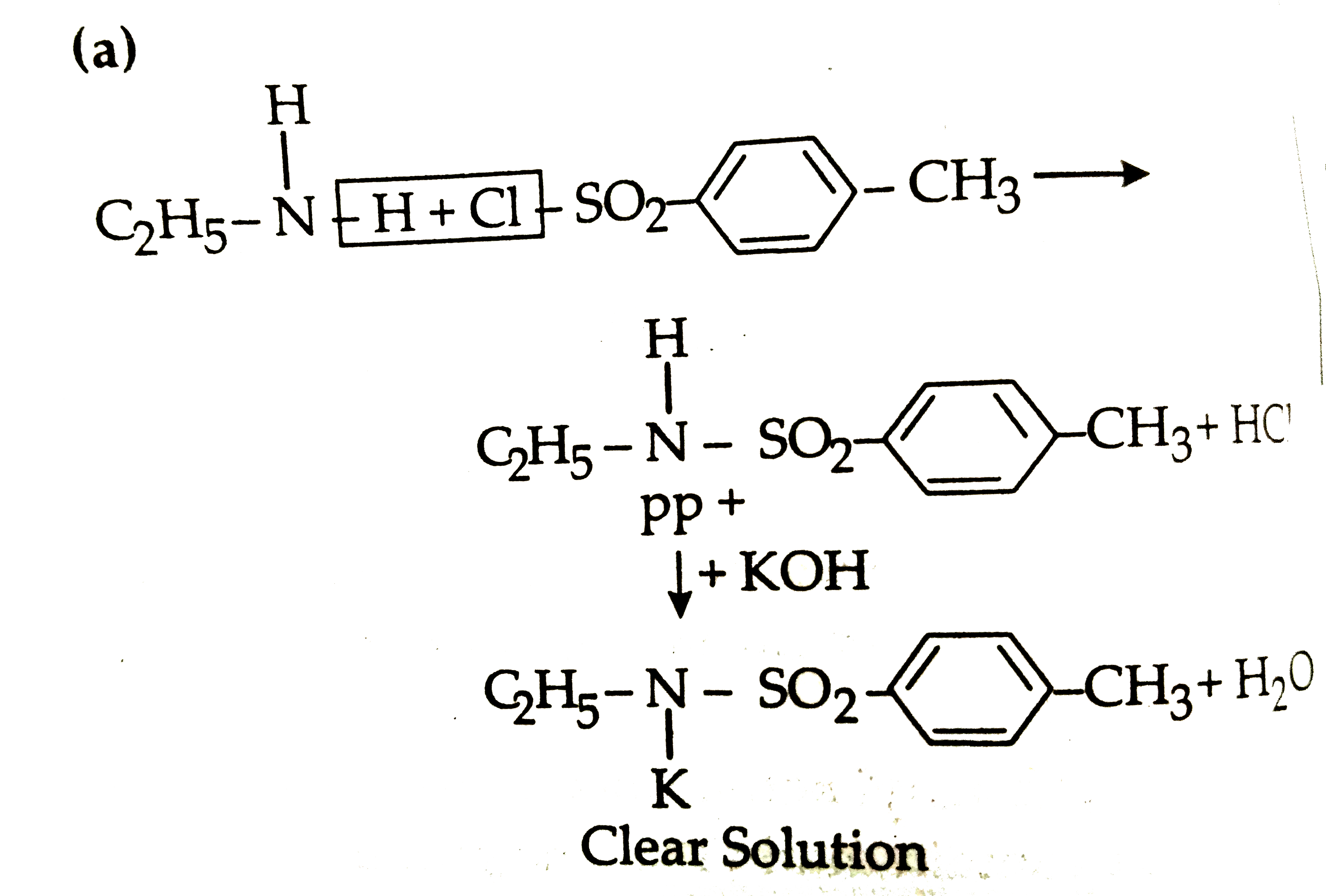
|
|
| 45. |
The amine, which reacts with p-toluenesulphonylchlorideto givea clearsolution, which on acidificationgives insoluble compoundis |
|
Answer» `C_(2)H_(5) NH_(2)` `1^(@)`amineon sulphonationgives a product, which isinsolublein ACID. `2^(@)` amine on sulphonationgivessameproduct butissolublein acid. `3^(@)`amine does notundergo this reaction. 
|
|
| 46. |
The amine which reacts with p-toluenesulphonylchlorideto give a clearsolution which on acidification gives insolublecompound is __________. |
|
Answer» `C_2H_5NH_2` |
|
| 47. |
The aminewhichreacts with p- toluenesulphonylchloride to givea clearsolutionwhich ofacidificationgives insolublecompound is |
|
Answer» `C_(2)H_(5) NH_(2)` |
|
| 48. |
Theaminewhichreactswithnitrous acidsto giveyellowoily compound is : |
|
Answer» Ethyl AMINE |
|
| 49. |
The amine which reacts with nitrous acid to give yellow oily compund is: |
|
Answer» ETHYL amine |
|
| 50. |
The amine salts commonly used for determination of molecular masses of amines arc |
|
Answer» nitrates |
|







