Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Sulphur forms for chlorides S_2 Cl_2 and SCl_2. The equivalent mass of sulphur in SCl_2 is 16 . The equivalent mass of sulphur in S_2 Cl_2 is |
|
Answer» 8 |
|
| 2. |
Sulphur combine with which of the following halogens to form a compound ? |
|
Answer» `Cl_2` |
|
| 3. |
Sulphur disappears when it is boiled in a solution of sodium sulphite. This is due to formation of |
|
Answer» `SO_2` |
|
| 4. |
Sulphur disappears when boiled with an aqueous solution of sodium sulphite. Why ? |
|
Answer» SOLUTION :S reacts with `Na_(2)SO_(3)` to form sodium THIOSULPHATE `(Na_(2)S_(2)O_(3))` and hence S DISAPPEARS. `Na_(2)SO_(3) + S rarr Na_(2)S_(2)O_(3)` |
|
| 5. |
Sulphur dioxide present in the industrial chimney exhaust causes : |
|
Answer» RESPIRATORY and LUNG disease |
|
| 6. |
Sulphur dioxide may be recognised by its: |
|
Answer» CHARACTERISTIC PUNGENT smell of BURNING sulphur |
|
| 7. |
Sulphur dioxide causes I. respiratory diseases in human beings. II. Red haze in the traffic III. Irritation of the eyes. The correct option is |
|
Answer» Both I and II |
|
| 8. |
Sulphur di oxide, how many times heavier than air? |
|
Answer» 2 TIMES |
|
| 9. |
Sulphur dioxide and sulphite an antioxidants. |
|
Answer» Solution :Sulphur dioxide is WIDELY used as preservative and antioxidant in food and beverage industries. (2) Sulphites are used as antioxidant, and they reduce discolourationof FRUITS, vegetables and dehydrated potatoes. They are also used preservative in wine, dairy PRODUCTS, sauce, jams, jellies, etc. (3) They are used as food additive as antimicrobial AGENTS, structure modifiers, enzyme inhibitors. (4) The salts usedare sodium or potassium sulphites or bisulphites. |
|
| 10. |
Sulphur burns in oxygen to form sulphur dioxide . The properties of sulphur dioxide are: |
|
Answer» totally different from sulphur and OXYGEN |
|
| 11. |
Sulphur atom of which oxoacid have non bonding electron pair ? |
|
Answer» SULPHUROUS ACID |
|
| 12. |
Sulphur-35 (34.96903 amu) emits a beta - particle but no gamma- rays, the product is chlorine -35 (34.96885 amu). The maximum energy emitted by the beta- particle is |
|
Answer» 0.016767 MeV `= 34.96903 - 34.96885 = 0.00018g` Binding ENERGY = mass defect `xx 931` `= 0.00018 xx 931` `= 0.1675 MeV` |
|
| 13. |
Sulphphuryl chloride (SO_(2) CI_(2)) reacts with white phosphorus (P_(4)) to give |
|
Answer» `PCI_(5), SO_(2)` |
|
| 14. |
Sulphur -35 (34.96903 amu) a beta-particle but no gamma-ray. The product is chlorine - 35 (34.96885 a.m.u). The maximum energy emitted by the beta- particle is |
|
Answer» `16.758` MEV Energy RELEASED = `931.5 xx 0.00018 MeV = 0.167 MeV` |
|
| 15. |
Sulphonation of phenol with conc. H_2SO_4 at373 K gives |
|
Answer» p-phenol sulphonic acid |
|
| 16. |
Sulphonation of benzene is done by which reagent? |
|
Answer» CONC. `HNO_3 + Conc. H_2SO_4` |
|
| 17. |
Sulphonation of nitrobenzene gives : |
|
Answer» <P>p-nitrosulphonic aciod |
|
| 18. |
Sulphonation of benzoic acid produces mainly |
|
Answer» o-sulphobenzoic acid 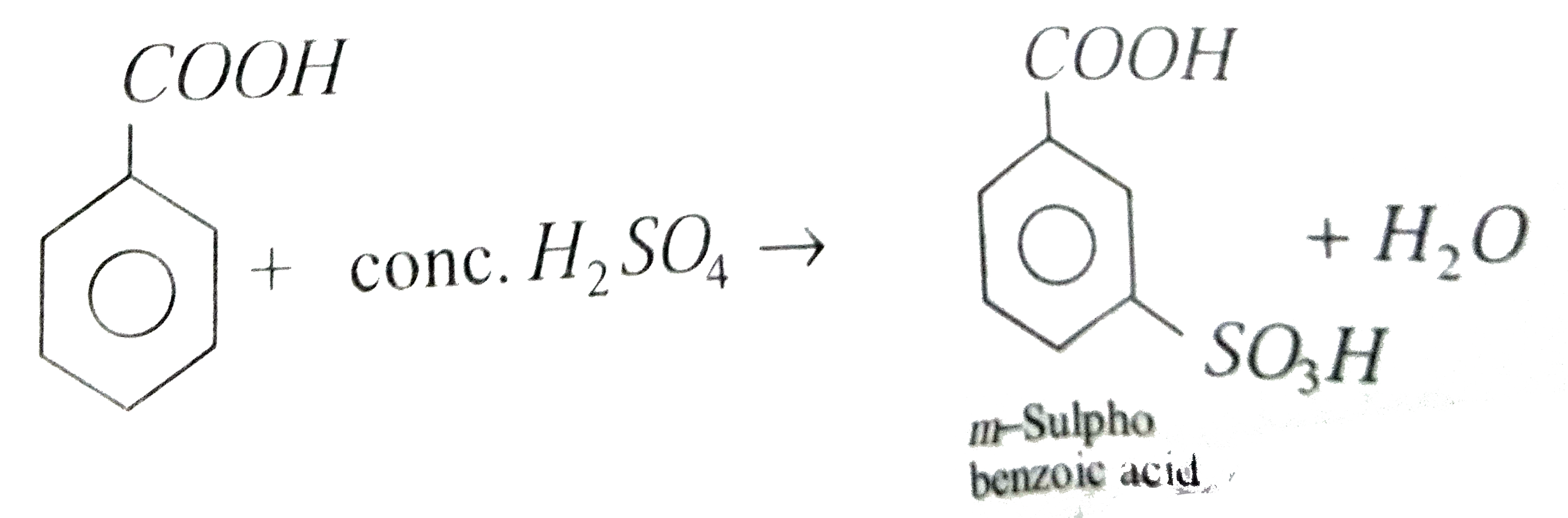
|
|
| 19. |
Sulphonation of benzene is carried out by treating benzene with concentrated sulphuric acid at 330 K in the presence of : |
|
Answer» `SO_(3)` |
|
| 20. |
Sulphonamides act as |
|
Answer» Antiseptic |
|
| 22. |
Sulphides of cations of analytic group IIB are precipitated by H_2S in: |
|
Answer» ACIDIC medium |
|
| 23. |
Sulphides ions in alkaline solution react with solid sulphur to form polyvalent sulphide ions. The equillibrium constant for the formationof S_2^(2-) and S_3^(2-) from S and S^(2-) ions are 1.7 and 5.3 respectively.What is the equillibrium constant for the formation of S_3^(2-) from S_2^(2-) and S ? |
|
Answer» 1.33 |
|
| 24. |
Sulphide ores of the metals are usually concentrated by froth floatation process. Which of the following sulphide ores offers an exception and is concentrated by chemical leaching ? |
|
Answer» Galena |
|
| 25. |
Sulphide ores of metals are usually concentrated by froth flotation process. Which one of the following sulphide ores offer an exception and concentrated by chemical leaching? |
|
Answer» Galena `Ag_(2)S+4NaCN to 2Na[Ag(CN)_(2)]+Na_(2)S` `4 Au+8 KCN+2H_(2)O+O_(2) to 4K[Au (CN)_(2)]+4KOH` |
|
| 26. |
Sulphide ores of metals are usually concentrated by froth floatation process. Which one of the following sulphide ores offers an exception and is concentrated by chemical leaching? |
|
Answer» Galena `underset("Flux")(CaO) + underset("Impurity")(SiO_2) to underset("Slag")(CaSiO_3)` |
|
| 27. |
Sulphideores ofmetalsare usuallyconcentrated by FrothFloatationprocess. Which one ofthefollowingsulphideoresoffers an exceptionandisconcentratedbychemicalleaching ? |
|
Answer» Sphalerite |
|
| 28. |
Sulphide ores are generally concentrated by.... |
|
Answer» Froth floatation |
|
| 29. |
Sulphide ores are common for the metals |
|
Answer» `Ag ,Cu and Pb` |
|
| 30. |
Sulphide ores are concentrated by froth floatation process. Write the name or formulae of any two sulphide ores of copper. |
|
Answer» |
|
| 31. |
Sulphide oresare commonfor metals |
|
Answer» Ag, Cu and Pb |
|
| 32. |
Sulphide ore is converted to oxide form by using the process _________. |
|
Answer» Calcination |
|
| 33. |
The sulphide ores are generally concentrated by : |
|
Answer» ELECTROMAGNETIC PROCESS |
|
| 34. |
Sulphide ore of zinc is concentrated by : |
|
Answer» FLOATATION PROCESS |
|
| 35. |
Sulphide ions react with Na_2[Fe(NO)(CN)_5] to form purple coloured compound Na_4[Fe(CN)_5 (NOS)] in the reaction, the oxidation state of iron changes from |
| Answer» Solution :No change | |
| 36. |
Sulphide ore is : |
|
Answer» COPPER pyrites |
|
| 37. |
Sulphide ions react with Na_(2)[Fe(NO)(CN)_(5)] to form a purple coloured compound Na_(4)[Fe(CN)_(5)(NOS)], in the reaction the oxidation state of ions |
|
Answer» CHANGES from + 2 to + 3 |
|
| 38. |
Sulphide ion reacts with [Fe(CN)_(5)NO] to form a purple coloured compound (X). In this reaction oxidation state of iron . |
|
Answer» CHANGES from +2 to +3 |
|
| 39. |
Sulphide ions react with Na_(2)[Fe(NO)(CN)_(5)] from a purple coloured compound Na_(4)[Fe(CN)_(5)(NOS)]. In the reaction, the oxidation state of iron |
|
Answer» changes from +2 to +3 `Na_(2)[Fe^(2+)(NO)(CN)_(5)]+Na_(2)S to Na_(4)[Fe^(+2)(NOS)(CN)_(5)]` |
|
| 40. |
Sulphide are of zinc//copper is concentrated by |
|
Answer» froth-floatation PROCESS |
|
| 41. |
Sulphate of alkaline earth metal which crystallises without water of hydration is that of |
|
Answer» `Be^(2+)` |
|
| 42. |
Which of the following alkali metal does not form alum: |
|
Answer» Cs |
|
| 43. |
Sulphate ion has____geometry. |
|
Answer» Trigonal |
|
| 44. |
Sulphapyridineis |
|
Answer» ANALGESICS |
|
| 45. |
Sulphate ion has …… geometry |
|
Answer» TRIGONAL |
|
| 46. |
Sulphanilic acid is not soluble in organic solvents. Explain ? |
|
Answer» `(##ALN_CHM_C09(II)_E01_899_Q01##)` Due to its Ionic CHARACTER it is INSOLUBLE in organic SOLVENTS. |
|
| 47. |
Sulphanilic acid although has acidic as well as basic group, it is soluble in alkali but insoluble in mineral acid. Sulphanilic acid is not soluble in organic solvents. |
Answer» SOLUTION :Sulphanilic ACID exist as Zwitterion  The weakly ACIDIC `-^(+)NH_(3)` transfers `H^(+)" to "OH^(-)` to form a soluble salt, `P-NH_(2)-C_(6)H_(4)-SO_(3)-Na^(+)` on the other hand `-SO_(3)^(-)` is too weakly basic to accept `H^(+)` from STRONG acids. Due to its ionic character it is insoluble in organic SOLVENTS. |
|
| 48. |
Sulphan' is |
|
Answer» a MIXTURE of `SO_(3)` and `H_(2)SO_(5)` |
|
| 49. |
Sulphaguanidine is used for : |
|
Answer» DYSENTERY |
|
| 50. |
Sulpha drugs are which type of drugs? |
|
Answer» ANTIMICROBIALS |
|