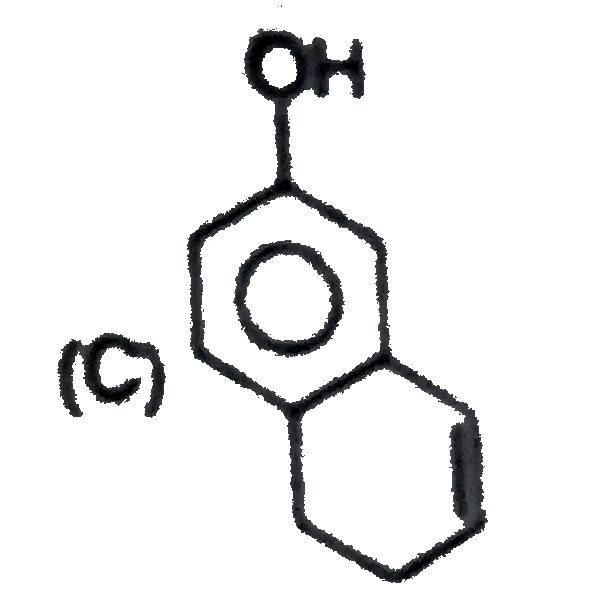
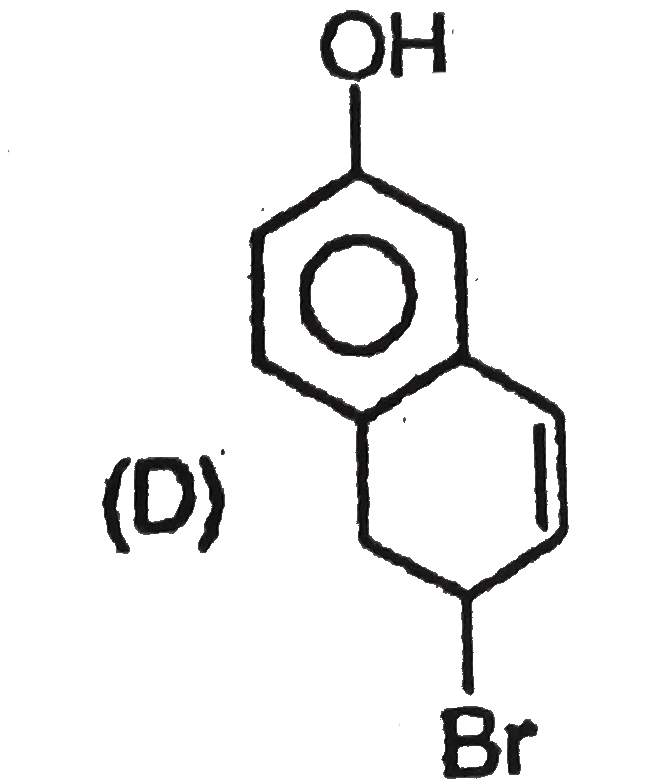
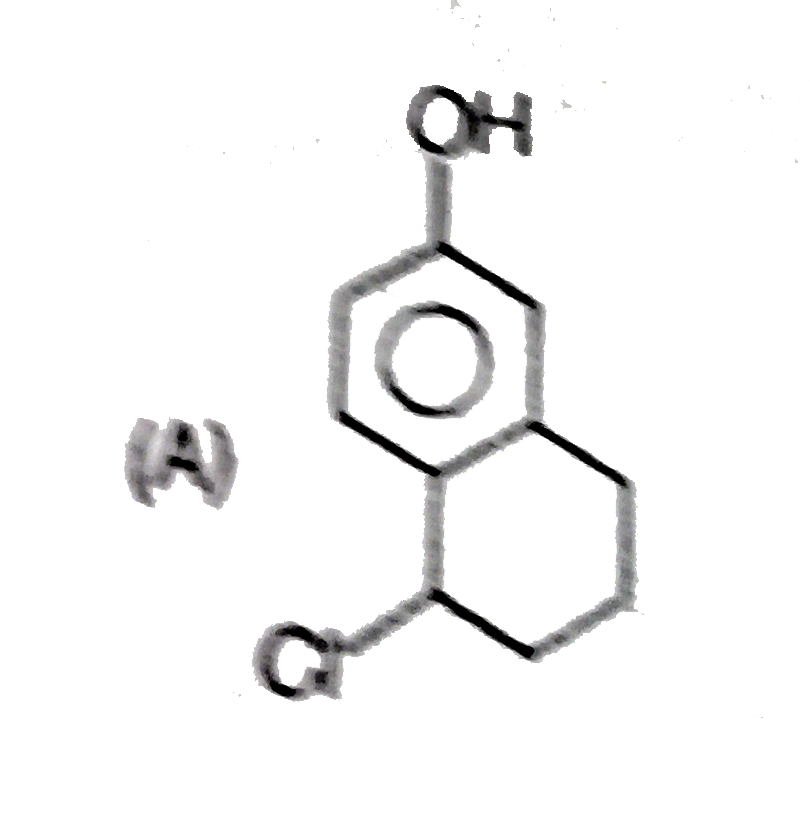
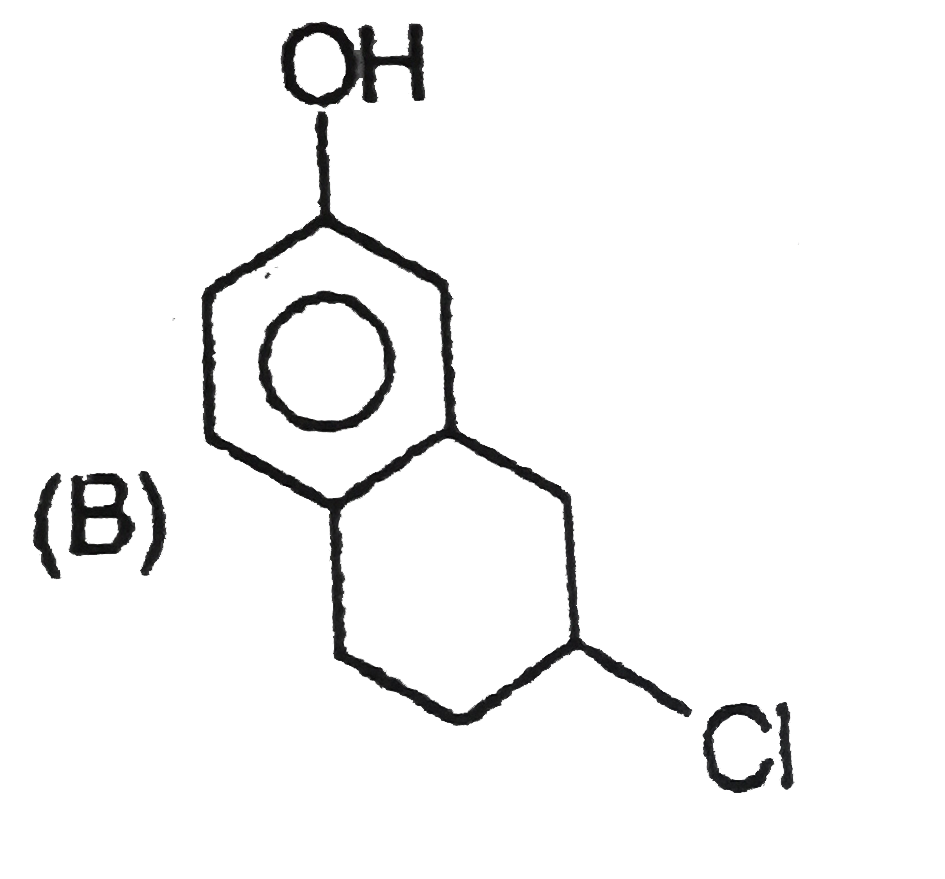
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Read the given passage and answer questions number 1 to 5 that follow : A variety of alkenes or dienes and their derivatives are polymerised in the presence of a free radical generating initiator (catalyst) like benzoyl peroxide, acetyl peroxide, tert-butyl peroxide, etc. For example, the polymerisation of ethene to polythene consists of heating or exposing to light a mixture of ethene with a small amount of benzoyl peroxide initiator. The process starts with the addition of phenyl free radical formed by the peroxide to the ethene double bond thus generating a new and larger free radical. This step is called chain initiating step. As this radical reacts with another molecule of ethene, another bigger sized radical is formed. The repetition of this sequence with new and bigger radicals carries the reaction forward and the step is termed as chain propagating step. Ultimately, at some stage the product radical thus formed reacts with another radical to form the polymerised product. This step is called the chain terminating step. How does the chain terminate to form the polymerised product? |
| Answer» Solution : At some STAGE, the PRODUCT RADICAL REACTS with ANOTHER radical to form the polymerised product. | |
| 2. |
Read the given passage and answer questions number 1 to 5 that follow : A variety of alkenes or dienes and their derivatives are polymerised in the presence of a free radical generating initiator (catalyst) like benzoyl peroxide, acetyl peroxide, tert-butyl peroxide, etc. For example, the polymerisation of ethene to polythene consists of heating or exposing to light a mixture of ethene with a small amount of benzoyl peroxide initiator. The process starts with the addition of phenyl free radical formed by the peroxide to the ethene double bond thus generating a new and larger free radical. This step is called chain initiating step. As this radical reacts with another molecule of ethene, another bigger sized radical is formed. The repetition of this sequence with new and bigger radicals carries the reaction forward and the step is termed as chain propagating step. Ultimately, at some stage the product radical thus formed reacts with another radical to form the polymerised product. This step is called the chain terminating step. How does the free radical combine with the monomer? Show the reaction with ethene as monomer and benzoyl peroxide as the initiator. |
Answer» SOLUTION :
|
|
| 3. |
Read the given passage and answer questions number 1 to 5 that follow : A variety of alkenes or dienes and their derivatives are polymerised in the presence of a free radical generating initiator (catalyst) like benzoyl peroxide, acetyl peroxide, tert-butyl peroxide, etc. For example, the polymerisation of ethene to polythene consists of heating or exposing to light a mixture of ethene with a small amount of benzoyl peroxide initiator. The process starts with the addition of phenyl free radical formed by the peroxide to the ethene double bond thus generating a new and larger free radical. This step is called chain initiating step. As this radical reacts with another molecule of ethene, another bigger sized radical is formed. The repetition of this sequence with new and bigger radicals carries the reaction forward and the step is termed as chain propagating step. Ultimately, at some stage the product radical thus formed reacts with another radical to form the polymerised product. This step is called the chain terminating step. Show the formation of a free radical from benzoyl peroxide. |
Answer» SOLUTION :
|
|
| 4. |
Read the given passage and answer questions number 1 to 5 that follow : A variety of alkenes or dienes and their derivatives are polymerised in the presence of a free radical generating initiator (catalyst) like benzoyl peroxide, acetyl peroxide, tert-butyl peroxide, etc. For example, the polymerisation of ethene to polythene consists of heating or exposing to light a mixture of ethene with a small amount of benzoyl peroxide initiator. The process starts with the addition of phenyl free radical formed by the peroxide to the ethene double bond thus generating a new and larger free radical. This step is called chain initiating step. As this radical reacts with another molecule of ethene, another bigger sized radical is formed. The repetition of this sequence with new and bigger radicals carries the reaction forward and the step is termed as chain propagating step. Ultimately, at some stage the product radical thus formed reacts with another radical to form the polymerised product. This step is called the chain terminating step. Name different initiators that can be used to perform polymerisation. |
| Answer» SOLUTION : BENZOYL PEROXIDE, ACETYL peroxide, tert-butyl peroxide. | |
| 5. |
Read the given passage and answer questionsfollow : The replacement of hydrogen atom(s) in an aliphatic or aromatic hydrocarbon by halogen atom(s) results in the formation of alkyl halide (haloalkane) and aryl halide (haloarene),respectively. Haloalkanes contain halogen atom(s) attached to the sp^(3)hybridised carbon atom of an alkyl group whereas haloarenes contain halogen atom(s) attached to sp^(2) hybridised carbon atom(s) of an aryl group. Many halogen containing organic compounds occur in nature and some of these are clinically useful. These classes of compounds find wide applications in industry as well as in day-to-day life. They are used as solvents for relatively non-polar compounds and as starting materials for the synthesis of wide range of organic compounds. Chlorine containing antibiotic, chloramphenicol, produced by microorganisms is very effective for the treatment of typhoid fever. Our body produces iodine containing hormone, thyroxine, the deficiency of which causes a disease called goiter. Synthetic halogen compounds, viz. chloroquine is used for the treatment of malaria, halothane is used as an anaesthetic during surgery. What is the use of halothane ? |
| Answer» SOLUTION : It is an ANAESTHETIC USED during SURGERY. | |
| 6. |
Read the given passage and answer questions follow : The replacement of hydrogen atom(s) in an aliphatic or aromatic hydrocarbon by halogen atom(s) results in the formation of alkyl halide (haloalkane) and aryl halide (haloarene),respectively. Haloalkanes contain halogen atom(s) attached to the sp^(3)hybridised carbon atom of an alkyl group whereas haloarenes contain halogen atom(s) attached to sp^(2) hybridised carbon atom(s) of an aryl group. Many halogen containing organic compounds occur in nature and some of these are clinically useful. These classes of compounds find wide applications in industry as well as in day-to-day life. They are used as solvents for relatively non-polar compounds and as starting materials for the synthesis of wide range of organic compounds. Chlorine containing antibiotic, chloramphenicol, produced by microorganisms is very effective for the treatment of typhoid fever. Our body produces iodine containing hormone, thyroxine, the deficiency of which causes a disease called goiter. Synthetic halogen compounds, viz. chloroquine is used for the treatment of malaria, halothane is used as an anaesthetic during surgery. What is thyroxine ? |
| Answer» Solution :It is an IODINE CONTAINING hormone PRODUCED by our body deficiency of which CAUSES goiter. | |
| 7. |
Read the given passage and answer questions number 1 to 5 that follow : A variety of alkenes or dienes and their derivatives are polymerised in the presence of a free radical generating initiator (catalyst) like benzoyl peroxide, acetyl peroxide, tert-butyl peroxide, etc. For example, the polymerisation of ethene to polythene consists of heating or exposing to light a mixture of ethene with a small amount of benzoyl peroxide initiator. The process starts with the addition of phenyl free radical formed by the peroxide to the ethene double bond thus generating a new and larger free radical. This step is called chain initiating step. As this radical reacts with another molecule of ethene, another bigger sized radical is formed. The repetition of this sequence with new and bigger radicals carries the reaction forward and the step is termed as chain propagating step. Ultimately, at some stage the product radical thus formed reacts with another radical to form the polymerised product. This step is called the chain terminating step. List the different steps involved in polymerisation reaction. |
|
Answer» Solution :(a) CHAIN INITIATING step. (B) Chain propagating step. (c) Chain terminating step. |
|
| 8. |
Read the given passage and answer questions follow : The replacement of hydrogen atom(s) in an aliphatic or aromatic hydrocarbon by halogen atom(s) results in the formation of alkyl halide (haloalkane) and aryl halide (haloarene),respectively. Haloalkanes contain halogen atom(s) attached to the sp^(3)hybridised carbon atom of an alkyl group whereas haloarenes contain halogen atom(s) attached to sp^(2) hybridised carbon atom(s) of an aryl group. Many halogen containing organic compounds occur in nature and some of these are clinically useful. These classes of compounds find wide applications in industry as well as in day-to-day life. They are used as solvents for relatively non-polar compounds and as starting materials for the synthesis of wide range of organic compounds. Chlorine containing antibiotic, chloramphenicol, produced by microorganisms is very effective for the treatment of typhoid fever. Our body produces iodine containing hormone, thyroxine, the deficiency of which causes a disease called goiter. Synthetic halogen compounds, viz. chloroquine is used for the treatment of malaria, halothane is used as an anaesthetic during surgery. Can we use halogen containing organic solvents to dissolve potassium chloride ? |
| Answer» Solution : No, ORGANIC SOLVENTS do not dissolve polar COMPOUNDS. | |
| 9. |
Read the given passage and answer questions follow : The replacement of hydrogen atom(s) in an aliphatic or aromatic hydrocarbon by halogen atom(s) results in the formation of alkyl halide (haloalkane) and aryl halide (haloarene),respectively. Haloalkanes contain halogen atom(s) attached to the sp^(3)hybridised carbon atom of an alkyl group whereas haloarenes contain halogen atom(s) attached to sp^(2) hybridised carbon atom(s) of an aryl group. Many halogen containing organic compounds occur in nature and some of these are clinically useful. These classes of compounds find wide applications in industry as well as in day-to-day life. They are used as solvents for relatively non-polar compounds and as starting materials for the synthesis of wide range of organic compounds. Chlorine containing antibiotic, chloramphenicol, produced by microorganisms is very effective for the treatment of typhoid fever. Our body produces iodine containing hormone, thyroxine, the deficiency of which causes a disease called goiter. Synthetic halogen compounds, viz. chloroquine is used for the treatment of malaria, halothane is used as an anaesthetic during surgery. Out of C_(2)H_(5)Cl " and " C_(6)H_(5)Clhalogen compounds, which has the halogen attached to sp^(2)hybridised carbon atom ? |
| Answer» Solution :`C_(6)H_(5)Cl` has chlrine attached to `SP^(2)` hybridised carbon . | |
| 10. |
Read the given passage and answer questions follow : The replacement of hydrogen atom(s) in an aliphatic or aromatic hydrocarbon by halogen atom(s) results in the formation of alkyl halide (haloalkane) and aryl halide (haloarene),respectively. Haloalkanes contain halogen atom(s) attached to the sp^(3)hybridised carbon atom of an alkyl group whereas haloarenes contain halogen atom(s) attached to sp^(2) hybridised carbon atom(s) of an aryl group. Many halogen containing organic compounds occur in nature and some of these are clinically useful. These classes of compounds find wide applications in industry as well as in day-to-day life. They are used as solvents for relatively non-polar compounds and as starting materials for the synthesis of wide range of organic compounds. Chlorine containing antibiotic, chloramphenicol, produced by microorganisms is very effective for the treatment of typhoid fever. Our body produces iodine containing hormone, thyroxine, the deficiency of which causes a disease called goiter. Synthetic halogen compounds, viz. chloroquine is used for the treatment of malaria, halothane is used as an anaesthetic during surgery. Name a chlorine containing compound produced by microorganisms for the treatment of typhoid fever. |
| Answer» SOLUTION :CHLORAMPHENICOL. | |
| 11. |
Read the given passage and answer questions follow: The hydroxyl group of an alcohol is replaced by halogen on reaction with concentrated halogen acids, phosphorus halides or thionyl chloride. Thionyl chloride is preferred because in this reaction alkyl halide is formed along with gases SO_(2) " and " HCl . The two gaseous products are escapable, hence, the reaction gives pure alkyl halides. The reactions of primary and secondary alcohols with HCl require the presence of a catalyst, ZnCl_(2). With tertiary alcohols, the reaction is conducted by simply shaking the alcohol with concentrated HCl at room temperature. Constant boiling with HBr (48%) is used for preparing alkyl bromide. Good yields of R- I may be obtained by heating alcohols with sodium or potassium iodide in 95% orthophosphoric acid. The order of reactivity of alcohols with a given haloacid is 3^(@) gt 2^(@) gt 1^(@). Phosphorus tribromide and triiodide are usually generated in situ (produced in the reaction mixture) by the reaction of red phosphorus with bromine and iodine respectively. How are PBr_(3) " and " PI_(3)prepared for halogenating the hydroxyl group of alcohols ? |
| Answer» Solution :These are obtained in situ by the REACTION between RED PHOSPHORUS and the halogen. | |
| 12. |
Read the given passage and answer questions follow: The hydroxyl group of an alcohol is replaced by halogen on reaction with concentrated halogen acids, phosphorus halides or thionyl chloride. Thionyl chloride is preferred because in this reaction alkyl halide is formed along with gases SO_(2) " and " HCl . The two gaseous products are escapable, hence, the reaction gives pure alkyl halides. The reactions of primary and secondary alcohols with HCl require the presence of a catalyst, ZnCl_(2). With tertiary alcohols, the reaction is conducted by simply shaking the alcohol with concentrated HCl at room temperature. Constant boiling with HBr (48%) is used for preparing alkyl bromide. Good yields of R- I may be obtained by heating alcohols with sodium or potassium iodide in 95% orthophosphoric acid. The order of reactivity of alcohols with a given haloacid is 3^(@) gt 2^(@) gt 1^(@). Phosphorus tribromide and triiodide are usually generated in situ (produced in the reaction mixture) by the reaction of red phosphorus with bromine and iodine respectively. Give the reactivity order of alcohols in halogenation using a haloacid. |
| Answer» SOLUTION :`3^(@) GT 2^(@) gt 1^(@)` . | |
| 13. |
Read the given passage and answer questions follow: The hydroxyl group of an alcohol is replaced by halogen on reaction with concentrated halogen acids, phosphorus halides or thionyl chloride. Thionyl chloride is preferred because in this reaction alkyl halide is formed along with gases SO_(2) " and " HCl . The two gaseous products are escapable, hence, the reaction gives pure alkyl halides. The reactions of primary and secondary alcohols with HCl require the presence of a catalyst, ZnCl_(2). With tertiary alcohols, the reaction is conducted by simply shaking the alcohol with concentrated HCl at room temperature. Constant boiling with HBr (48%) is used for preparing alkyl bromide. Good yields of R- I may be obtained by heating alcohols with sodium or potassium iodide in 95% orthophosphoric acid. The order of reactivity of alcohols with a given haloacid is 3^(@) gt 2^(@) gt 1^(@). Phosphorus tribromide and triiodide are usually generated in situ (produced in the reaction mixture) by the reaction of red phosphorus with bromine and iodine respectively. Which catalyst is required when we prepare halogen compounds using concentrated HCI? |
| Answer» SOLUTION :`ZnCl_(2)`. | |
| 14. |
Read the given passage and answer questions follow: The hydroxyl group of an alcohol is replaced by halogen on reaction with concentrated halogen acids, phosphorus halides or thionyl chloride. Thionyl chloride is preferred because in this reaction alkyl halide is formed along with gases SO_(2) " and " HCl . The two gaseous products are escapable, hence, the reaction gives pure alkyl halides. The reactions of primary and secondary alcohols with HCl require the presence of a catalyst, ZnCl_(2). With tertiary alcohols, the reaction is conducted by simply shaking the alcohol with concentrated HCl at room temperature. Constant boiling with HBr (48%) is used for preparing alkyl bromide. Good yields of R- I may be obtained by heating alcohols with sodium or potassium iodide in 95% orthophosphoric acid. The order of reactivity of alcohols with a given haloacid is 3^(@) gt 2^(@) gt 1^(@). Phosphorus tribromide and triiodide are usually generated in situ (produced in the reaction mixture) by the reaction of red phosphorus with bromine and iodine respectively. Name the reagents that can be used for halogenation of alcohols. |
| Answer» SOLUTION :CONCENTRATED halogen acid, phosphorus HALIDES and thionyl CHLORIDE. | |
| 15. |
Read the given passage and answer questions follow: The hydroxyl group of an alcohol is replaced by halogen on reaction with concentrated halogen acids, phosphorus halides or thionyl chloride. Thionyl chloride is preferred because in this reaction alkyl halide is formed along with gases SO_(2) " and " HCl . The two gaseous products are escapable, hence, the reaction gives pure alkyl halides. The reactions of primary and secondary alcohols with HCl require the presence of a catalyst, ZnCl_(2). With tertiary alcohols, the reaction is conducted by simply shaking the alcohol with concentrated HCl at room temperature. Constant boiling with HBr (48%) is used for preparing alkyl bromide. Good yields of R- I may be obtained by heating alcohols with sodium or potassium iodide in 95% orthophosphoric acid. The order of reactivity of alcohols with a given haloacid is 3^(@) gt 2^(@) gt 1^(@). Phosphorus tribromide and triiodide are usually generated in situ (produced in the reaction mixture) by the reaction of red phosphorus with bromine and iodine respectively. Why do we prefer thionyl chloride for the preparation of alkyl halides? |
| Answer» Solution : The side products are GASES and escape. This GIVES the PURE product. | |
| 16. |
Read the given passage and answer questionsfollow : Tetrachloromethane is produced in large quantities for use in the manufacture of refrigerants and propellants for aerosol cans. It is also used as feedstock in the synthesis of chlorofluorocarbons and other chemicals, pharmaceutical manufacturing, and general solvent use. Until the mid 1960s, it was also widely used as a cleaning fluid, both in industry, as a degreasing agent, and in the home, as a spot remover and as fire extinguisher. There is some evidence that exposure to carbon tetrachloride causes liver cancer in humans. The most common effects are dizziness, light headedness, nausea and vomiting, which can cause permanent damage to nerve cells. In severe cases, these effects can lead rapidly to stupor, coma, unconsciousness or death. Exposure to C Cl_(4)can make the heart beat irregularly or stop. The chemical may irritate the eyes on contact. When carbon tetrachloride is released into the air, it rises to the atmosphere and depletes the ozone layer. Depletion of the ozone layer is believed to increase human exposure to ultraviolet rays, leading to increased skin cancer, eye diseases and disorders, and possible disruption of the immune system. How does carbon tetrachloride affect the heart? |
| Answer» SOLUTION : It causes the heart beat irregularly or STOP. | |
| 17. |
Read the given passage and answer questionsfollow : Tetrachloromethane is produced in large quantities for use in the manufacture of refrigerants and propellants for aerosol cans. It is also used as feedstock in the synthesis of chlorofluorocarbons and other chemicals, pharmaceutical manufacturing, and general solvent use. Until the mid 1960s, it was also widely used as a cleaning fluid, both in industry, as a degreasing agent, and in the home, as a spot remover and as fire extinguisher. There is some evidence that exposure to carbon tetrachloride causes liver cancer in humans. The most common effects are dizziness, light headedness, nausea and vomiting, which can cause permanent damage to nerve cells. In severe cases, these effects can lead rapidly to stupor, coma, unconsciousness or death. Exposure to C Cl_(4)can make the heart beat irregularly or stop. The chemical may irritate the eyes on contact. When carbon tetrachloride is released into the air, it rises to the atmosphere and depletes the ozone layer. Depletion of the ozone layer is believed to increase human exposure to ultraviolet rays, leading to increased skin cancer, eye diseases and disorders, and possible disruption of the immune system. What is the role of CCl4 to ozone layer ? |
| Answer» SOLUTION : It DEPLETES ozone layer and increases exposure of ULTRAVIOLET RAYS to humans. | |
| 18. |
Read the given passage and answer questionsfollow : Tetrachloromethane is produced in large quantities for use in the manufacture of refrigerants and propellants for aerosol cans. It is also used as feedstock in the synthesis of chlorofluorocarbons and other chemicals, pharmaceutical manufacturing, and general solvent use. Until the mid 1960s, it was also widely used as a cleaning fluid, both in industry, as a degreasing agent, and in the home, as a spot remover and as fire extinguisher. There is some evidence that exposure to carbon tetrachloride causes liver cancer in humans. The most common effects are dizziness, light headedness, nausea and vomiting, which can cause permanent damage to nerve cells. In severe cases, these effects can lead rapidly to stupor, coma, unconsciousness or death. Exposure to C Cl_(4)can make the heart beat irregularly or stop. The chemical may irritate the eyes on contact. When carbon tetrachloride is released into the air, it rises to the atmosphere and depletes the ozone layer. Depletion of the ozone layer is believed to increase human exposure to ultraviolet rays, leading to increased skin cancer, eye diseases and disorders, and possible disruption of the immune system. What are the adverse health effects of exposure to carbon tetrachloride ? |
| Answer» SOLUTION : It causes liver CANCER in HUMANS. It causes dizziness, light HEADEDNESS, nausea and vomiting. | |
| 19. |
Read the given passageand answer question number1 to 5 that follow : Hormones are molecules that act as intercellular messengers . Theseare produced by endocrine glands in the body andare poured directly in the bloodstream which transports them to the siteofaction . In terms of chemical nature ,some of thesesteriod ,e.g., estrogens and androgens, someotherareamino acid derivatives such as epinephrineand norepinephrine. Hormones haveseveral function in the body. The help to maintain theh balanceof biologicalactivities in the body .Therole of insulin in keeping the bloodglucose level within the narrowlimits is an exampleof this function . Insulin is relased in response to the rapidrisein bloodglucoselevel . Onthe otherhand hormore glucagon tends to increase the glucose level in theblood . Thetwo hormones togetherregulate teh glucose level in the blood. Epinephrine andnorepinephrine mediate respones toexternal stimuli.Growth hormones and sex hormones play role in the grwoth and development. Thyroxineproduced in the thyroidgland is an iodinatedderivative of amino acidtyrosine. Whatisthyroxine ? |
| Answer» SOLUTION :Thyroxineis an iodinated DERIVATIVE ofamion ACID TYROSINE. | |
| 20. |
Read the given passage and answer questionsfollow : Tetrachloromethane is produced in large quantities for use in the manufacture of refrigerants and propellants for aerosol cans. It is also used as feedstock in the synthesis of chlorofluorocarbons and other chemicals, pharmaceutical manufacturing, and general solvent use. Until the mid 1960s, it was also widely used as a cleaning fluid, both in industry, as a degreasing agent, and in the home, as a spot remover and as fire extinguisher. There is some evidence that exposure to carbon tetrachloride causes liver cancer in humans. The most common effects are dizziness, light headedness, nausea and vomiting, which can cause permanent damage to nerve cells. In severe cases, these effects can lead rapidly to stupor, coma, unconsciousness or death. Exposure to C Cl_(4)can make the heart beat irregularly or stop. The chemical may irritate the eyes on contact. When carbon tetrachloride is released into the air, it rises to the atmosphere and depletes the ozone layer. Depletion of the ozone layer is believed to increase human exposure to ultraviolet rays, leading to increased skin cancer, eye diseases and disorders, and possible disruption of the immune system. Give two uses of tetrachloromethane. |
|
Answer» Solution :(a) MANUFACTURE of refrigerants and propellants. (B) SYNTHESIS of CHLOROFLUROCARBONS and other chemicals. |
|
| 21. |
Read the given passage and answer questionsfollow : Tetrachloromethane is produced in large quantities for use in the manufacture of refrigerants and propellants for aerosol cans. It is also used as feedstock in the synthesis of chlorofluorocarbons and other chemicals, pharmaceutical manufacturing, and general solvent use. Until the mid 1960s, it was also widely used as a cleaning fluid, both in industry, as a degreasing agent, and in the home, as a spot remover and as fire extinguisher. There is some evidence that exposure to carbon tetrachloride causes liver cancer in humans. The most common effects are dizziness, light headedness, nausea and vomiting, which can cause permanent damage to nerve cells. In severe cases, these effects can lead rapidly to stupor, coma, unconsciousness or death. Exposure to C Cl_(4)can make the heart beat irregularly or stop. The chemical may irritate the eyes on contact. When carbon tetrachloride is released into the air, it rises to the atmosphere and depletes the ozone layer. Depletion of the ozone layer is believed to increase human exposure to ultraviolet rays, leading to increased skin cancer, eye diseases and disorders, and possible disruption of the immune system. Why was the use of tetrachloromethane as cleaning and degreasing agent stopped aftermid 1960s ? |
| Answer» SOLUTION : It was FOUND to be HARMFUL to the HEALTH. | |
| 22. |
Read the given passageand answer question number1 to 5 that follow : Hormones are molecules that act as intercellular messengers . Theseare produced by endocrine glands in the body andare poured directly in the bloodstream which transports them to the siteofaction . In terms of chemical nature ,some of thesesteriod ,e.g., estrogens and androgens, someotherareamino acid derivatives such as epinephrineand norepinephrine. Hormones haveseveral function in the body. The help to maintain theh balanceof biologicalactivities in the body .Therole of insulin in keeping the bloodglucose level within the narrowlimits is an exampleof this function . Insulin is relased in response to the rapidrisein bloodglucoselevel . Onthe otherhand hormore glucagon tends to increase the glucose level in theblood . Thetwo hormones togetherregulate teh glucose level in the blood. Epinephrine andnorepinephrine mediate respones toexternal stimuli.Growth hormones and sex hormones play role in the grwoth and development. Thyroxineproduced in the thyroidgland is an iodinatedderivative of amino acidtyrosine. Whatis theroleof epinephrine andnorepinephrine ? |
| Answer» SOLUTION :Epinerphrineand norepineplhrinemediaterespones to EXTERNAL STIMULI. | |
| 23. |
Read the given passageand answer question number1 to 5 that follow : Hormones are molecules that act as intercellular messengers . Theseare produced by endocrine glands in the body andare poured directly in the bloodstream which transports them to the siteofaction . In terms of chemical nature ,some of thesesteriod ,e.g., estrogens and androgens, someotherareamino acid derivatives such as epinephrineand norepinephrine. Hormones haveseveral function in the body. The help to maintain theh balanceof biologicalactivities in the body .Therole of insulin in keeping the bloodglucose level within the narrowlimits is an exampleof this function . Insulin is relased in response to the rapidrisein bloodglucoselevel . Onthe otherhand hormore glucagon tends to increase the glucose level in theblood . Thetwo hormones togetherregulate teh glucose level in the blood. Epinephrine andnorepinephrine mediate respones toexternal stimuli.Growth hormones and sex hormones play role in the grwoth and development. Thyroxineproduced in the thyroidgland is an iodinatedderivative of amino acidtyrosine.Whatare different functions of hormoes ? |
| Answer» SOLUTION :They HELP to MAINTAIN the balanceof BIOLOGICAL ACTIVITY. | |
| 24. |
Read the given passageand answer question number1 to 5 that follow : Hormones are molecules that act as intercellular messengers . Theseare produced by endocrine glands in the body andare poured directly in the bloodstream which transports them to the siteofaction . In terms of chemical nature ,some of thesesteriod ,e.g., estrogens and androgens, someotherareamino acid derivatives such as epinephrineand norepinephrine. Hormones haveseveral function in the body. The help to maintain theh balanceof biologicalactivities in the body .Therole of insulin in keeping the bloodglucose level within the narrowlimits is an exampleof this function . Insulin is relased in response to the rapidrisein bloodglucoselevel . Onthe otherhand hormore glucagon tends to increase the glucose level in theblood . Thetwo hormones togetherregulate teh glucose level in the blood. Epinephrine andnorepinephrine mediate respones toexternal stimuli.Growth hormones and sex hormones play role in the grwoth and development. Thyroxineproduced in the thyroidgland is an iodinatedderivative of amino acidtyrosine. Whatkindof hormoneis insulin ? |
| Answer» SOLUTION :INSULIN is a POLYPEPTIDE horome. | |
| 25. |
Read the given passageand answer question number1 to 5 that follow : Hormones are molecules that act as intercellular messengers . Theseare produced by endocrine glands in the body andare poured directly in the bloodstream which transports them to the siteofaction . In terms of chemical nature ,some of thesesteriod ,e.g., estrogens and androgens, someotherareamino acid derivatives such as epinephrineand norepinephrine. Hormones haveseveral function in the body. The help to maintain theh balanceof biologicalactivities in the body .Therole of insulin in keeping the bloodglucose level within the narrowlimits is an exampleof this function . Insulin is relased in response to the rapidrisein bloodglucoselevel . Onthe otherhand hormore glucagon tends to increase the glucose level in theblood . Thetwo hormones togetherregulate teh glucose level in the blood. Epinephrine andnorepinephrine mediate respones toexternal stimuli.Growth hormones and sex hormones play role in the grwoth and development. Thyroxineproduced in the thyroidgland is an iodinatedderivative of amino acidtyrosine. Definehormoneis insulin ? |
| Answer» SOLUTION :HORMONES are MOLECULES thatact asintercellular (between the CELLS) MESSENGERS. | |
| 26. |
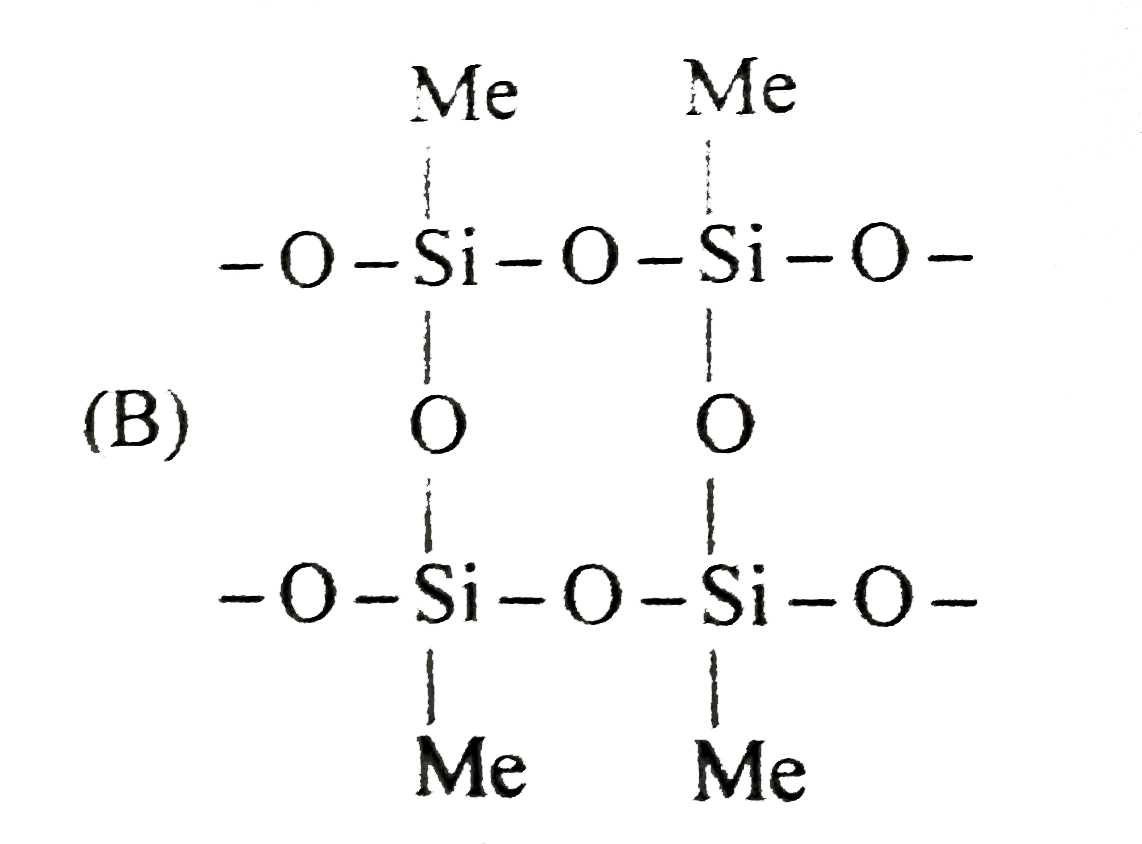
Read the following write-ups and answer the questions at the end of it. Silicons are synthetic polymers containing repeated R_(2)SiO unit. Since, the imperical formula of a ketone (R_(2)CO), the name silicone has been given to this materials. silicons can be made into oils, rubberly elastamors and resins. They find a variety of applications because of their chemical inertness,water repelling nature, heat-resistence and good electrical insulating property. Commercial silicone polymers are usally methyl derivatives and to lesser a extent phenyl derivatives and are synthesised by the hydrolysis of R_(2)SiCl_(2) [R =methyl (Me) or phenyl (phi)] Me_(2)SiCl_(2) overset(H_(2)O)to O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O- If we start with MeSiCl_(3) as the starting material, silicons formed is : |
|
Answer» `Me-overset(Me)overset(|)underset(Me)underset(|)(SI)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-Me` |
|
| 27. |
Read the following write-ups and answer the questions at the end of it. Silicons are synthetic polymers containing repeated R_(2)SiO unit. Since, the imperical formula of a ketone (R_(2)CO), the name silicone has been given to this materials. silicons can be made into oils, rubberly elastamors and resins. They find a variety of applications because of their chemical inertness,water repelling nature, heat-resistence and good electrical insulating property. Commercial silicone polymers are usally methyl derivatives and to lesser a extent phenyl derivatives and are synthesised by the hydrolysis of R_(2)SiCl_(2) [R =methyl (Me) or phenyl (phi)] Me_(2)SiCl_(2) overset(H_(2)O)to O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O- If we mix Me_(3)SiCl with Me_(2)SiCl_(2), we get silicones of the type |
|
Answer» `Me-overset(Me)overset(|)UNDERSET(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-O-overset(Me)overset(|)underset(Me)underset(|)(Si)-Me` |
|
| 28. |
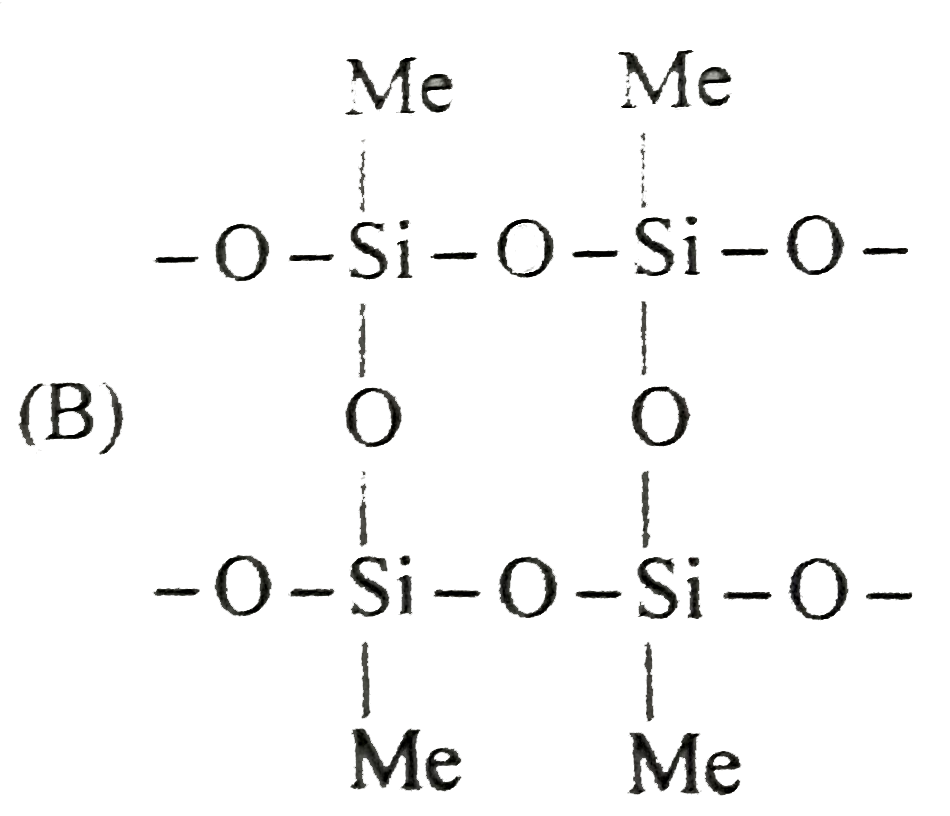
Read the following write-ups and answer the question at the end of it. Silicones are synthetic polymers containing repeated R_(2)SiO units. Since, the empirical formula is that of a ketone (R_(2)CO) , the name silicone has been given to these materials. Silicones can be made intooils, rubbery elastomers and resins. They find a variety of applications because of their chemical inertness, water repelling nature, heat-resistance and good electrical insulating property. Commerical silicon polymers are usually methyl derivatives and to a lesser extent phenyl derivatives and are synthesised by the hydrolysis of R_(2)SiCl_(2)[R="methyl (Me) or phenyl"(phi)] Me_(2)SiCl_(2) overset(H_(2)O) rarrO-underset(Me)underset(|)overset(Me)overset(|)(Si)-O-underset(Me)underset(|)overset(Me)overset(|)(Si)-O-underset(Me)underset(|)overset(Me)overset(|)(Si)-O If we start with MeSiCl_(3) as the starting material, silicones formed is : |
|
Answer»
|
|
| 29. |
Read the following write-ups and answer the question at the end of it. Silicones are synthetic polymers containing repeated R_(2)SiO units. Since, the empirical formula is that of a ketone (R_(2)CO) , the name silicone has been given to these materials. Silicones can be made intooils, rubbery elastomers and resins. They find a variety of applications because of their chemical inertness, water repelling nature, heat-resistance and good electrical insulating property. Commerical silicon polymers are usually methyl derivatives and to a lesser extent phenyl derivatives and are synthesised by the hydrolysis of R_(2)SiCl_(2)[R="methyl (Me) or phenyl"(phi)] Me_(2)SiCl_(2) overset(H_(2)O) rarrO-underset(Me)underset(|)overset(Me)overset(|)(Si)-O-underset(Me)underset(|)overset(Me)overset(|)(Si)-O-underset(Me)underset(|)overset(Me)overset(|)(Si)-O If we mix Me_(3)SiClwith Me_(2)SiCl_(2), we get silicones of the type: |
|
Answer»
|
|
| 30. |
Read the following statements :- (I) Al has greater affinity than that of Cr, for oxygen (II) Al can be used for reduction of Fe_(2)O_(3) (III) Carbon is used for reduction of copper oxide (CuO) (IV) SnO_(2) can be reduced by carbon Choose the correct set of statement(s) |
|
Answer» I, III |
|
| 31. |
Read the following statements: (I) Atomic size of following pair are almost same Fe, Co, Al,Ga , Zr, Hf (II) E.A. order of Li, Be,B,C,N,O is Be lt N lt B lt Li lt C lt O (III) Ionisation energy of Al lt Ga & Zr lt Hf (IV) Ionic radius O^(-2) lt F^(-) lt Na^(+) lt Mg^(+2) Select the correct statements(s) |
|
Answer» I, III, IV |
|
| 32. |
Read the following statements: Choose the correct set of statement(s): (P) Al has greater affionity than that of Fe. For oxygen (Q) Cast iron of nickel impurity of zinc and lead (R ) Refining of nickel is done by vapour phase refning (S) In cyanide proces, oxygen and zinc dust are used as oxidising agent and reducing agent respectively: |
| Answer» Answer :C | |
| 33. |
Read the following statements carefully (A) A secondary alcohol on oxidationgives a ketone (B) Ethanol reacts with conc. H_(2)SO_(4) at 180^(@)C to yield ethylene (C) Methanol reacts with iodine and sodium hydroxide to given a yellow precipitate of iodoform (D) Hydrogen gas is liberated when sodium is added to alcohol. Select the correct statements from the above set |
|
Answer» A,B `(b) CH_(3)-CH_(2)-OH underset(180^(@)C)overset("Conc." H_(2)SO_(4))to CH_(2)=CH_(2)+H_(2)O` (d) `2CH_(3)CH_(2)OH + 2Na to 2CH_(3)-CH_(3)-CH_(2)-ONA +H_(2)` |
|
| 34. |
Read the following statements carefully (A) Albumin is a simple protein (B)The amino acid alanine contains an acidic side chain (C) Insulin is a hormone (C) Muscles contain the protein keratin |
|
Answer» A,B  alanine contain SIDE chain of methyl group. |
|
| 35. |
Read the following statement and choose the correct option. (1) Alnico and aluminium bronze are used in preparation of parts of the aeroplanes. (2) Copper is used in the preparation of tubes of boiler, Deltametal and Muntz metal. (3) Copper and Aluminium forms alloys like duralumin and aluminium bronze. (4) Copper and Zinc are used in the preparation of German Silver. |
|
Answer» T F T T |
|
| 36. |
Read the following statements and choose the correct answer (i) The boiling points of isomeric haloalkanes decrease with increase in branching. (ii) Among isomeric dihalobenzenes the para-isomers have higher melting point than their ortho and metaisomers. (iii) The isomeric dihalobenzene have large difference in their boi I ing and melting points (iv) The isomeric dihalobenzene have nearly same boiling point. |
|
Answer» (i), (ii) and (iii) are correct  Boiling points of isomeric dihalobenzenes are very nearly the same. However, the para-isomers are high melting as compared to their ortho and meta-isomers. It is DUE to symmetry of para-isomers that FITS in CRYSTAL Ia nice better as compared to ortho- and meta-isomers. 
|
|
| 37. |
Read the following statements and choose the correct option? (i) Starch is a polymer of alpha - glucose. (ii) Starch consists of amylose and amylopectin. (iii) Amylose is insoluble in water. (iv) Amylopectin is soluble in water. |
|
Answer» (i) (iii) and(iv) |
|
| 38. |
Read the following statement regarding Na_(6)P_(6)O_(18) compound. (I) It is used as a water softner. (II) It is called as calogone. (III) It's structure has 5, (sigma)/(pi) bond ratio. (IV) All Phosphorous has sp^(3) hybridisation. (V) It's IUPAC name is sodium hexa-meta phosphate How many total number of above statements are correct ? |
|
Answer» Only three 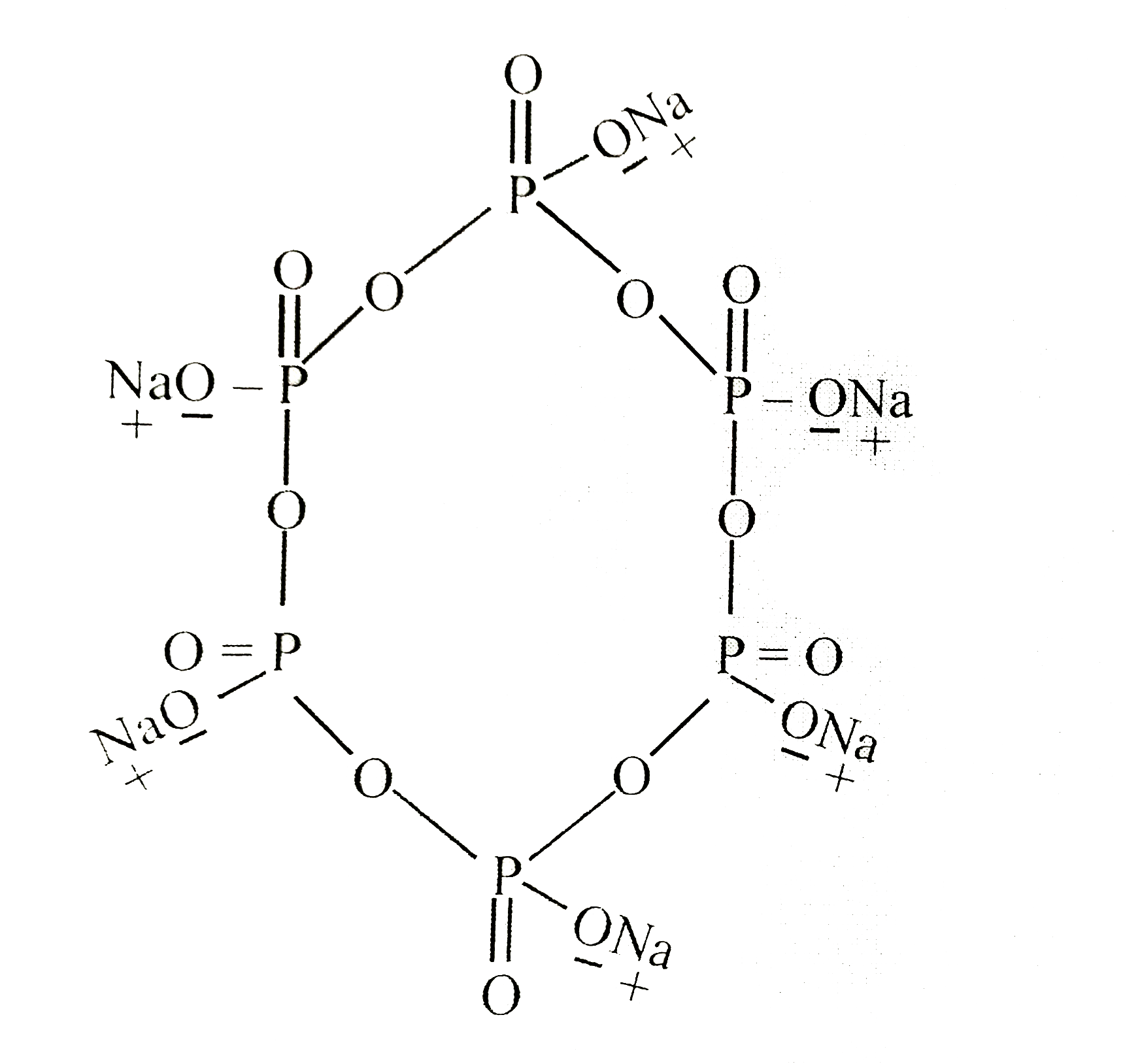
|
|
| 39. |
Read the following statement and explanation and answer as per the options given below: Assertion : HNO_(3) is a stronger acid than HNO_(2) Reason : In HNO_(3) there are two nitrogen-to-oxygen bonds whereas in HNO_(2) there is only one. |
|
Answer» If both assertion and reason are correct, and reason is the correct explanation of the assertion Structure of `HNO_(2) : O-H-N=O`, Strcuture of `HNO_(3)`:  LTBGT The assertion is true but the reason is wrong as can be clearly seen from the above structures. LTBGT The assertion is true but the reason is wrong as can be clearly seen from the above structures.
|
|
| 40. |
Read the following short write-up and answer the questions at the end of it. The name 'silica' covers an entire group of minerals, which have the general formula SiO_(2), the most common of which is quartz. Quartz is a framework silicate with SiO_(4) tetrahedra arranged in spirals. The spirals can turn in a clockwise to anticlockwise direction-a frature that results inthere being two mirror images, optically active, varieties of quartz. The silicate anion in the mineral kionite is a chain of three SiO_(4) tetrahedra that share corners with adjacent tetrahedra. The mineral also contains Ca^(2+) ions, Cu^(2+) ions, and water molecules in a 1:1:1 ratio mineral is represented as : |
|
Answer» `CaCuSi_(3)O_(10).H_(2)O` |
|
| 41. |
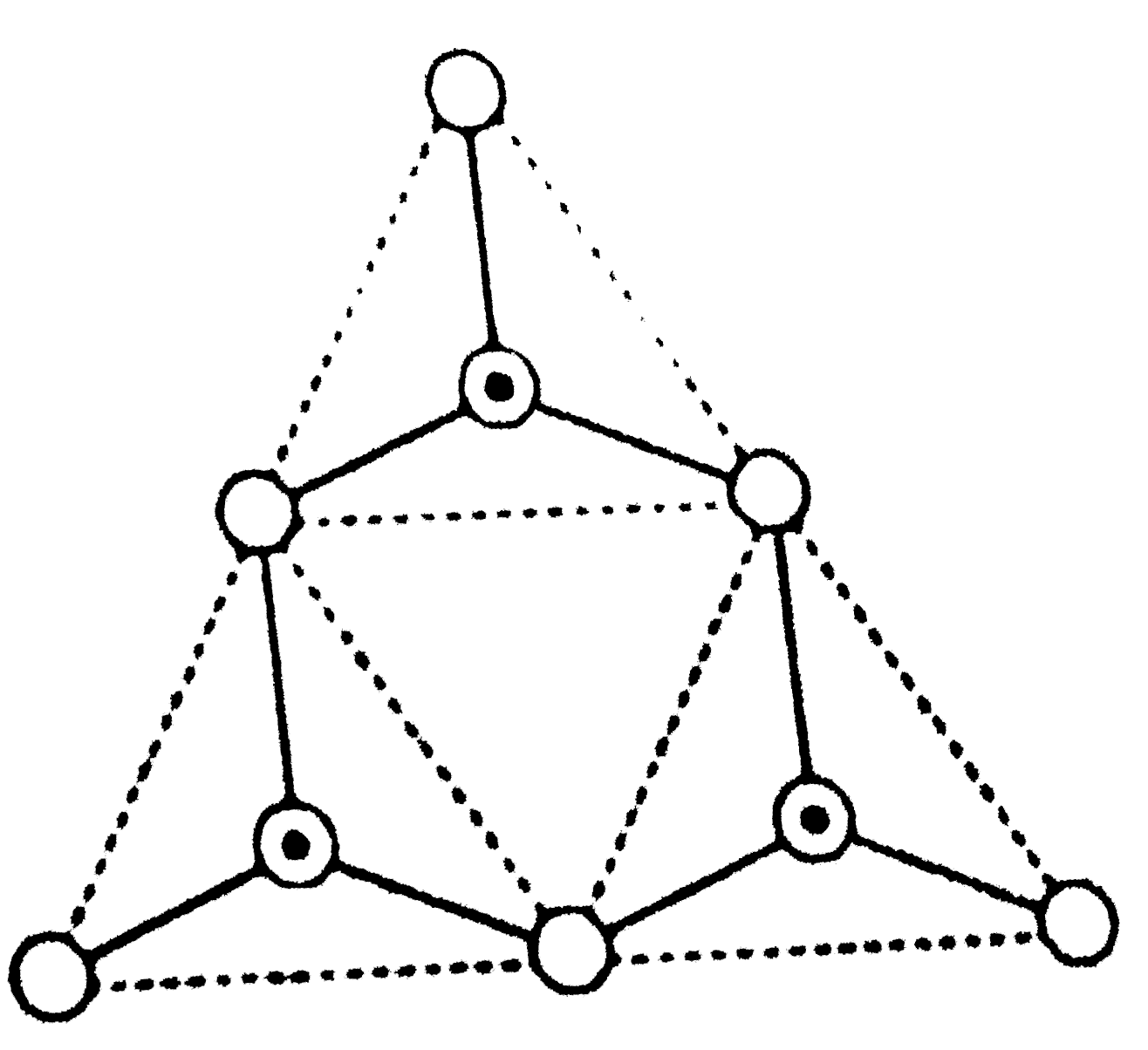
Read the following short write-up and answr the questions at the end of it The name 'silica' covers an entire group of minerals, which have the general formula SiO_(2), the most common of which is quartz. Quartz is a framworksilicate with SiO_(4) tetrahdera arranged in spirals. the spirals can turnin a clockwise or anticlockwise direction-a feature that results in there being two mirror images, optically active, varieties of quartz. Si_(3)O_(9)^(6-) (having three tetrahderal) is represent as : |
|
Answer»
|
|
| 42. |
Read the following short write-up and answr the questions at the end of it The name 'silica' covers an entire group of minerals, which have the general formula SiO_(2), the most common of which is quartz. Quartz is a framworksilicate with SiO_(4) tetrahdera arranged in spirals. the spirals can turnin a clockwise or anticlockwise direction-a feature that results in there being two mirror images, optically active, varieties of quartz. In the given following emperical formulae of silicates shared and unshared oxygen corner are same by each tetrahedron in. |
|
Answer» `Si_(2)O_(7)^(6 Theta)` |
|
| 43. |
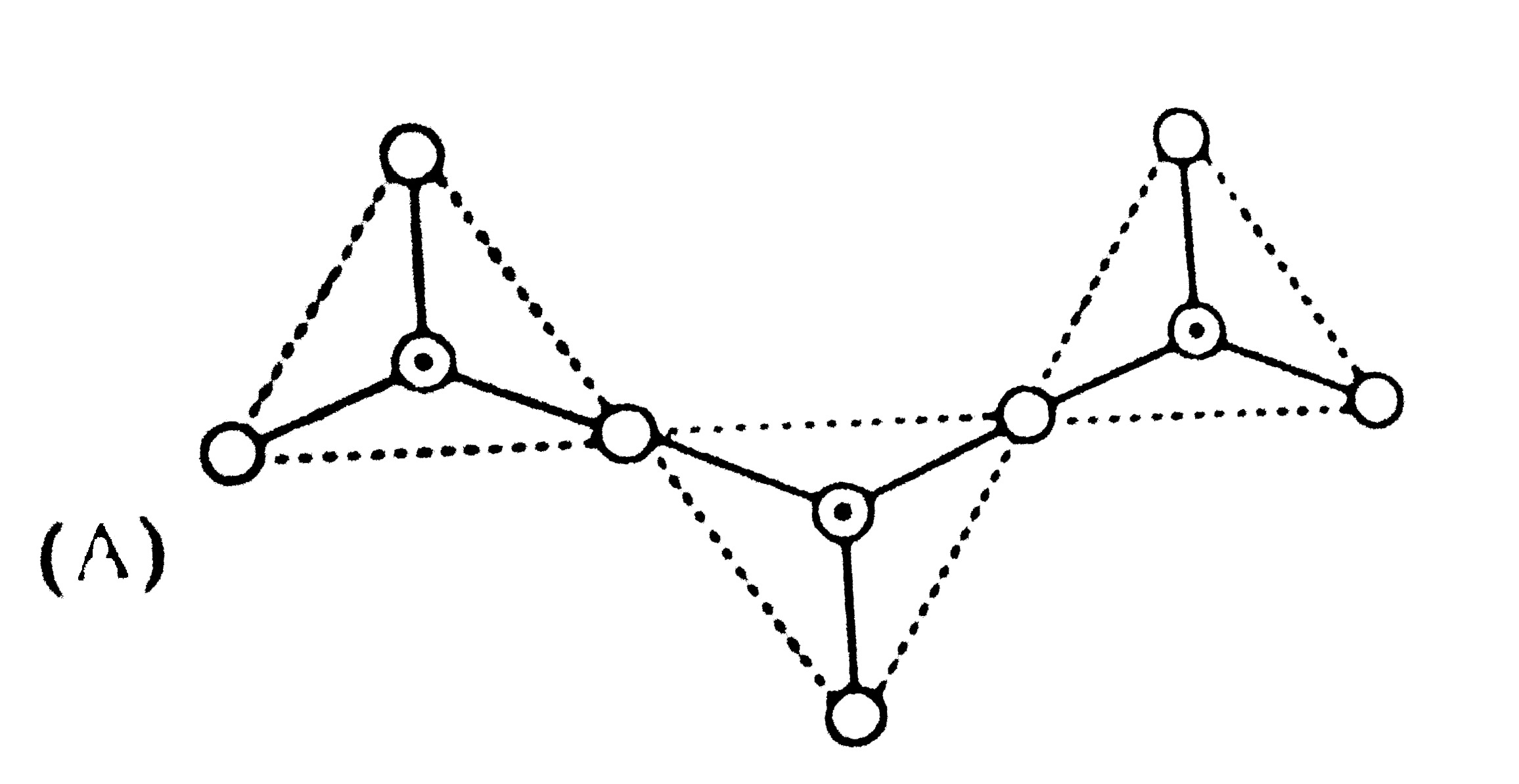
Read the following short write-up and answr the questions at the end of it The name 'silica' covers an entire group of minerals, which have the general formula SiO_(2), the most common of which is quartz. Quartz is a framworksilicate with SiO_(4) tetrahdera arranged in spirals. the spirals can turnin a clockwise or anticlockwise direction-a feature that results in there being two mirror images, optically active, varieties of quartz. The following picture represents various silicate anions. their formulae are respectively. |
|
Answer» `SiO_(3)^(2-),Si_(3)O_(7)^(2-)` |
|
| 44. |
Read the following short write-up and answer the questions at the end of it. The name 'silica' covers an entire group of minerals, which have the general formula SiO_(2), the most common of which is quartz. Quartz is a framework silicate with SiO_(4) tetrahedra arranged in spirals. The spirals can turn in a clockwise to anticlockwise direction-a frature that results inthere being two mirror images, optically active, varieties of quartz. Si_(3)O_(9)^(6-) ( having three tetrahedral units) is represented as: |
| Answer» Answer :B | |
| 45. |
Read the following short write-up and answer the questions at the end of it. The name 'silica' covers an entire group of minerals, which have the general formula SiO_(2), the most common of which is quartz. Quartz is a framework silicate with SiO_(4) tetrahedra arranged in spirals. The spirals can turn in a clockwise to anticlockwise direction-a frature that results inthere being two mirror images, optically active, varieties of quartz. The following pictures represent various silicate anions. Their formulae are respectively |
|
Answer» `SiO_(3)^(2-) ""Si_(3)O_(7)^(2-)` |
|
| 46. |
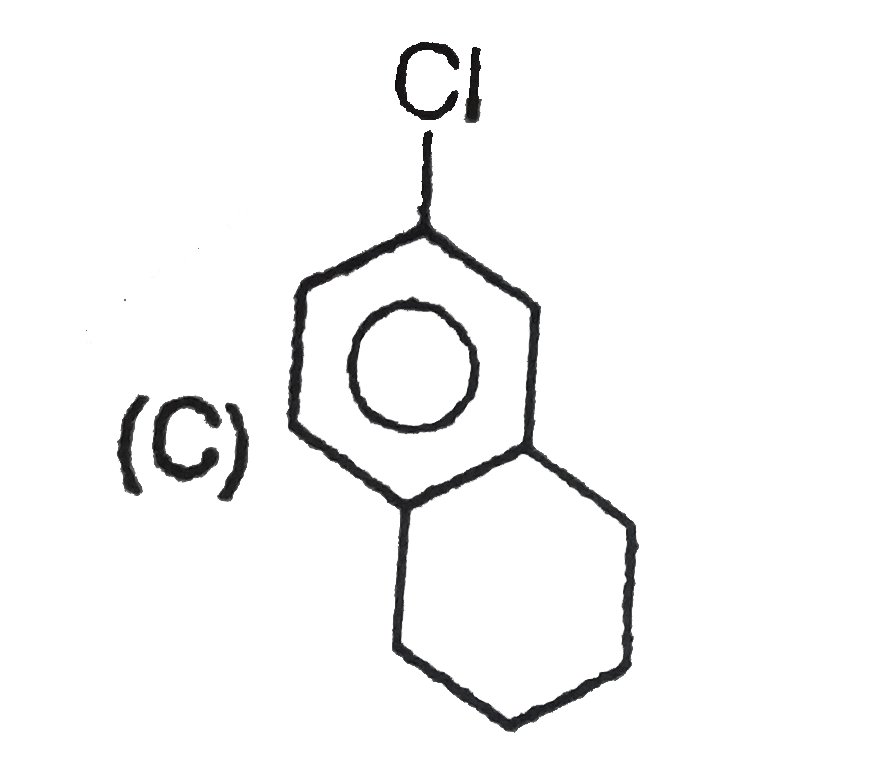
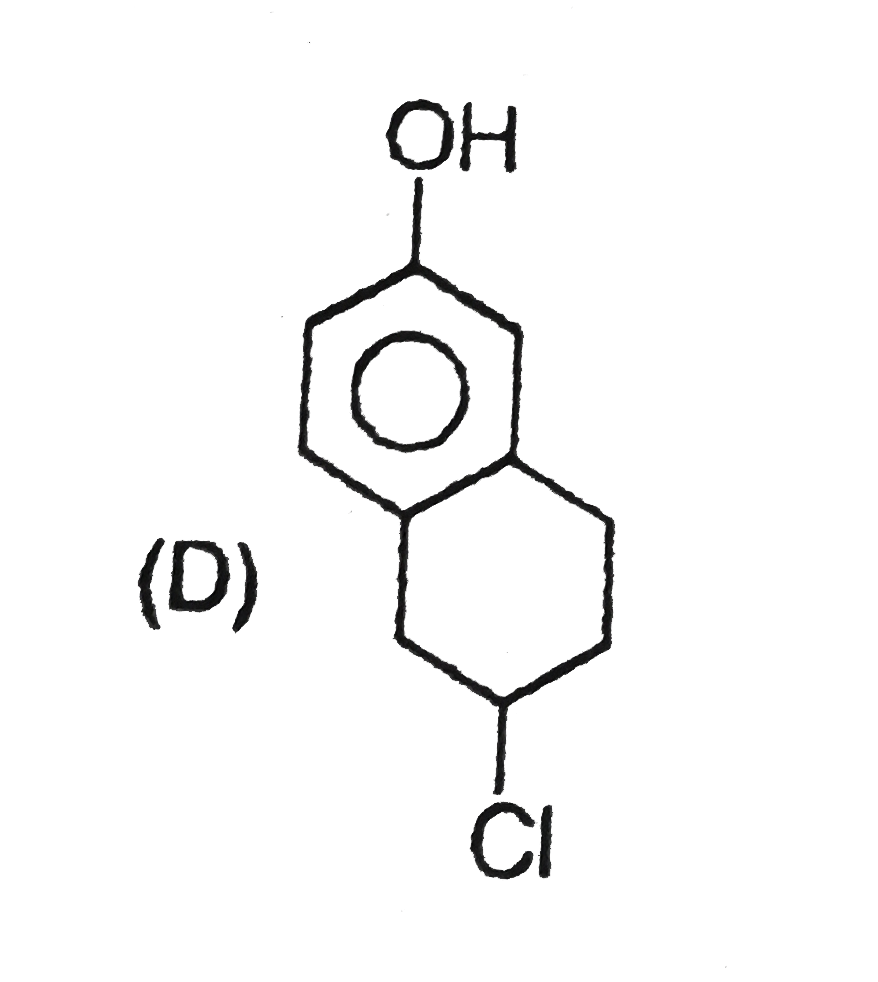
Read the following road map carefully |
|
Answer» Both the ethers OBTAINED by the TWO routes have OPPOSITE but equal OPTICAL rotation 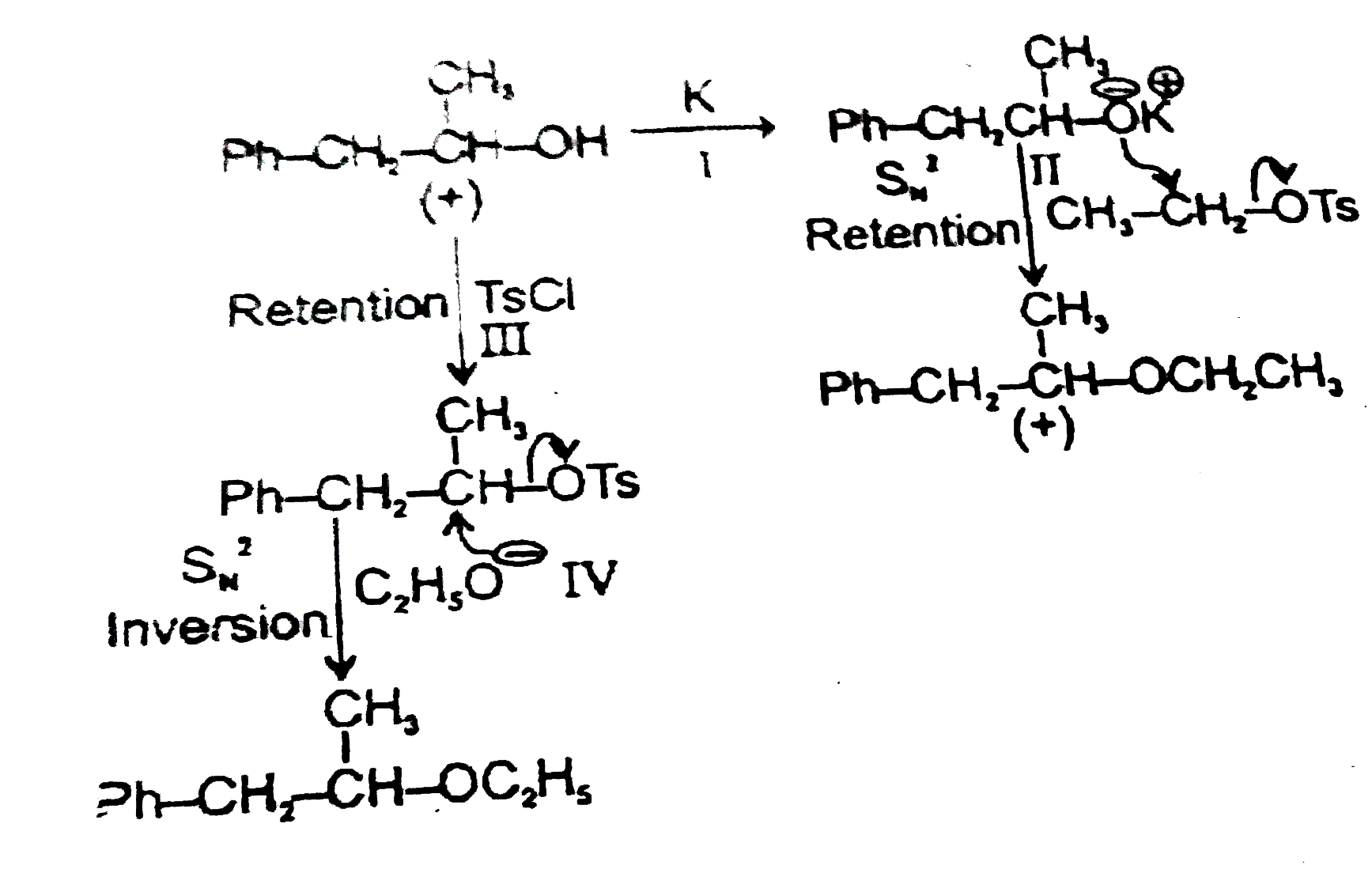
|
|
| 48. |
Read the following reaction When treating 'S' with strong base, product obtained is |
|
Answer»
|
|
| 49. |
Read the following reaction CH-=CHoverset(NaOCl)toPunderset(Ether)overset(Mg)toQoverset(undersetunderset(Cl)(|)(2CH_2)-CHO)toRoverset(KOH)toSunderset(H_2O)overset(NaOH)to Tunderset((i)DiL KMnO_4)overset((i)H2//Pd.//BaSO_4)toU & U' 'Q' is |
|
Answer» ClMgC=CMgCl |
|
| 50. |
Read the following reaction Compound 'Q' is |
|
Answer»
|
|