Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
R-C-=N can be reduced to RCH_(2)NH using the reducing agent. |
| Answer» Solution :`R-C-=N overset(H_(2)//Ni)underset("or "LiAH_(4))toR-CH_(2)NH_(2)` | |
| 2. |
R - C -= underset((2)CH_3CH_2O)overset((1)NaNH_2)(to) product |
|
Answer» `R - C-= C - R ` |
|
| 3. |
R - 2 bromooctane underset((ii)CO_(2))overset((i)Mg)(rarr) (iii) H^(+) X_(i)[X] is |
| Answer» ANSWER :C | |
| 4. |
R-(-)-2-Bromooctane on treatment with aqueous KOH mainly gives 2-octanol. Which of the following is true ? |
|
Answer» Optically active with R-CONFIGURATION |
|
| 5. |
(R)-2-bromooctane undergoes alkaline hydrolysis to yield (S)-2-octanol. Predict the stereochemistry (inversion or retention) of the reaction. |
|
Answer» Solution :`##CHY_CHE_ORG_XII_P2_U10_E01_073_S01.png" width="80%"> Inversion of CONFIGURATION occurs. |
|
| 6. |
(R)-2-Bromooctane reacts with hydrogen sulphide (HS^(-)) ion and gives (S)-2-octanethiol with inversion of configuration at the stereocentre. Can we plan to get (R)-2-octanethiol from (R)-2-bromooctane? |
Answer» Solution :(R)-2-octanethiol can be OBTAINED from (R)-2-bromooctane by DOUBLE inversion to GET net retention of configuration. For EXAMPLE, an `S_(N)2` reaction with `I^(-)` ion would give (S)-2-io- dooctane. Then, it is reacted with HS-to give (R)-2-octanethiol. 
|
|
| 7. |
(R)-2-Bromooctane reacts with NaSH to form (S)-2-octanethiol with inversion of configuration at the stereocentre. How cann we obtain (R)-2-octanethiol from (R)-2-bromoctane? |
Answer» Solution :We know that `S_(N)2` reactions proceeds with inversion of configuration at the STEREOCENTRE. If however, two `S_(N)2` reactions are carried out at the same stereocentre of a compound, retention of configuration will OCCUR. Thus, (R)-2-octanethiol can be obtained from (R)-2-bromooctane by first reacting it with NaI in ACETONE and then with NASH in ethanol. 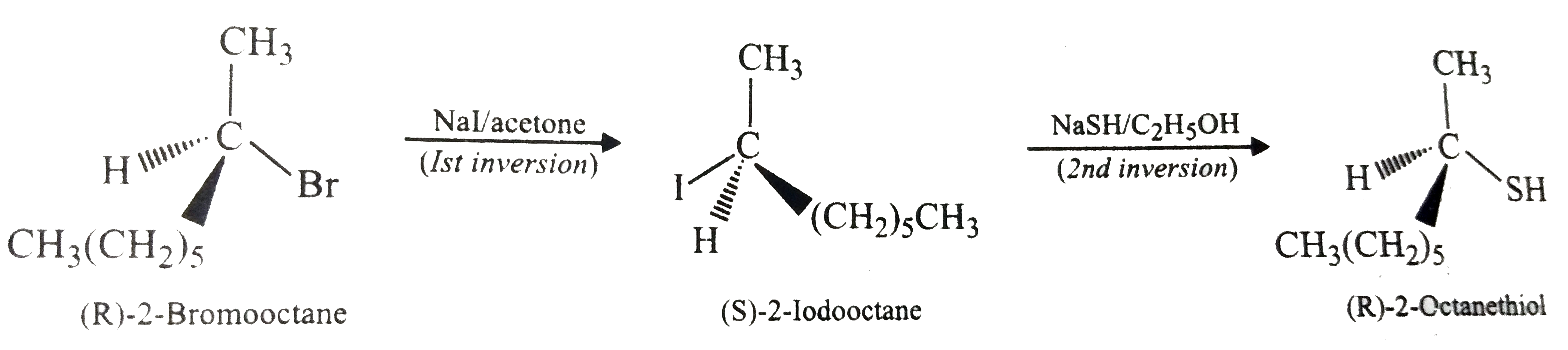
|
|
| 8. |
Quinones are good electron acceptors, party because reduction restores aromaticity. Q+2e^(-)+2H^(o+)rarr H_(2)Q Give the decreasing order of E^(c-)._(reduction) of the following quinones : |
|
Answer» Solution :`a.` Decreasing order or `E^(c-)._(reduction):` `IIIgtIgtII` `b. IIIgtIIgtI""c.IIgtI` `a.` After reduction, products are `:` 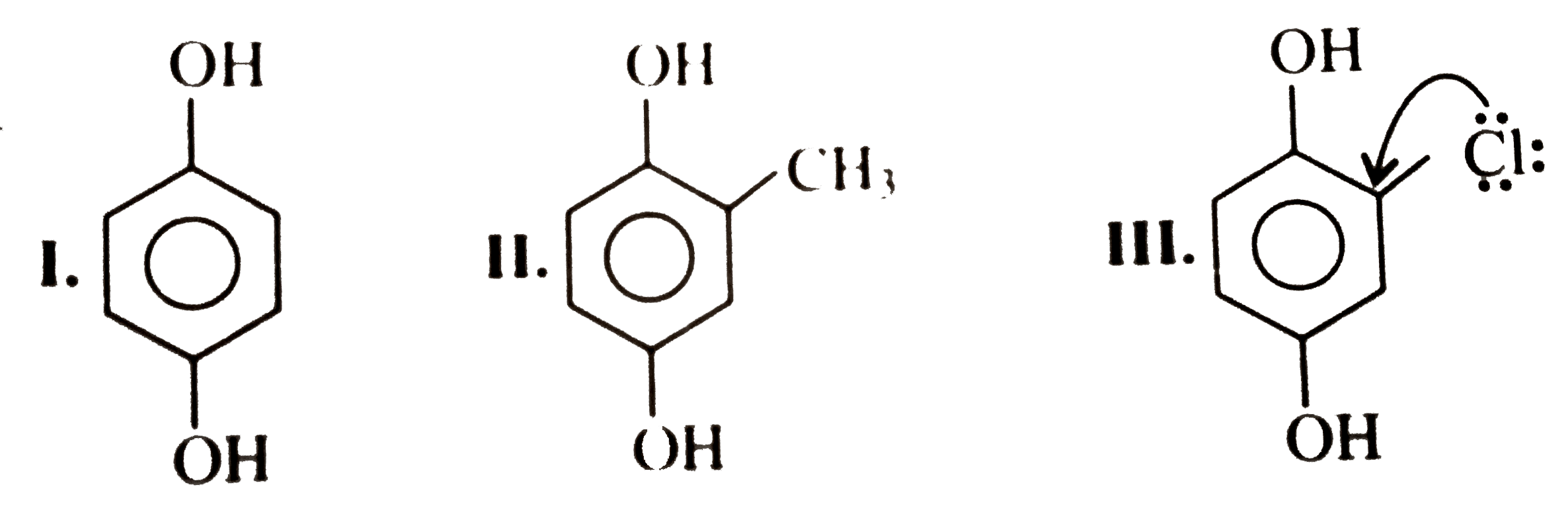 Reactivity order `: IIgtIgtIII` Stability order `: IIIgtIgtII` `III`is more stable due to `+R(` or `+M)` effect of `Cl` than `+I` and `H.C.` effect of `CH_(3)`. More stable the product means FASTER the reduction of quinone to hydroquinone and therefore higher `E^(c-)._(red)` value `:.` Decreasing order of `E^(c-)._(red)` value in `(a): IIIgtIgtII.` `b.` Stability order `: IgtIIgtIII` Reactivity order `: IIIgtIIgtI` `(III)` is more easily reducedt to  because it generates two aromatic rings which have more resonance energy and greater stability than the reaction of `(I)` and `(II)`, which would generate only one benzene ring. because it generates two aromatic rings which have more resonance energy and greater stability than the reaction of `(I)` and `(II)`, which would generate only one benzene ring. Both `(I)` and `(II)` on reduction GIVE one benzene ring, but `(II)` is less stable `(` more reactive `)` than `I` because adjacent `(C=O)` groups in `(II)` make it less stable . 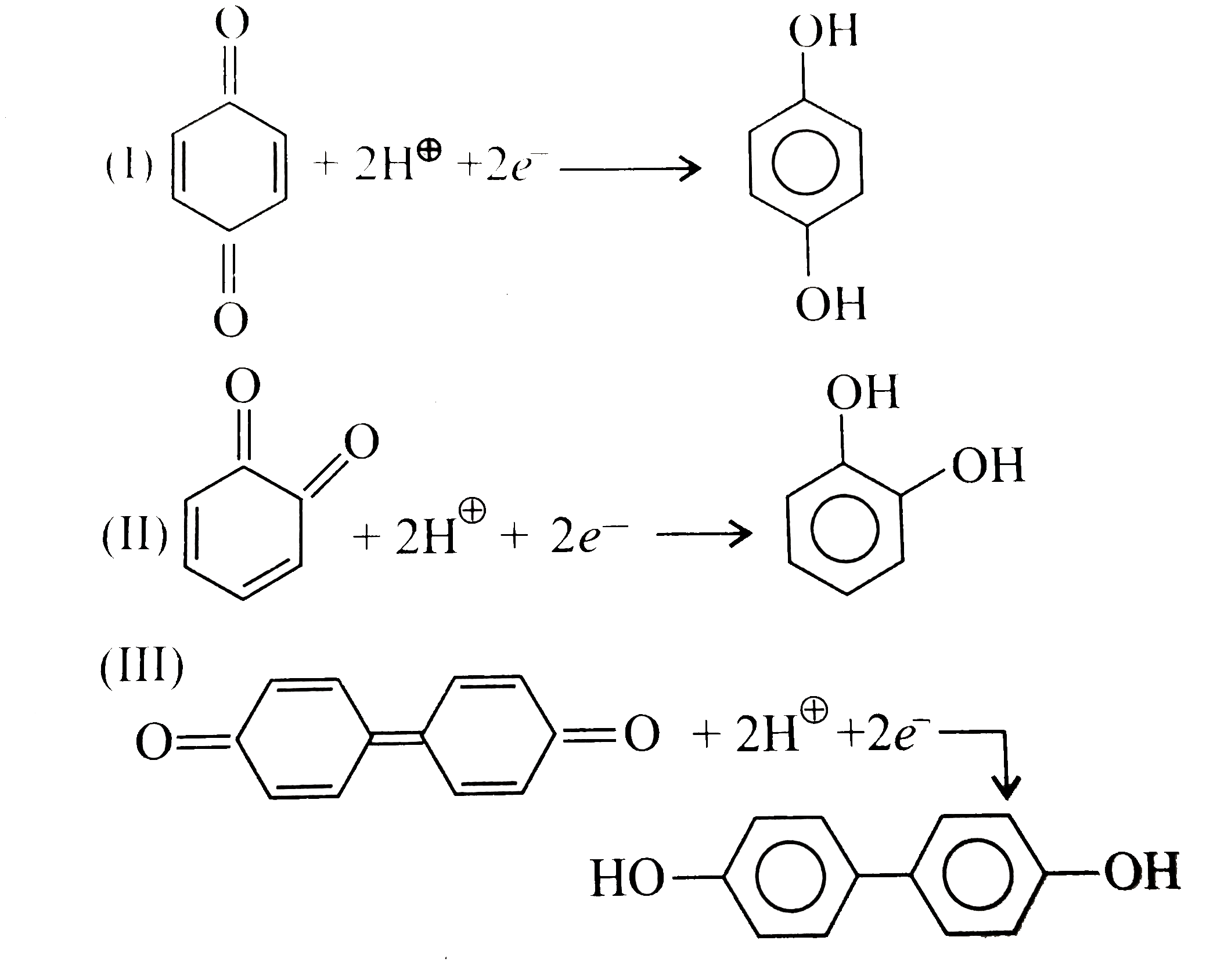 The reaction is representes as follows `:` `Q+2H^(o+)+2e^(-) rarr H_(2)O` `:.` Reduction potential `(E^(c-)._(Q|H_(2)O)):IIgtI` . `c.` Stability order `: IgtII` Reactivity order `: IIgtI` Standard reduction potential `(E^(c-)._(Q|H_(2)O)):IIgtI` 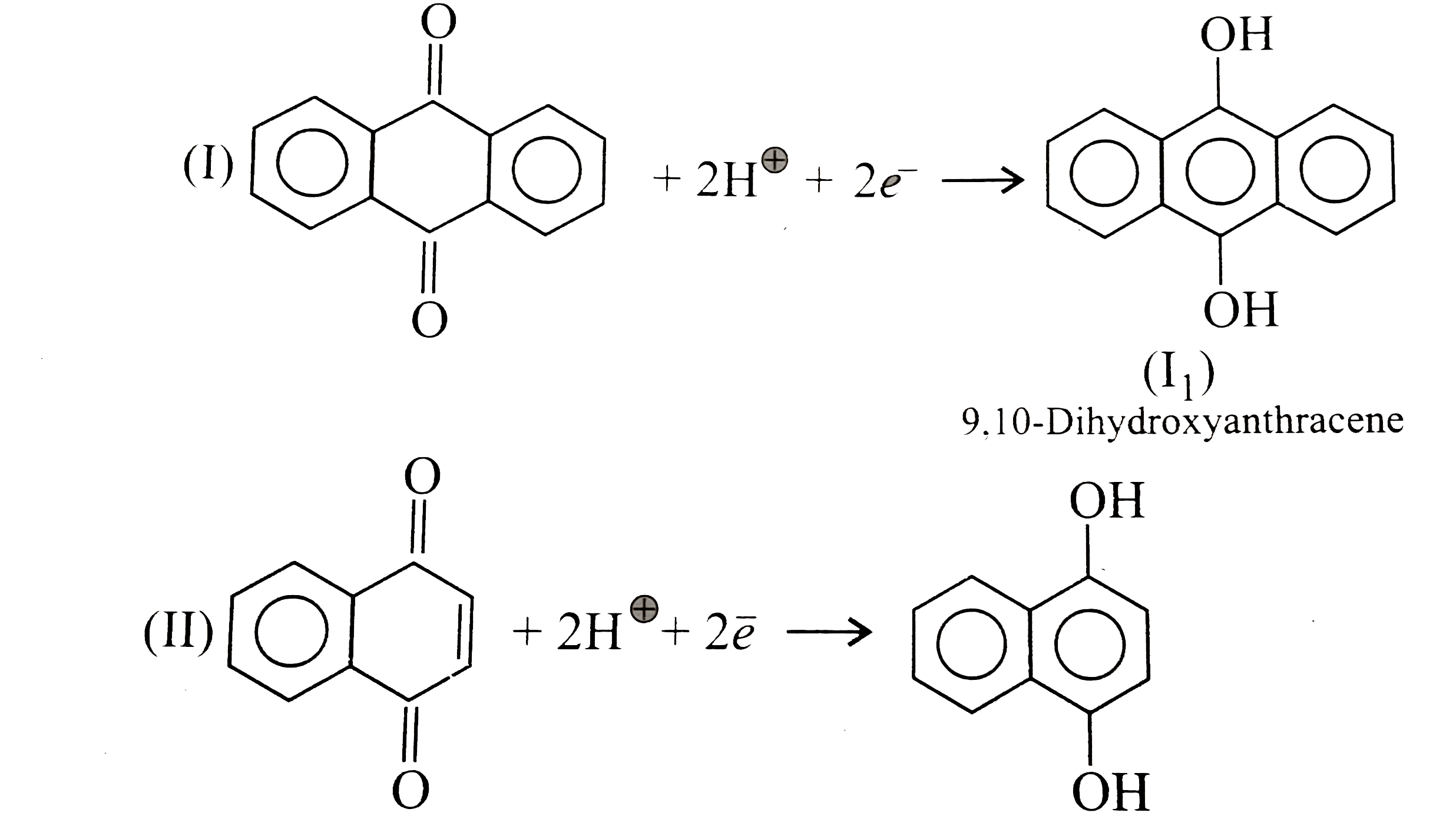 `(I)` has two individual benzene rings whose combined resonance energy along with that ot two `(C=O)` groups is more than the resonance energy of three benzene rings of reduced product `(I_(1))`. |
|
| 10. |
Question given below are based on electronic configuration of the element. The three X,Y and Z with the electronic configurations shown below all formhydrides: Element "" Electronic configuration X""1s^(2),2s^(2),2p^(3) Y""1s^(2),2s^(2),2p^(6),3s^(1) Z""1s^(2),2s^(2),2p^(6),3s^(2),3d^(10),4s^(2),4p^(5) Whcih of the properties (A,B,C or D) correctly list properties of the hydrides of these elements |
|
Answer» |
|
| 11. |
Quaternay ammoniumhalideis heatedwith moistsilveroxide gives |
|
Answer» quaternaryammoniumhydroxide |
|
| 12. |
Quaternary structure of haemoglobin consists of .............subunits , two of the subunits are identical and contain ............amino acid residues and the other two subunits are also identical and contain. Amino acid residues. |
| Answer» SOLUTION :FOUR , 146,141 | |
| 13. |
Quaternay ammonium hydroxidecontainingall groupare methyl, onheatedgives . |
| Answer» Answer :C | |
| 14. |
Quaternary ammoniumhalide reacts with moist silver oxide to produce _________ |
|
Answer» PRIMARY AMINE |
|
| 15. |
Quaternary ammonium salts. |
| Answer» Solution :The tetra alkyl derivatives of ammonium salts `(Noverset(+)(NH_(4))X^(-))` in which all the four hydrogen atoms ATTACHED to nitrogen are replaced by four, same or different alkyl groups are called QUATERNARY ammonium salts `(R_(4)overset(+)NX^(-))` . | |
| 16. |
Quartz is an example of: |
|
Answer» IONIC crystal |
|
| 17. |
Quantium efficiency or quantum yield (phi) of photochemical reaction is given by: phi = ("moles of the substance reacted")/("moles of photons absorbed") Absorption of UV radiation decomposes A according to the reaction 2A overset (hv) rarr B+3C In a first order reaction 75% of the reactant disappears in 1.386 h the rate constant of the reaction is close to: |
|
Answer» `7.2xx10^(-1) s^(-1)` |
|
| 18. |
Quantium efficiency or quantum yield (phi) of photochemical reaction is given by: phi = ("moles of the substance reacted")/("moles of photons absorbed") Absorption of UV radiation decomposes A according to the reaction 2A overset (hv) rarr B+3C The quantum yield of the reaction at 330 nm is 0.4 A sample of 'A' absorbs monochromatic radiation at 330 nm at the rate of 7.2xx10^(-3)Js^(-1) (Given N_(A)=6xx10^(23),h=6.6xx10^(-34) in S.I unit) The rate of formation of C (mol //s) is |
| Answer» Answer :A | |
| 19. |
Quantity of electricity which liberates 108 g of Ag from AgNO_(3) solution is equal to_________. |
|
Answer» ONE ampere |
|
| 20. |
Quantity of electricity is measured in |
|
Answer» `"AMP"^(-1)` SEC |
|
| 21. |
Quantity of electricity is measured in : |
| Answer» Answer :A | |
| 22. |
Quantitative analysis of a compound shows that it contains 0.110 mole of 'C'. 0.055 mole of 'N' and 0.165 mole of 'O' its molecular mass is about 270. how many atoms of carbon are there in empirical and molecular formulae of the compound respectively? |
|
Answer» `{:("EMPIRICAL FORMULA","MOLECULAR formula"),(1,3):}` |
|
| 23. |
Q. When H_(2)S gas was passed into filtrate (P), a coloured precipitate was obtained, then cation present in the filtrate is: |
|
Answer» `Zn^(2+)(AQ.)` |
|
| 24. |
Q. The process which occurs in going from BtoC is: |
|
Answer» isothermal |
|
| 25. |
Q. The compound C and metal M are: |
| Answer» | |
| 27. |
Q. 'T' cannot be identify by: |
|
Answer» `NH_3` solution * `Fe^(3+)+NH_(4)SCNtoFe(SCN)_(3)` BLOOD red. |
|
| 28. |
Q. The compound B is : |
|
Answer» `Mg(NH_(4))PO_(4)` |
|
| 29. |
Q. The compound A is : |
|
Answer» `CaCl_(2)*2H_(2)O` |
|
| 30. |
Q. Species P and S are respectively: |
|
Answer» `SO_(3)^(2-)(AQ.),S` 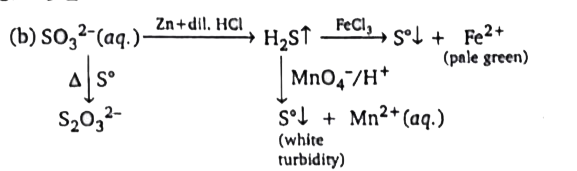
|
|
| 31. |
Q. Precipitate (Q) was treated withdil. HCl and coloured solution was obtained. On passing H_(2)S gas into this solution no precipitate was obtained but colour of the solution changes, then cation present in the precipitate (Q) can be identified by: |
|
Answer» `Na_(2)S_(2)O_(3)` SOLUTION |
|
| 32. |
Q. Incorrect statement 'Y' changes on heating: |
|
Answer» Colour of 'Y' changes on heating |
|
| 33. |
Q. identify salt 'A'? |
|
Answer»
|
|
| 35. |
Q. Compound 'X' is: |
|
Answer» `NaNO_(3)` 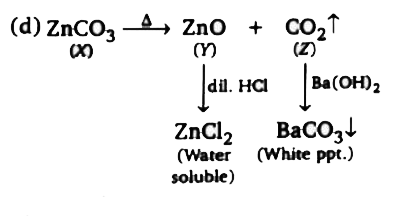
|
|
| 39. |
Pyruvic acid is obtained by |
|
Answer» Oxidation of acetaldehyde cyanohydrin |
|
| 40. |
Pyrosilicate ion is |
|
Answer» `SiO_(2)^(2-)` |
|
| 41. |
Pyrosulphurous acid is |
|
Answer» `H_(2)S_(2)O_(5)` |
|
| 42. |
Pyrophosorus acidH_(2)P_(2)O_5 |
|
Answer» <P>Cantain P + 5 oxidation state |
|
| 44. |
Pyrophosphoric acid on heating to 600^(@)C gives : |
|
Answer» `HPO_(3)` |
|
| 45. |
Pyrolustie on heating with KOH in the presence of air gives dark green compound (A).The solution of (A) on treatment with H_(2)SO_(4) gives a purple colouredcompound (B) which gives the following reactions : (i) Alkaline solution of B on reaction with KI changes it into compound (C ). (ii) The color of the compound (B) disappears on treatment with acidic soluiont of FeSO_(4). (iii) On reactionwith conc. H_(2)SO_(4) compound (B) gives another compound (D) which can decompose to produce compound (E ) along with oxygen gas. Identify compounds (A) to (E )and write balanced chemical equation involvedin eachcase. |
|
Answer» Solution :Chemical equation for the formation of A `:` ` underset("Pyrolusite")(2MnO_(2)) + 4KOH + O_(2) rarr underset("Potassium manganate ") underset((A))(2K_(2)MnO_(4)) +2H_(2)O` Chemical equation for theformation of B form A `:` `3K_(2)MnO_(4) + 2H_(2)SO_(4) rarr underset("POTASSIUMPERMANGANATE ")underset((B))(2KMnO_(4))+ MnO_(2) + 2K_(2)SO_(4) + 2H_(2)O` Chemical equationsof reaction of COMPOUND B `:` (i)` 2KMnO_(4) + H_(2)O+ KI rarr 2MnO_(2) + 2KOH + underset("Potassium iodate")underset((C ))(KIO_(3))` (ii) `2KMnO_(4)+ 10 FeSO_(4) + 8 H_(2)SO_(4) rarr K_(2)SO_(4) + 2MnSO_(4)+ 5Fe_(2)(SO_(4))_(3) + 8 H_(2)O` `2Mn_(2)O_(7) rarr underset((E )) (4 MnO_(2)) + 3O_(2)` |
|
| 46. |
Pyrolusite is the main ore of manganese in which it is present asIts Mn content is determined by reducing it under acidic condition to Mn^(2+)with the help of oxalate (C_(2)O_(4)^(2-)) ion which in turn gets oxidized to CO_(2) The analytical determination is carried out by adding a known excess volume of (C_(2)O_(4)^(2-)) solution to a suspension of the pyrolusite and digesting the mixture on a hot water bath until all the MnO_(2) has been reduced. The excess unreacted oxalate solution is then titrated with standardized KMnO_(4) solution. Thereby Mn content of ore can be calculated. KMnO_(4)solution is also standardized under acidic condition against oxalate ion wherein MnO_(4)^(-) ion is reduced to Mn^(2+) and (C_(2)O_(4)^(2-)) ion is oxidized to CO_(2)Qsample containing MnO_(2)is treated with HCl liberating Cl_(2) The Cl_(2) is passed into a solution of KI and 30cm^(3) ofhypo is required to titrate the liberated iodine. What is the percentage of MnO_(2) in the sample ? |
|
Answer» `22.64%` `Cl_(2)+2KIto2KCl+I_(2)` REACTION involed `I_(2)+2S_(2)O_(3)^(2-)to2I+S_(4)O_(6)` |
|
| 47. |
Pyrolusite is the main ore of manganese in which it is present asIts Mn content is determined by reducing it under acidic condition to Mn^(2+)with the help of oxalate (C_(2)O_(4)^(2-)) ion which in turn gets oxidized to CO_(2) The analytical determination is carried out by adding a known excess volume of (C_(2)O_(4)^(2-)) solution to a suspension of the pyrolusite and digesting the mixture on a hot water bath until all the MnO_(2) has been reduced. The excess unreacted oxalate solution is then titrated with standardized KMnO_(4) solution. Thereby Mn content of ore can be calculated. KMnO_(4)solution is also standardized under acidic condition against oxalate ion wherein MnO_(4)^(-) ion is reduced to Mn^(2+) and (C_(2)O_(4)^(2-)) ion is oxidized to CO_(2)Qof pyrolusite ore were treated withof pure ferrous ammonium sulphate FeSO_(4).(NH_(4))_(2)SO_(4)6h_(2)O) and dil. H_(2)SO_(4)After the reaction solution was diluted to.of dilute solution required 10 mL of 0.1NK_(2)Cr_(2)O_(7) solution. What is the amount of ferrous ammonium sulphate in 500 mL of solution, which is neutralized by K_(2)Cr_(2)O_(7)solution ? |
|
Answer» 16.08g therefore In 500mL, MEQ of ferrous ammounium sulphate=10meq `implies10=g/(392)xx1000` `implies g=3.92grams` |
|
| 48. |
Pyrolusite is the main ore of manganese in which it is present asIts Mn content is determined by reducing it under acidic condition to Mn^(2+)with the help of oxalate (C_(2)O_(4)^(2-)) ion which in turn gets oxidized to CO_(2) The analytical determination is carried out by adding a known excess volume of (C_(2)O_(4)^(2-)) solution to a suspension of the pyrolusite and digesting the mixture on a hot water bath until all the MnO_(2) has been reduced. The excess unreacted oxalate solution is then titrated with standardized KMnO_(4) solution. Thereby Mn content of ore can be calculated. KMnO_(4)solution is also standardized under acidic condition against oxalate ion wherein MnO_(4)^(-) ion is reduced to Mn^(2+) and (C_(2)O_(4)^(2-)) ion is oxidized to CO_(2)Q What is the percentage of pure MnO_(2) in the above question ? |
|
Answer» `50.9%` or number of equivalent of `MnO_(2)=(16.08)/(392)=0,041` `implies x=1.781 therefore %purity=1.781/3xx100=59.4%` |
|
| 49. |
Pyrolusite in MnO_(2) is used to prepare KMnO_(4). steps are MnO_(2) overset(I) MnO_(4)^(2-) overset(II) to MnO_(4)^(-). The I and II are, |
|
Answer» I=Fused with KOH/airII=electrolytic OXIDATION |
|
| 50. |
Pyrolusite is the main ore of manganese in which it is present asIts Mn content is determined by reducing it under acidic condition to Mn^(2+)with the help of oxalate (C_(2)O_(4)^(2-)) ion which in turn gets oxidized to CO_(2) The analytical determination is carried out by adding a known excess volume of (C_(2)O_(4)^(2-)) solution to a suspension of the pyrolusite and digesting the mixture on a hot water bath until all the MnO_(2) has been reduced. The excess unreacted oxalate solution is then titrated with standardized KMnO_(4) solution. Thereby Mn content of ore can be calculated. KMnO_(4)solution is also standardized under acidic condition against oxalate ion wherein MnO_(4)^(-) ion is reduced to Mn^(2+) and (C_(2)O_(4)^(2-)) ion is oxidized to CO_(2)Q An alternative method for this analysis may be the replacement of oxalate with Fe^(2+) But this is less frequently used because it requires some special care otherwise we get low analytical results. The probable reaction responsible for this fact is : |
|
Answer» presence of oxygen can cause OXIDATION, `Fe^(2+)toFe^(3+)` |
|