Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Tertiary alkyl halide is practically inert to substitution by S_(N) 2 mechanism because of |
|
Answer» INSOLUBILITY |
|
| 2. |
Tertiary alkyl halide among the following is |
|
Answer» 2 - Chlorobutane |
|
| 3. |
Tertiary alkyl amines on oxidation by KMnO_(4) gives |
|
Answer» `1^(@)` - nitroalkane |
|
| 4. |
Tertiaryalkyl amineis |
|
Answer» `1^(0)` - AMINE |
|
| 5. |
Tertiary alkali halides are practically inert to substitution by SN2 mechanism because of : |
|
Answer» insolubility |
|
| 6. |
Tertiary alcohols when passed over heated copper undergo_____to form_____. |
| Answer» SOLUTION :DEHYDRATION, ALKENES. | |
| 7. |
Tertiary alcohols undergo dehydration by……………… . |
|
Answer» `SN^(1)` MECHANISM |
|
| 8. |
Tertiary alcohols are resistant to oxidation because |
|
Answer» they do not have `alpha`-hydrogen atom |
|
| 9. |
Tertiary alcohol on treatment with cyanide in presence cone. H_(2)SO_(4) gives corresponding primary amine. T reaction is called as: |
|
Answer» SCHMIDT REACTION |
|
| 10. |
Tertiary alcohols are formed by reaction of Grignard reagennt with____or______. |
| Answer» SOLUTION :a KETONE or an ESTER other than FORMIC ester. | |
| 11. |
Tertiary alcohols are difficult to oxidise as ________. |
|
Answer» there is no hydrogen ATOM attached to FUNCTIONAL carbon |
|
| 12. |
Tertiary alcohol is obtained when Grignard reagent reacts with : |
|
Answer» Acetone |
|
| 13. |
Tertiary alcohol is not ordinarily oxidised, but on restrict oxidation, it yields _____ having one_____ carbon atom. |
| Answer» SOLUTION :KETONE, LESS. | |
| 14. |
tert-Butylbromide reacts with aq. NaOH by S_(N)1 mechanism while n - butylbromide reacts by S_(N)2 mechanism. Why ? |
Answer» SOLUTION :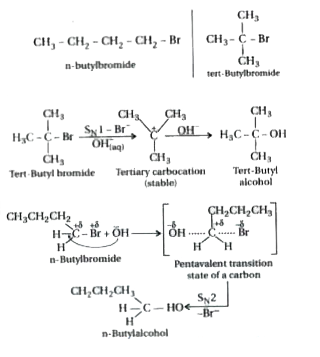
|
|
| 15. |
Tertiary alcohol (3^(@)) having atleast four carbon atoms upon drastic oxidation yield carboxylic acid with |
|
Answer» One carbon atom less 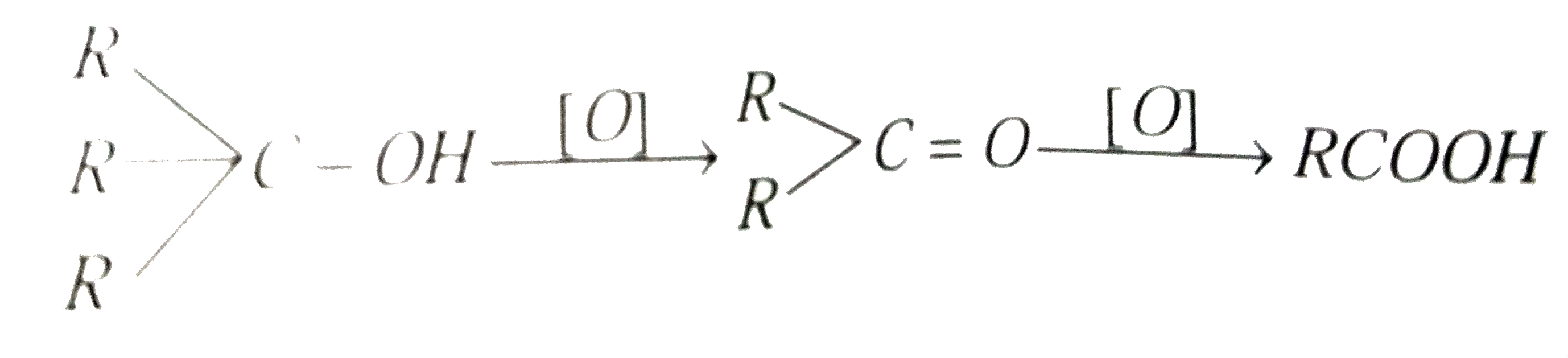 . .
|
|
| 16. |
tertiary alcohol is not ordinarily oxidized, but on restrict oxidation, it yields____ having one______carbon atom. |
| Answer» SOLUTION :KETONE, LESS. | |
| 17. |
Tert-Butylbenzene does not benzoic acid on oxidation with acidic KMnO_(4). Give reason. |
Answer» SOLUTION :Due to absence of benzylic hydrogens, tert-butylbenzene does not undergo oxidation to GIVE benzoic acid while other alkylbenzenes such as toluene, ethylbenzene, n-propylbenzene and isopropylbenzene which CONTAIN one or more benzylic hydrogens (marked as `ALPHA`) undergo oxidation to form benzoic acid. 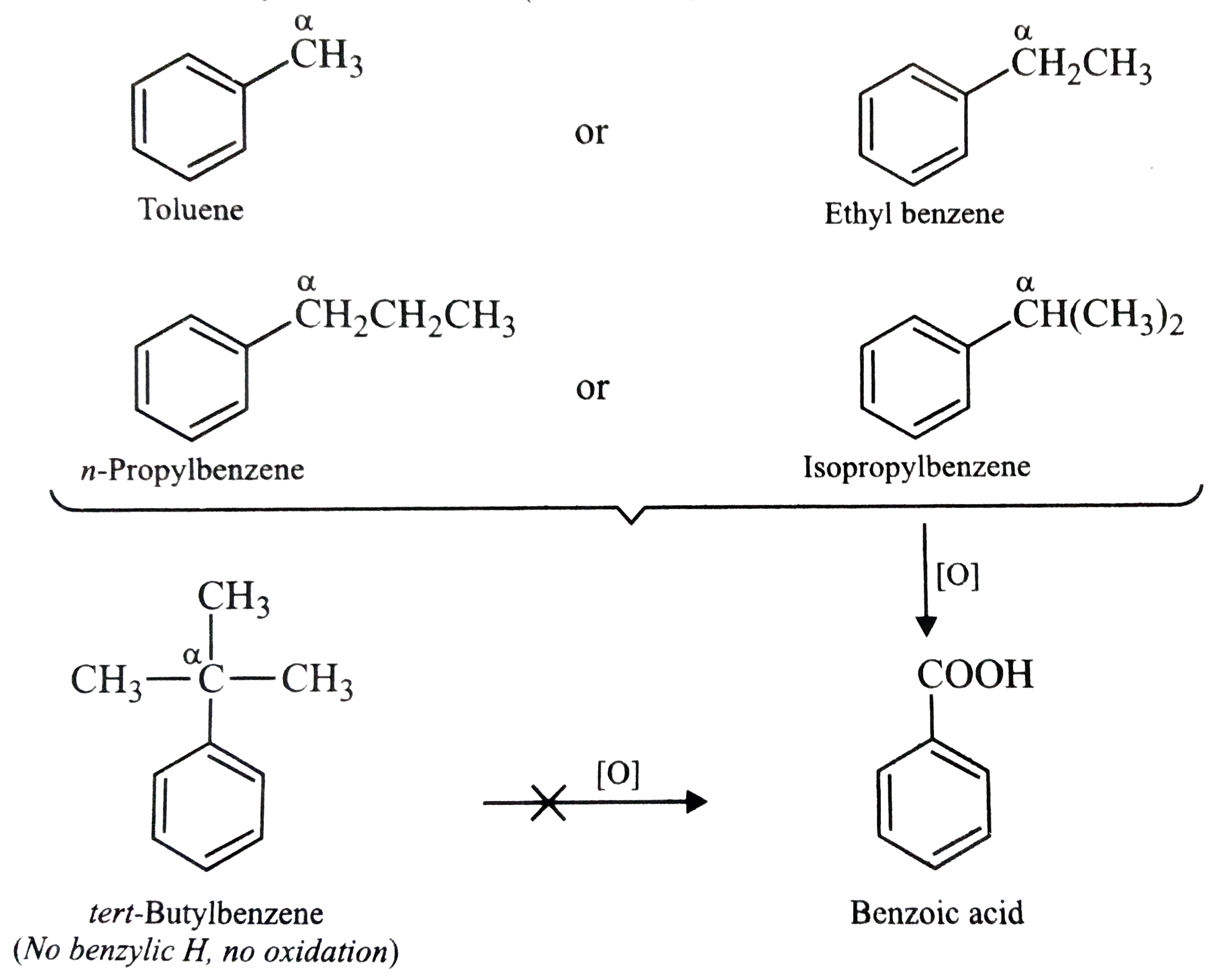
|
|
| 18. |
Tert -Butyl methyl ether on heating with HI of one molar concentration gives |
|
Answer» ` CH_3 OH+ (CH_3) _3 CL` |
|
| 19. |
Tert-butylmethylether ontreatment with hydrogeniodinein coldgives |
|
Answer» tert-butyl iodide and methyl iodide  `S_(N)1` mechanism is followedis followedas `(CH_(3))_(3)C^(+)`is most stable carbocation and `I^(-)` is attachedto it. |
|
| 20. |
Tert-butyl methyl ether on treatment with hydrogen iodide in cold gives |
|
Answer» tert-butyl iodide and METHYL iodide The reaction carried by using `SN^(1)` mechanism. |
|
| 22. |
tert-Butyl methyl ether on heating with HI gives a mixture of |
|
Answer» tert-butyl ALCOHOL and METHYL IODIDE |
|
| 23. |
Tert-butyl chloride to 2-methylpropene |
|
Answer» SOLUTION :Tert-butyl chloride to 2-methyl propene : `CH_(3)-underset(CH_(3))underset("|")overset(CH_(3))overset("|")("C ")-Cl+underset(("alc."))(KOH)overset("HEAT")rarrunderset("2-methyl propene")(CH_(3)-overset(CH_(3))overset("|")("C ")=CH_(2))+KBr+H_(2)O` |
|
| 24. |
Tert butyl ethyl ether is prepared by williamson synthesis byreacting |
|
Answer» SODIUM ethoxide and tert butoxide |
|
| 25. |
tert-Butyl chloride on treatment with sodium alkoxide yields |
|
Answer» an ether |
|
| 26. |
tert-Butyl bromide reacts with aq. NaOH by S_(N)1 mechanism while n-butyl bromide reacts by S_(N)2 mechanism. Why? |
Answer» Solution :tert-Butyl readily loses `Br^(-)` ion to form STABLE tert-butyl carbocation. Therefore, it undergoes reaction by `S_(N)1` mechanism which occurs in TWO steps. In the first step, tert-butyl carbocation is formed. This step is slow and hence is the rate determining step of the reaction. In the second step, the tert-butyl carbocation is readily attacked by `OH^(-)` ion to form tert-butyl alcohol. 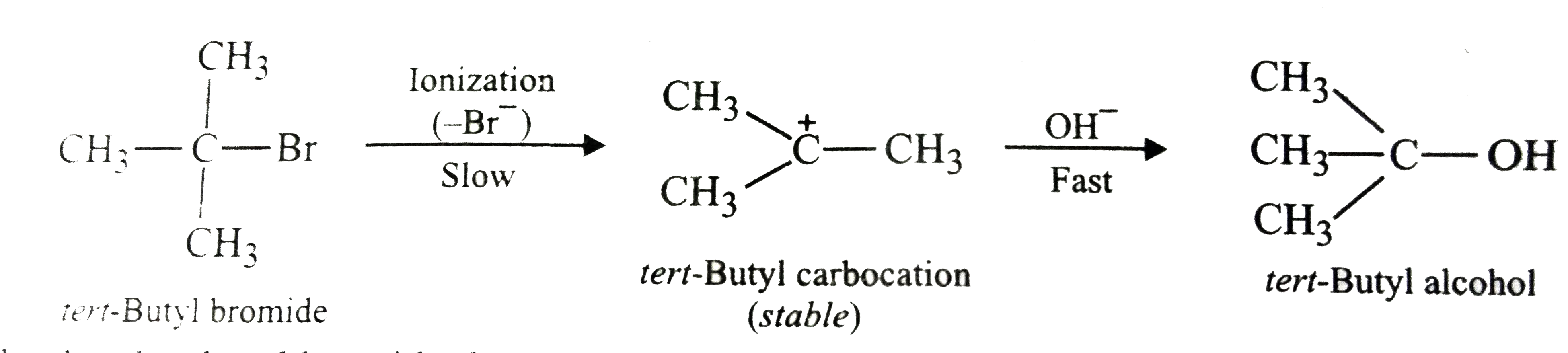 On the other hand, n-butyl bromide does not undergo ionization to PRODUCE n-butyl carbocation because it is not stable. Therefore, it prefers to undergo reaction by `S_(N)2` mechanism which occurs in one step through a transition state involving NUCLEOPHILIC ATTACK of the `OH^(-)` ion from the back side with simultaneous expulsion of `Br^(-)` ion from the front side. 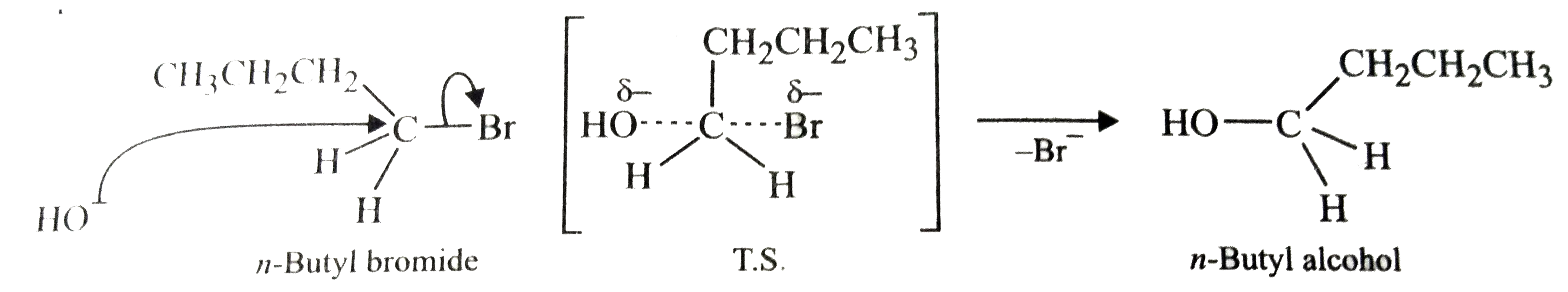
|
|
| 27. |
Write the relavant chemical equations : tert-butyl bromide to isobutyl bromide |
Answer» SOLUTION :
|
|
| 28. |
tert-butyl alcohol is more soluble in water than n-butyl alcohol. |
|
Answer» |
|
| 29. |
tert-Butyl alcohol is much more soluble in water than n-butyl alcohol. Explain. |
| Answer» Solution :In tert-butyl alcohol, ALKYL group is HIGHLY branched and hence APPROACHES almost spherical shape. As a result, non-polar part of the molecule becomes less prominent and hence it can have STRONGER intermolecular hydrogen bonds with water molecules. On the other hand, n-butyl alcohol has predominently non-polar character. | |
| 30. |
Terrorists often use ammonium nitrate fertilizer as an ingredient in car bombs . When ammonium nitrate explodes, it decomposes to gaseous nitrogen , oxygen and water vapour. The force of the explasion results from the sudden production of a huge volume of hot gas. It has been claimed that NH_(4) NO_3 fertilizer (NH_4)_(2)HPO_4 Analysis of such a disensitized sample of NH_(4) NO_3 showed the mass % nitrogen to be 33.81%An explosive mixture called Amatol contains : |
|
Answer» 80% `NH_(4) NO_(3) + 20% ` TNT 
|
|
| 31. |
Terrorists often use ammonium nitrate fertilizer as an ingredient in car bombs . When ammonium nitrate explodes, it decomposes to gaseous nitrogen , oxygen and water vapour. The force of the explasion results from the sudden production of a huge volume of hot gas. It has been claimed that NH_(4) NO_3 fertilizer (NH_4)_(2)HPO_4 Analysis of such a disensitized sample of NH_(4) NO_3 showed the mass % nitrogen to be 33.81%NH_4NO_3 on explosion gives |
|
Answer» `NO_2` |
|
| 32. |
Terpen - 4 - ol is an active ingredient in tea tree oil has the following structure The correct observations for terpen - 4 - ol is/are I. It rotates the plane of plane polarized light. IIIt reacts with baeyer's reagent to form a triol III. On reaction with NaBr and H_2SO_4 , it gives a di bromo compound IV On ozonolysis it gives a compound withmolecular formula C_10H_18O_3. |
|
Answer» I,II,III and IV |
|
| 33. |
Terpenes are |
|
Answer» FOUR RING CYCLIC structure |
|
| 34. |
Termolite is a silicate mineral with fibrous nature and having tetrahedral unit. The molecular formula of tremolite is Ca_(2)Mg_(x)[Si_(8)O_(22)(OH)_(2)] identify the value of x . |
| Answer» | |
| 35. |
Terminal carbon in butane is ____hybridised . |
|
Answer» |
|
| 36. |
Teritiary alkyl halides are practically inert to substitution by S_N2 mechanism because of |
|
Answer» Insolubility |
|
| 37. |
Teritaryalcohols can be prepared using R mg Xwith : |
|
Answer» Formaldehyde |
|
| 38. |
Teritary alcohols are dehydrated by using boiling dilute H_(2)SO_(4) and not con. H_(2)SO_(4) because |
|
Answer» with conc. `H_(2)SO_(4)` teriary alcohol GIVES mixture of isomerric alkenes 
|
|
| 39. |
Terflon is polymer of |
|
Answer» `FCH=CH_2` |
|
| 40. |
Terflon polymer monomer of |
|
Answer» difluoroethene |
|
| 41. |
Terfenadine is commonly used as alan |
|
Answer» Tranquilizer |
|
| 42. |
Terfenadine is commonly used as a/an |
|
Answer» Tranquilizer |
|
| 43. |
Terephthalic acid is obtained by oxidation of which of the following compounds |
|
Answer»
|
|
| 44. |
ter-Butyl methyl ether on heating with HI gives a mixture of |
|
Answer» ter-Butyl ALCOHOL and METHYL iodide |
|
| 45. |
Terephthalic acid is heated with ammonia gives |
|
Answer» PHTHALAMIDE |
|
| 46. |
Terfenadine is commonly used as an |
|
Answer» tranquilizer |
|
| 47. |
ter-Butyl methyl ehter on heating with HI (g) in the presence of ehter gives a mixture of |
|
Answer» ter-Butyl alcohol and methyl iodide |
|
| 48. |
ter-Butyl methyl ehter on heating with anhydrous HI in dry ether gives a mixture of |
|
Answer» ter-Butyl alcohol and METHYL iodide |
|
| 49. |
tendency of forming complex µ charge densityprop(1)/("size of atom") Select those species for which white fumes appear on heating:- BeCl_(2)*4H_(2)O,MgCl_(2)*6H_(2)O,CaCl_(2)*6H_(2)O,SrCl_(2)*6H_(2)O |
|
Answer» 1 |
|
| 50. |
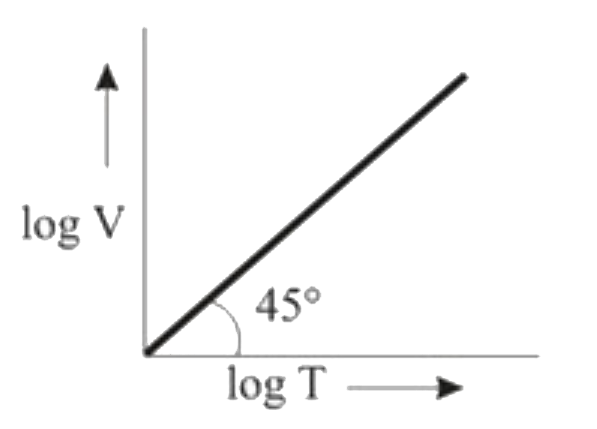
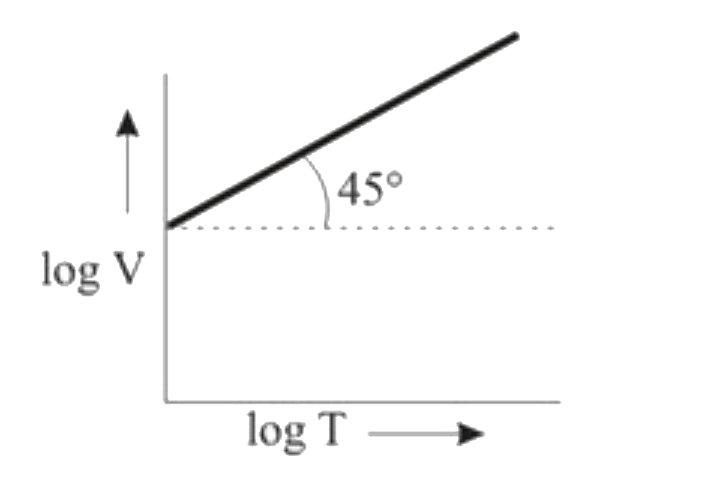
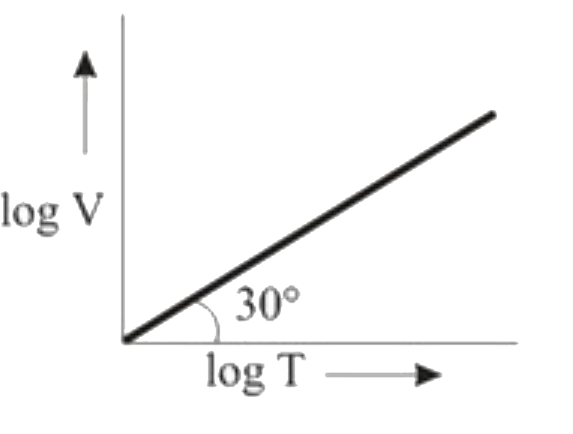
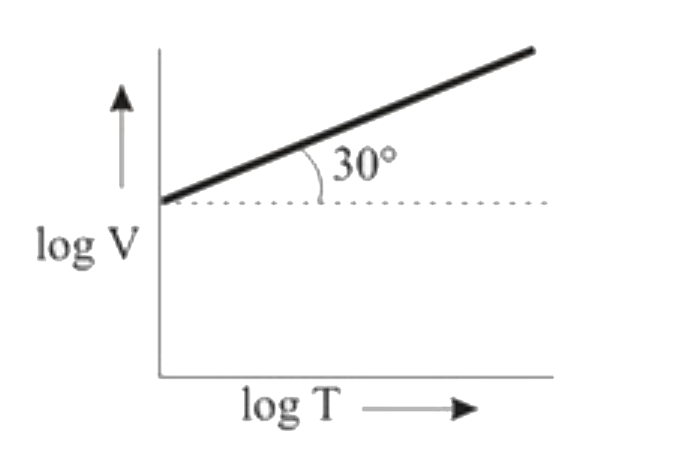
Ten moles of an ideal gas are filled in a closed vessel. The vessel has cylinder and piston type arrangement and pressure of the gas remains constant at 0.821 atm. Which of the following graph represents correct variation of log V vs log T? (V = Volume in litre and T = temperature in Kelvin) |
|
Answer»
|
|