Saved Bookmarks
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Shyam went to a grocery shop to purchase some food items. The shopkeeper packed all the ftems in polythene bags and gave them to Shyam. But Shyam refused to accpt the polythene bags and asked the shopkeeper to pack the items in paper bags. He informed the shopkeeper about the heavy penalty imposed by the government for using polythene bags. The shopkeeper promised that he would use paper bags in future in place of polythene bags. Answer the following Write the values (at least two) shown by Shyam |
| Answer» Solution :Concerned about ENVIRONMENT, CARING, SOCIALLY alert, law abiding CITIZEN ( or any other 2 values | |
| 3. |
Shown how the following alcohols can be prepared by the action of suitable Grignard reagard reagent on methanal ? (i) CH_(3)-underset(CH_(3))underset(|)(CH)-CH_(2)OH (ii) |
Answer» SOLUTION :
|
|
| 4. |
Show with equations how the following compounds are repared (equations need not be balanced): (i) 4-nitroaniline to 1,2,3-tribromobenzene. (ii) Aniline rarr Benzylamine (in 3 steps) Convert in not more than four steps. |
|
Answer» |
|
| 5. |
Show with the help of a diagram arrangement of stearate ions inside the bulk of water (ionic micelle) at critical micelle concentration of soap. |
Answer» SOLUTION :
|
|
| 6. |
Show the relation between the osmotic pressure of the solutions prepared in 1 liter water by dissolving 10 gram glucose (P_(1)), 10 gram urea (P_(2)) and 10 gram Sucrose, (P_(3)) respectively. |
|
Answer» `P_(2)GT P_(1)gt P_(3)` |
|
| 7. |
Show two different Friedel-Crafts acylation reac- tions that can be used to prepare the following com- pound. |
|
Answer» |
|
| 8. |
Show the mechanism of acylation of ethanamine and write the IUPAC name of the product formed. |
Answer» SOLUTION :MECHANISM of ACETYLATION of ETHANAMINE is GIVEN below : 
|
|
| 9. |
Show the fundamental unit of structure common to all polypeptides and proteins and show how cross linking occurs between two chains by H-bonding. |
Answer» SOLUTION :
|
|
| 10. |
Show the exact structure of the product (or products) of each EAS reaction below.State whether each reaction is expected to be faster or slower than the corresponding reaction of benzene. |
|
Answer»
 Each phenyl group acts as an activating ortho/para director to the other (equivalent)ring. This reaction would be faster than the nitration of benzene. (b)  The carbonyl group acts as a deactivating META director to both(equivalent ) rings. This reaction will be slower than the nitration of benzene. The carbonyl group acts as a deactivating META director to both(equivalent ) rings. This reaction will be slower than the nitration of benzene. (c)  The OXYGEN -containing ring can be viewed as an OR group and an R group attached to the benzene ring. Although both groups are activating , the OR group is more strongly activating , so the position ortho and para to it will be brominated. This reaction will be faster than the bromination of benzene, which requires a CATALYST. (d)  The reaction will be faster than that of benzene because of the activating NATURE of `OCH_(3)` group will be dominating over the deactivating group CN. |
|
| 11. |
Show the formation of a peptide bond with an equation. (OR) What is a peptide bond? Illustrate the formation of a peptide bond in glycin alanine. |
|
Answer» Solution :(i) The bond formed between TWO amino ACIDS by the elimination of a water molecule is called peptide linkage or bond, (ii) The amino GROUP of one amino acid and a carboxyl group of other amino acid undergoes condensation to remove a water molecule and it results in the formation of bond.  (iii) The product formed by linking amino acid molecules through peptide linkages is called a peptide (iv) In peptide formation.the two different amino acid molecules may react in one of the two ways. 
|
|
| 12. |
Show the arrowhead steps for the preparation of acetic acid by using the following substances in the correct order: dry C_(2)H_(5)OC_(2)H_(5),I_(2),Mg, red P, CH_(3)OH,CO_(2), dilute HCl. |
| Answer» Solution :`UNDERSET("Methanol")(CH_(3)OH) overset(I_(2),red" "P)to underset("Methyl iodide")(CH_(3)-I) underset(("Grignard reaction")) overset(Mg,dryC_(2)H_(5)OC_(2)H_(5))to underset("Methylmag. Iodide")(CH_(3)-MgI) underset(C_(2)H_(5)OC_(2)H_(5))overset(CO_(2)(s))to CH_(3)COOMgI overset(Dil.HCl)to underset("ACETIC acid")(CH_(3)COOH)+MgICl` | |
| 13. |
Show that Volume ofa fixedamount of an ideal gas is a statefunction |
|
Answer» |
|
| 14. |
Show that time required for 99% completionis twice the time required for the completion of 90% of reactions for a first order reaction. The decomposition of hydrocarbon follow the equation K=(4.5xx10^(11)5^(-1))e^(-28000K//T)" Calculate Ea". |
|
Answer» Solution :`"For first ORDER reaction, t"=(2.303)/(R )LOG.(a_(0))/(a_(0))` 1)=(2)/(1)` `rArr""t_(99%)=2xxt_(90%)` `k=Ae^(-Ea//RT)` `(Ea)/(RT)=(28000)/(T )` `E_(a)=28000xxR` `=28000xx8.314` `E_(a)=232.79" KJ mol"^(-1)` |
|
| 15. |
Show that the reaction CO(g) +1/2 O_2(g) to CO_2(g)at 300 K is spontaneous and exothermic, when the standard entropy change is -0.094 kJ "mol"^(-1) K^(-1) . The standard Gibbs free energies of formation for CO_2and CO are -394.4 and -137.2 kJ "mol"^(-1)respectively. |
|
Answer» Solution :`DELTA G^@ = Delta H^@ - T Delta S^@` `Detla G^@ = -257.2 kJ` (SPONTANEOUS) , `Delta H^@ = -285kJ` (exothermic) |
|
| 16. |
Show that the rate of first order reaction is doubled when concentration of the reactant is doubled. |
|
Answer» SOLUTION :Consider FIRST order reaction `A to P` `r_(1) = k[A]^(1) "" ….. (1)` When CONCENTRATION is doubled `r_(2) = k[2A]^(1) "" r_(2) = 2k [A} "" …(2)` Comparing equation (1) and (2) `r_(2) = 2r` |
|
| 17. |
Show that the radius of the particle that can be placed in a tetrahedral void of an hcp or a ccp structure of particles, without disturbing the close-packed structure, should not exceed 0.225 times the radius of the packed particles. |
Answer» Solution :In an hcp or ccp structure, the void that is surrounded by 4 nearest NEIGHBOUR particles occupying the 4 CORNERS of a regular tetrahedron is called a tetrahedral void. 4 particles surrounding a tetrahedral void are in contact with one another. A tetrahedral void is created when 4 particles are placed at alternate corners of a cube as shown in the figure, Suppose, the radius of each packed particle = R & that of tetrahedral void = r.  As the particles are in contact, face diagonal, AE = R + R = 2R If the edge length of the cube be a, then it is clear from the figure that the face diagonal, AE = `sqrt(a^(2)+a^(2))=asqrt2` `therefore" "2R=asqrt2or,a=Rsqrt2` It is also clear from the figure that the body diagonal, `AD=sqrt(AE^(2)+ED^(2))=sqrt((asqrt2)^(2)+a^(2))=asqrt3=Rsqrt2xxsqrt3=Rsqrt6` If a particle with radius 'r' is placed in the tetrahedral void, AF = `1/2xx` body diagonal = `1/2xxAD=1/2xxRsqrt6=Rsqrt(3/2)=R+r` or, `R(sqrt(3/2)-1)=ror,Rxx0.225=r` Therefore, radius of the particle that can be placed in a tetrahedtral void without disturbing the close-packed structure should be equal to 0.225 times the radius of packed particles. |
|
| 18. |
Show that the potentials are additive for the process in which half reaction are added to yield an overall reaction bu they are not additive when added to yield a third half reaction. |
|
Answer» |
|
| 19. |
Show that the following results illustrate the law of reciprocal proportions: (i) 0.92g Mg produces 1.54g mangesium oxide. (ii) 0.41 g Mg illustrates 380mL of hydrogen at NTP from on acid. (iii) 1.25g of water results from the union of 1.11 g of oxygen with hydrogen. (Density of hydrogen at NTP=0.00009g mL^(-1)) |
|
Answer» SOLUTION :(i) Mass of oxygen =(1.54-0.92)=0.62g 0.92g MG combines with 0.62g of oxygen 1G Mg combines with oxygen `=(0.62)/(0.92)=0.6739g` (ii) Mass of 380mL of hydrogen at NTP `=380xx0.00009g` `=0.0342g` 0.41g Mg liberates hydrogen =0.0342g 1g Mg liberates hydrogen `=(0.0342)/(0.41)=0.0834g` The RATIO of hydrogen and oxygen which combine with 1g Mg is `0.0834:0.6739`, i.e., 1:8 (iii) Mass of hydrogen `=(1.25-1.11)=0.14g` The ratio of hydrogen and oxygen in water =0.14:1.11 or 1:8 Sine, the ratio of hydrogen and oxygen is same, the LAW of reciprocal is illustrated. |
|
| 20. |
Show that the differential dV of the molar volume of an ideal gas is an exact differential and hence V is a state function. |
|
Answer» <P> SOLUTION :`[V=(RT)/(p),V=f(p,T)]` |
|
| 21. |
Show that the excluded volume is four times the actual volume of a molecule. |
| Answer» Solution : Excluded VOLUME PER two molecules ` = 4/3 PI (2R)^3` | |
| 22. |
Show that t_(99%) = 2 xx t_(90%) |
|
Answer» Solution :Formula : `K = (2.303)/(t) LOG""([R]_(0))/([R])` `(t_(99%))/(t_(90%)) ==((2.303)/(t)log""(100)/(1))/((2.303)/(k) log""(100)/(10)) rArr (t_(99%))/(t_(90%)) = 2` |
|
| 23. |
Show that SHE can act both as a anode as well as cathode. |
|
Answer» Solution :(i) When it is placed on the right-hand side of the zinc electrode, the hydrogen electrode reaction is, `""2H^(+)+2E^(-) rarr H_(2)` The electrons flow to the SHE and it acts as the cathode. (ii) When the SHE is placed on the LEFT hand side, the electrode reaction is `""H_(2) rarr 2H^(+)+2e^(-)` The electrons flow to the copper electrode and the hydrogen electrode acts as the anode. |
|
| 24. |
Show that pressure of a fixed amount of an ideal gas is a state function V=(nRT)/P |
|
Answer» SOLUTION :`((delV)/(delT))_(p)=(NR)/PrArrdel/(delP)[((delV)/(delT))_(p)]_(T)=(-nR)/(P^(2))` `((delV)/(delT))_(T)=-(nRT)/P^(2)=DEL/(delT)[((delV)/(delP))_(T)]_(P)=(-nR)/P^(2)` |
|
| 25. |
Show that phosphine is weakly basic? |
|
Answer» Solution :PHOSPHINE is WEAKLY basic and forms phosphonium salts with HALOGEN acids. `PH_(3)+HIrarrPH_(4)I` `PH_(4)I+H_(2)Ooverset(DELTA)rarr PH_(3)+H_(3)O^(+)+I^(-)` |
|
| 26. |
Show that, pK_a+pK_b=pK_b where K_b and K_a are the ionisation constants of weak base (B) and its conjugateacid. (BH^+) respectively. |
| Answer» | |
| 27. |
Show that is case of first order reaction , the time required for 99.9% completion is nearly ten times the time required for half completion of the reaction. |
|
Answer» SOLUTION :Let `[A_0]=100, ` When `t=t_(99.9%),[A]=(100-99.9)=0.1` `k=(2.303)/t LOG (([A_0])/([A]))` `t_(99.9%)=(2.303)/k(100)/(0.1)impliest_(99.9%)=(2.303)/klog100 ` `t_(99.9%)=(2.303)/k(3)impliest_(99.9%)=(6.906)/k` `t_(99.9%)~=10xx(0.69)/k` `t_(99.9%)~=10t_(1//2)` |
|
| 28. |
Show that in the first order reaction, time required for completion of 99.9% is 10 times that of half-life (t_(1//2)) of the reaction. |
|
Answer» Solution :The EQUATION for first order REACTION is `t=(2.303)/(k)"log"([R]_(0))/([R])""…(i)` For 99.9% completion, `[R]=0.001xx[R]_(0)` For half-reaction,`[R]=(1)/(2)[R]_(0)` Substituting the valuesin the above equation, we have `t_(99.9%)=(2.303)/(k)"log"([R]_(0))/(0.001xx[R]_(0))=(2.303)/(k) log 10^(3) ""...(ii)` `t_(50%)=(2.303)/(k)"log"([R]_(0))/(1//2xx[R]_(0))=(2.303)/(k)log2 ""...(iii)` DIVIDING (ii) by (iii), we get `(t_(99.9%))/(t_(50%))=(log 10^(3))/(log2)=(3)/(0.3010)=10` |
|
| 29. |
Show that in case of a first order reaction, the time taken for completion of 99.9% reaction is ten times the time required for half change of the reaction. |
|
Answer» SOLUTION :To show that`t_(99.9%) = 10 xx t_(1//2)` `K = (2.303)/(t) LOG ""([R]_(0))/([R])` `(t_(99.9))/(t_(1//2)) = ((2.303)/(k)log""(100)/(0.1))/((2.303)/(k)xxlog""(100)/(50))rArr (t_(99.9%))/(t_(1//2)) = (3)/(0.3)` `t_(99.9%) = 10 t_(1//2)` |
|
| 30. |
Show that in case of a first order reaction, the time required for 99.9% of the reaction to take place is about ten times than that required for half the reaction. |
|
Answer» SOLUTION :For REACTION of first order, `t_(1//2)=(2.303)/(k)log""(a)/(a-(a)/(2))=(2.303)/(k)log""2=(2.303)/(k)(0.3010)` `t_(99.9%=(2.303)/(k)log=(a)/(a-0.999a)=(2.303)/(k)LOG10^(-3)=(2.303)/(k)xx3:.(t_(99.9%))/(t_(1//2))=(3)/(0.3010)=10` |
|
| 31. |
Show that in a first order reaction,time required for completion of 99.9 % is 10 times of half-life (t_((t)/(2))) of the reaction. |
|
Answer» Solution :When reaction is completed 99.9 % `[R]_(n)[R]_(0)-0.999[R]_(0)` `K=(2.303)/(t)` log `([R]_(0))/([R])` `k=(2.303)/(t)`=log`([R]_(0))/([R]_(0)-0.999[R]_(0))` `=(2.303)/(t)=log 10^(3)` t=6.909 /K For HALF -life of the reaction `t((t)/(2))=(0.693)/(k)` `(t)/(t(1)/(2))=(6.909)/(k)XX(6.909)/(k)=10` |
|
| 32. |
Show that in a cubic close packed structure, eight tetrahedral voids are present per unit cell. |
|
Answer» Solution : cubicpackedstruturecontainsoneatomat eachcornerof anunitcellandatforcecentre of echunit cellEachunit cellconsistsof 8smallcubes. Each smallleadto CREATIONOF atetrahedralvoidas SHOWNBELOW  sincethere are total8 SMALLERCUBES PRESENTAT oneunitcelleachcubeshasonetetrahedralvoidtotalnumberof tetrahedralvoid PRESENTIN eachunitcellis equaltoright As weknown ccp struturehasatomsper unitcellthustotalnumberof tetrahedralvoidin oneccpunitcellequalto 8. |
|
| 33. |
Show that for first order reaction t(87.5%) = 3t_(50%) |
|
Answer» Solution :`k =2.303/t_(87.5%)"LOG" (100)/(12.5)``k=2.303/t_(87.5%)xx0.9031` EXPRESSION of K when 50% of REACTION is reacted `k=2.303/t_(50%) "log" (100)/(50)``k= 2.303/t_(50%)xx0.3010`k= EQUATION `(1)//`Equation(2) `t_(87.5%)=t_(50%)` |
|
| 34. |
Show that chromates and dichromates are interconvertible. |
|
Answer» Solution :The chromate and dichromates are interconvertible in aqueous solution DEPENDING upon pH of the solution. At low pH (acidic MEDIUM) : Chromate CONVERTS into dichromate `2CrO_(4)^(2-)+2H^(+)rarrCr_(2)O_(7)^(2-)+H_(2)O` At high pH (alkaline medium) : dichromate is converted into chromate `Cr_(2)O_(7)^(2-)+2OH^(-)rarr2CrO_(4)^(2-)+H_(2)O` |
|
| 35. |
Show how you might synthesize 2-propanamine from a three-carbon starting material that is a ketone, aldehyde, nitrile, or amide. |
| Answer» Solution :We begin by recognizing that 2-propanamine has a primary AMINE group bonded to a secondary carbon. Neither a three-carbon nitrile nor a three-carbon amide can LEAD to this structural unit from a `C_(3)` starting material. An oxime can lead to the proper structure, but we must start with a three-carbon ketone rather than an aldehyde. THEREFORE, we choose PROPANONE as our starting material, convert it to an oxime, and then reduce the oxime to an amine. | |
| 36. |
Show how will you synthesise : (i) 1- phenylethanol from a suitable alkene, (ii) cyclohexylmethanol using an alkyl halide by an S_(N)2 reaction. (iii) pentan-1-ol using a suitable alkyl halide? |
Answer» Solution :(i) ADDITION of `H_(2)O` to Ethenylbenzene in presence of dil. `H_(2)SO_(4)` gives 1- Phenylethanol.  (ii) Hydrolysis of cyclohexylmethyl bromide by aqueous `NaOH` gives cyclohexylmethanol.  (iii) Hydrolysis of 1- Bromopentane by aqueous `NaOH` gives Pentan -1- ol. `underset("1 - Bromopentane")(CH_(3)-CH_(2)-CH_(2)-CH_(2)-CH_(2)-Br)+NaOH(aq)overset(Delta)underset("Hydrolysis")RARR underset("Pentan-1-ol")(CH_(3)-CH_(2)-CH_(2)-CH_(2)-CH_(2)-OH+NaBr)` |
|
| 37. |
Showhowyoucould synthesize allyl phenyl etherphenoland allyl bromide |
Answer» SOLUTION :USEA williamsonethersynthesis
|
|
| 38. |
Show how will you synthesize : Pentan-1-ol using suitable alkyl halide ? |
|
Answer» SOLUTION :`? to (CH_(2))_(3)- (CH_(2))_(3)- CH_(2)OH` Alkyl haide `UNDERSET("1-Chloropentane")(CH_(3)-(CH_(2))_(3)- CH_(2)-Cl+ NaOH) underset(Delta)OVERSET(S_(N)@" hydrolysis")to underset("Pentan-1-ol")(CH_(3)- (CH_(2))_(3)- CH_(2)-OH)` |
|
| 39. |
Show how will you synthesize (i) 1-phenylethanol from a suitable alkene. (ii) cyclohexylmethanol using an alkyl halide by an S_(N)2 reaction. (iii) pentan-1-ol using a suitable alkyl halide. |
Answer» Solution :(i) Addition of `H_(2)O` to ethenylbenzene (or styrene) in presence of dil. `H_(2)SO_(4)` gives 1-phenylethanol. 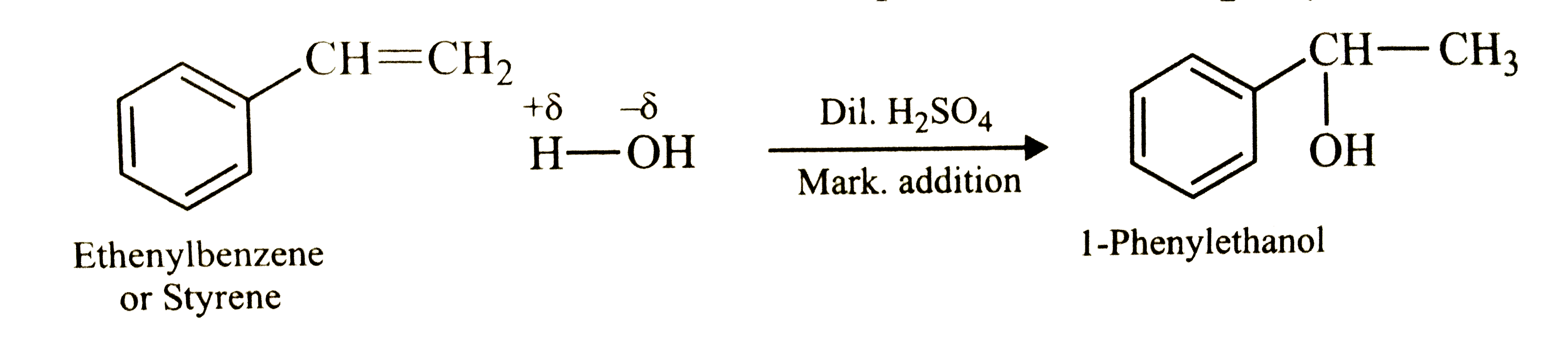 (II) Hydrolysis of cyclohexylmethyl bromide by aqueous NAOH gives cyclohexylmethanol 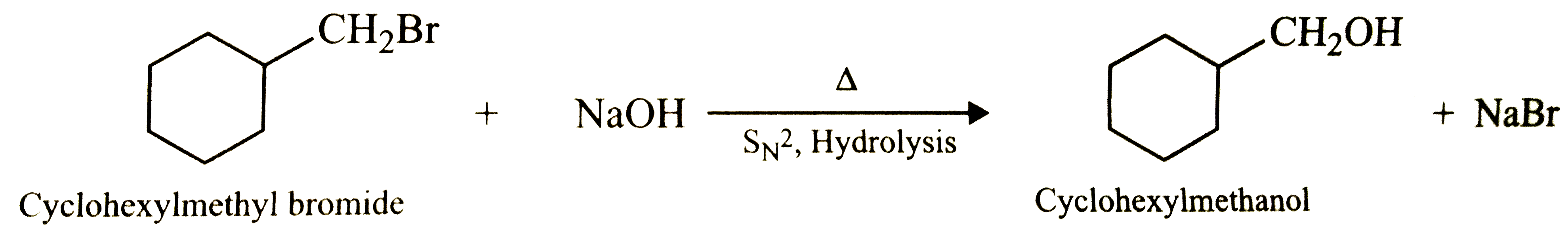 (iii) Hydrolysis of 1-bromopentane by aqueous `NaOH` gives pentan-1-ol. `underset("1-Bromopentane")(CH_(3)CH_(2)CH_(2)CH_(2)CH_(2))-Br+NaOH(aq) underset(S_(N)2,"Hydrolysis")OVERSET(Delta)to underset("Pentan-1-ol")(CH_(3)CH_(2)CH_(2)CH_(2)CH_(2))-OH+NaBr` |
|
| 40. |
Show how will you synthesize : Cyclohexylmethanol using alkyl halide by an S_(N)2 reaction. |
Answer» SOLUTION :
|
|
| 41. |
Show how will you synthesize : 1-Phenylethanol from a suitable alkene. |
Answer» SOLUTION :
|
|
| 42. |
Show how the following alcohols are prepared by the reaction of a suitable Grignard reagent on methanal. |
Answer» SOLUTION :
|
|
| 43. |
Show how the following alcohols are prepared by the reaction of a suitable Grignard reagent on methanal. |
Answer» SOLUTION :
|
|
| 44. |
Show how each of the four products shown at the beginning of this section is formed in the crossed aldol addition between ethanol and propanal. |
|
Answer» Solution :In the BASIC aqueous solution, four organic entities will initially be PRESENT: molecules of ethanal, molecules of PRO panal, enolate ANIONS derived from ethanal, and enolate anions derived from propanal. 3-Hydroxy-2-methylbutanal results when the enolate of propanal REACTS with ethanal.  and finally, 3-hydroxypentanal results when the enolate of ethanal reacts with propanal. 
|
|
| 46. |
Show how each of the following compounds could be converted to benzoic acid (i) Ethylbenzene (ii) Acetophenone (iii) Bromobenzene (iv) Phenyethene (Styrene). |
Answer» SOLUTION :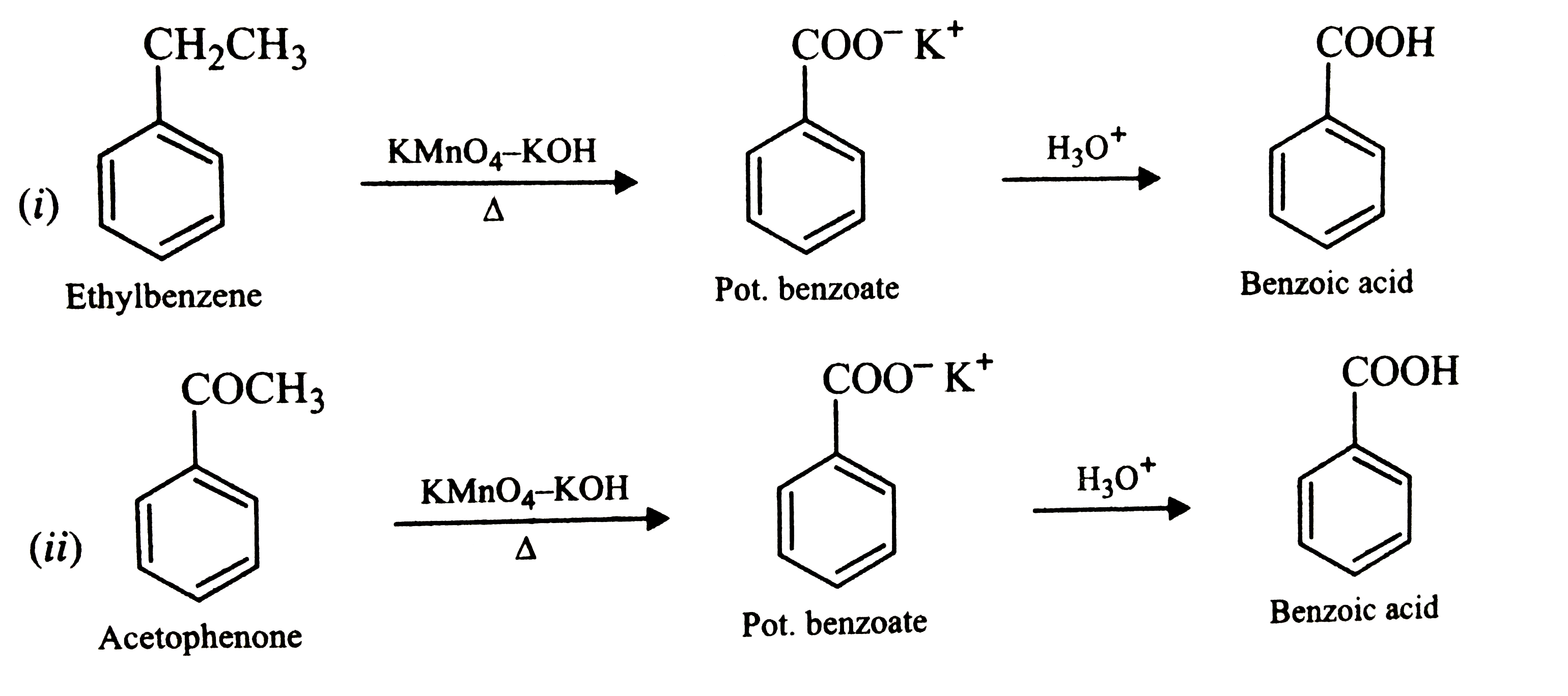 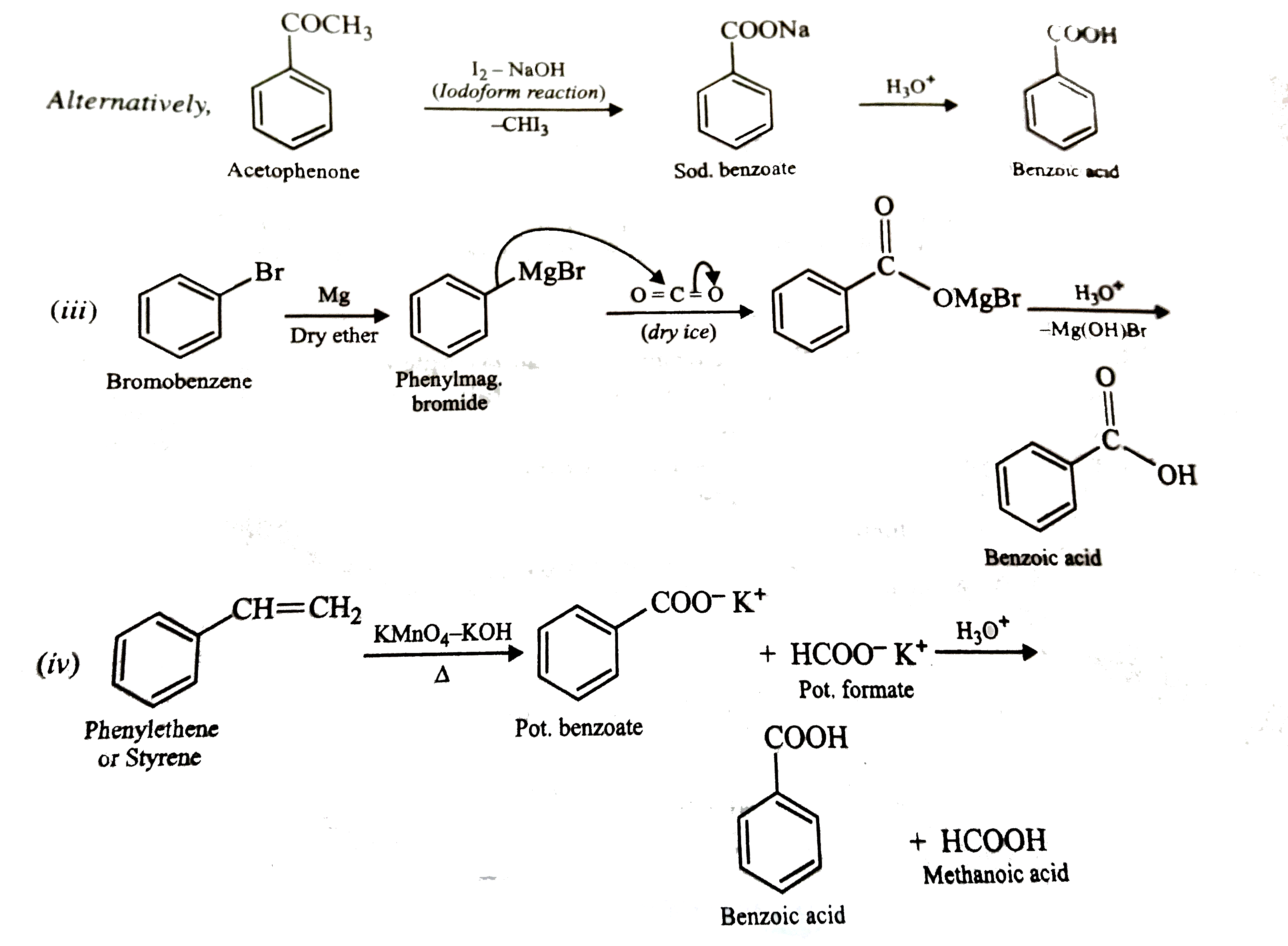 . .
|
|
| 47. |
Show how each of the following compounds can be converted to benzoic acid. (iv) Phenylethene (styrene) |
|
Answer» |
|
| 48. |
Show how each of the following compounds can be converted to benzoic acid. (iii) Bromobenzene |
|
Answer» |
|
| 49. |
Show how each of the following compounds can be converted to benzoic acid. (ii) Acetophenone |
|
Answer» |
|
| 50. |
Show how are the following alcohols prepared by the reaction of a suitable Grignard reagent on methanal? (i) CH_(3)-underset(CH_(3))underset("|")"CH"-CH_(2)OH (ii) |
|
Answer» Solution :(i) `CH_(3)-underset(CH_(3))underset("|")"C "-CH_(2)OH` METHANAL REACTS with Grignard reagent to FORM primary alcohol. To identify the Grignard reagent, look for the part other than `-CH_(2)OH`. THEREFORE, the Grignard reagent should be isopropyl MAGNESIUM bromide. Thus,   Identify the Grignard reagent, look for the part of the product other than `-CH_(2)OH`. Therefore the Grignard reagent must be cyclohexylmagnesium bromide. Reaction takes place as under : 
|
|




