Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Pair of smallest and largest species of radii among Mg, Al, Mg^(+2), Al^(+3) is :- |
|
Answer» `MG^(+2), Mg` |
|
| 2. |
Paris green is: |
|
Answer» `CU(CH_3COO)_2` |
|
| 3. |
Pair of isobar is : |
|
Answer» `._(6)^(77)C,_(7)^(13)N` |
|
| 4. |
Pair of compound which is planar :- |
|
Answer» <P>`[NI(CN)_(4)]^(-4), [PtCl_(4)]^(-2)` |
|
| 6. |
Packing efficiency is the percentage of total space filled by the particles.Which of the following lattices has the highest packing efficiency? Simple cubic lattice, body centered cubic lattice, hexagonal close packed lattice |
| Answer» SOLUTION :HEXAGONAL CLOSE PACKED LATTICE. | |
| 7. |
Packing fraction is related to : |
|
Answer» NUCLEAR spin |
|
| 8. |
Packing efficiency by arrangement of atoms in two dimensional hexagonal close packing is |
|
Answer» 60.43  let radius of the sphere=r AREA occupied by sphere in hexagonal close packing `pir^(2)+6XX((1)/(6)xxpir^(2))=2pir^(2)` Area of hexagonal`=6xx[(SQRT(sqrt(3))/(4)xx(2r)^(2)]` `=6xx(sqrt(3))/(4)xx4r^(2)` `=6sqrt(3)xxr^(2)` % occupied by `=(2pir^(2))/(6xxsqrt(3)xxr^(2))xx100` `=(2xx3.14)/(6xxsqrt(3))xx100=60.43%` |
|
| 9. |
Packing efficiency (Packing Fraction) |
| Answer» Solution :It is the percentage of total SPACE in the tunit cell OCCUPIED by the constituent particles like ATOMS, ions or MOLECULES of the crystal. | |
| 10. |
P_(A)=320 torr, whenX_(A)=x torr If X_(A)=x+0.1,P_(A)=440 then P_(A)^(0)=200x, find x. |
|
Answer» |
|
| 11. |
P_(A)^(0)=10 torr, 1g of B is dissolved in 20g of A, its vapour pressure reduced to 9.0 torr . If mwt of A is 200, M.wt of B is 30x is |
|
Answer» |
|
| 12. |
P_A and P_B are the vapour pressure of pure liquid components. A and B, respectively of an ideal binary solution. If x_A represents the mole fraction of component A, the total pressure of the solution will be |
|
Answer» `P_B+x_A (P_B-P_A)` |
|
| 13. |
p_(A) and p_(B) are the vapour pressure of pure liquid components, A and B, respectively of an ideal binary solution. If x_(A) represents the mole fraction of component A, the total pressure of the solution will be |
|
Answer» <P>`p_(A) + x_(A)(p_(B)-p_(A))` `p=x_(A)p_(A)+x_(B)p_(B)` For binary solution, `x_(A)+x_(B)=1` or `[x_(B)=1-x_(A)]` `p=x_(A)p_(A)+(1-x_(A))p_(B)` `=x_(A)p_(A)+p_(B)-x_(A)p_(B)` or `=p_(B)+x_(A)(p_(A)-p_(B))` |
|
| 14. |
P_(A)and P_(B) are the vapour pressure of pure liquid components ,Aand B respectively of an ideal binary solution,If x_(A) represents the mole fraction of component A, the total pressure of the solution will be |
|
Answer» `p_(A)+x_(A)(P_(B)-P_(A))` `P=P_(A)x_(A)+P_(B)x_(B)` For BINARY solutions `x_(A)+x_(B)=1,x_(B)=1-x_(A)` ` P=P_(A)x_(A)+P_(B)(1-x_(A))` `P=P_(B)+x_(A)(P_(A)-P_(B))` |
|
| 15. |
P_(4)S_(3) +8O_(2) to P_(4)O_(10)+3SO_(2) Calculate mass of P_(4)S_(3) is required at least 9.6 gm of each product. |
|
Answer» |
|
| 16. |
P_(4)("white")+SOCl_(2)rarrX("halide of P")+Y("oxide")+Z("halide of S") Incorrect statement among the following is : |
|
Answer» X on hydrolysis gives a dibasic acid |
|
| 17. |
P_(4)overset(Ba(OH)_(2))underset(Delta)(to)underset("salt")(X) + underset("gas")(Y) uarr Xoverset("dil." H_2SO_4)rarrZdarr+underset("acid")WWhich is incorrect about Y? |
|
Answer» It usually IGNITES due to PRESENCE of `P_2H_4` Y is `PH_3`  - less soluble than `NH_3` in water as it is weak base - less soluble than `NH_3` in water as it is weak base `-Ca_(3)P_(2)+6H_(2)Orarr3Ca(OH)_2+2PH_(3)` `-PH_3` contains impurity diphosphine `(P_2H_4)` - which catches fire readily and ignites `PH_3` |
|
| 18. |
P_(4)overset(Ba(OH)_(2))underset(Delta)(to)underset("salt")(X) + underset("gas")(Y) uarr Xoverset("dil." H_2SO_4)rarrZdarr+underset("acid")W Which is correct in view of 'W' ? |
|
Answer» It's shape is planar Y is `PH_3` is WEAKER base than `NH_3` 
|
|
| 19. |
P_(4)overset(Ba(OH)_(2))underset(Delta)(to)underset("salt")(X) + underset("gas")(Y) uarr Xoverset("dil." H_2SO_4)rarrZdarr+underset("acid")W Which statment is correct ? |
|
Answer» The anion of salt 'X' cannot have a LEWIS base `H_(2)PO_(2)^(-)` Anion of SALF .X.  No Lone pairs on phosphorous - so not a lewi.s base No Lone pairs on phosphorous - so not a lewi.s base
|
|
| 20. |
P_4O_6 reacts with water according to equation P_4O_6 + 6H_2O to4H_3PO_3. Calculate the volume of 0.1 M NaOH solution required to neutralise the acid formed by dissolving 1.1 g of P_(4)O_(6)in H_2O . |
|
Answer» Solution :MOL. Mass of `P_(4)O_(6) = 220` g/mol Moles of `P_(4)O_(6)` dissolved in `H_(2)O = (1.1)/220` `underset(1 mol)(P_(4)O_(6)) + underset(6 "mol")(6H_(2)O) to underset(4 "mol")(4H_(3)PO_(3))` `therefore` Moles of `H_(3)PO_(3)` produced `=4.4/(220)` `underset(1 mol)(H_(3)PO_(3)) + 2NaOHto Na_(2)HPO_(3) + 2H_(2)O` `therefore (4.4)/(220) mol` Moles of NaOH consumed `=(2 xx 4.4)/220 = 8.8/(220)` `therefore` Volume of NaOH REQUIRED `=("Moles of NaOH" xx 1000)/("Molarity of NaOH")` `=(8.8 xx 1000)/(220 xx 0.1)` = 400 mL `V_(NaOH) = 400 mL` |
|
| 21. |
P_(4)O_(6) reacts with water according to equation, P_(4)O_(6) + 6 H_(2)O rarr 4 H_(3)PO_(3). Calculate the volume of 0.1 M NaOH solution required to neutralise the acid formed by dissolving 1.1 g of P_(4)O_(6) in H_(2)O. |
|
Answer» <P> Solution :The chemical equations for the reactions involved are :`{:(""P_(4)O_(6) + 6H_(2)O rarr 4 H_(3)PO_(4)""("Hydrolysis reaction")),(ul(H_(3)PO_(3) + 2 NaOH rarr [4 Na_(2)HPO_(3)+ 2H_(2)O]xx 4) " "("Neutralisation reaction")),(" "underset("= 220 g")underset(4 xx 31 + 6 xx 16)(P_(4)O_(6))+underset("= 320 g")underset(8 xx 40)(8 NaOH) rarr 4 Na_(2)HPO_(3)+2H_(2)O""("Overall reaction")):}` Now 220 g of `P_(4)O_(6)` require NaOH for neutralization = 320 g `therefore` 1.1 g of `P_(4)O_(6)` will require NaOH `= (320)/(220) xx 1.1 = 1.6 g` Now 1000 ML of 0.1 MHCl contain NaOH `= 40 xx 0.1 = 4 g` In other words, 4 g of NaOH are present in 0.1 M NaOH = 1000 mL `therefore` 1.6 g of NaOH will be present in 0.1 M NaOH `=(1000)/(4) xx 1.6 =400 mL = 0.4 L` |
|
| 22. |
P_4O_(6) reacts with water according to equation P_4O_6 + 6H_2O to 4H_3PO_3 . Calculate the volume of 0.1 M NaOH solution required to neutralise the acid formed by dissolving 1.1 g of P_4O_6 in H_2O. |
Answer» SOLUTION :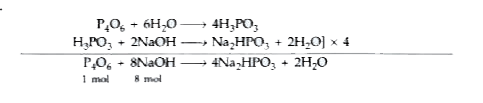 No. of moles in `P_4O_6=(1.1)/(220)` 1 MOL `P_4O_6` requires = 8 mol NAOH `(1.1)/(220)` mol `P_4O_6` required = `8 XX (1.1)/(220)` mol NaOH `=(8.8)/(220) =0.04 mol` NaOH Molarity of NaOH solution is 0.1 M 0.1 mol of NaOH is present in =1 litre 0.04 mol NaOH is present in `=(1)/(0.1) xx 0.04 `litre = 400 mL |
|
| 23. |
P_(4)O_(6) reacts with cold water to give |
| Answer» Solution :`H_(3)PO_(3)` | |
| 24. |
P_(4)O_(6) is the anhydride of the following |
|
Answer» `H_3PO_2` |
|
| 25. |
P_4O_6 and P_4O_10are anhydrides of : |
|
Answer» `H_3PO_3 and H_3PO_4` |
|
| 26. |
P_(4)O_(10) is the anhydride of the following |
|
Answer» `H_3PO_2` |
|
| 27. |
P_(4)O_(10) is not used to dry NH_(3) gas because |
|
Answer» `P_(4)O_(10)` is not a drying agent `P_(4)O_(10) + 12NH_(3) + 6H_(2)O rarr 4(NH_(4))_(3) PO_(4)` |
|
| 28. |
P_(4)O_(10) is not used to dry NH_(3) gas because: |
|
Answer» It is very costly |
|
| 29. |
P_4O_10 is an anhydride of which compound ? |
|
Answer» `H_(3)PO_(2)` |
|
| 30. |
P_(4)O_(10) is an anhydride of : |
|
Answer» `H_(3)PO_(2)` |
|
| 31. |
P_4O_10 has short and long P-O bonds. The number of short P-O bonds in this compound is: |
|
Answer» 1 |
|
| 32. |
P_(4)+8SOCl_(2)to X +4SO_(2)+YIn this equation mention formula of X and Y |
|
Answer» `X=PCl_(5(S)), Y=S_((S))` |
|
| 33. |
P_4 O_(10) is the anhydride of |
|
Answer» `H_(3)PO_(2)` |
|
| 34. |
P^(3-) ion has the valence shell electronic configuration |
|
Answer» `3s^(2)3p^(3)` |
|
| 35. |
P_2O_5 when treated with cold water gives: |
|
Answer» Orthophosphoric acid |
|
| 36. |
P_2O_5 is used extensively as a: |
|
Answer» DEHYDRATING agent |
|
| 37. |
P_(2)O_(5) is anhydride of |
|
Answer» `H_(3)PO_(3)` |
|
| 38. |
P_2O_5 can dimerise but N_2O_5 cannot. Explain with structure. |
|
Answer» Solution :Nitrogen on account of small size and high. electronegativity has a strong tendency to form multiple bonds (`ppi-ppi`) with oxygen and form oxide in open chain structure. HOWEVER DUE to larger and diffused p-orbital, phosphorus is unable to construct such `pi`-bonds and prefers to form single bonds in order to gain STABILITY. As a result, `N_2O_5` does not undergo DIMERISATION whereas `P_2O_5` dimerises to form `P_4O_(10)` For structural detail |
|
| 39. |
P_(2)H_(4) can be romoved from phosphine containing traces of it |
|
Answer» by passing impure `PH_(3)` gas through a freezing mixture When passed through freezing miture the `P_(2)H_(4)` present condenses and pure `PH_(3)` is OBTAINED. When passed through `HI, PH_(3)` is obsorbed FORMING `PH_(4)I.PH_(4)I` when treated with KOH(aq) yields pure phosphine. `PH_(3)+HI to PH_(4)I` `PH_(4)I+KOH(aq) to KI+H_(2)O+PH_(3) uparrow` |
|
| 40. |
P_2 O_5 is used extensively as a/an |
|
Answer» <P>REDUCING agent |
|
| 41. |
{:((P),1/2A_2+3/2B_2hArrD_2,(1),1//4),((Q),2D_2hArrA_2+3B_2,(2),64),((R ),A_2+3B_2hArr2D_2,(3),1//64),((S),D_2hArrB_2+1/2C,(4),8):} |
|
Answer» <P>`{:(P,Q,R,S),(4,3,2,1):}` |
|
| 42. |
P_1:Chloride of A imparts brick red colour to the flame.Select the correct statement P_2:Shape of D is trigonal pyramidal.P_3: E does not decompose on heating.P_4: is used to remove temporary hardness of water... |
|
Answer» `P_2,P_3,P_4` |
|
| 43. |
P-V plots for two gases during an adiabatic process are given in the figure: Plot A and plot B should correspond to : (Assume ideal behaviour) |
|
Answer» He and `O_2` |
|
| 44. |
P-Vplots for three gases (assuming ideal behaviour and similar condition) for reversible adiabatic compression are given in the figure below : Plots X,Y and Z should correspond to respectively : |
|
Answer» `CO_2,Cl_2` and Ne As `gamma` increases, for COMPRESSION graph RISES up. |
|
| 45. |
p-type semi-conductors are made by mixing silicon with impurities of- |
|
Answer» germanium |
|
| 46. |
p-Toluidine overset(Br_(2)-H_(2)O)to A. A in the above sequence is |
|
Answer»

|
|
| 47. |
P+Q rarr Anisol e overset(HI)rarrR+S Correct statement among the following is |
|
Answer» P and Q are `C_(6)H_(5)` Ona and `C_(2)H_(5)` CI |
|
| 48. |
P + Q toAnisoleoverset(HI)(to)R + SCorrect statement among the following is |
|
Answer» <P>P and Q are `C_6H_5ONa` and `C_2H_5Cl` |
|
| 49. |
p pi - d pi bonding is not formed between oxygen and |
|
Answer» <P>Phosphours in `P_(4)O_(10)` |
|
| 50. |
(P), (P) will be : |
|
Answer»
|
|