Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On warming formic acid with ammoniacal silver nitrate the product formed is: |
| Answer» Answer :A | |
| 2. |
On warming an aqueous solution of benzenediazonium chloride, the product obtained is |
|
Answer» Benzene sulphonic ACID |
|
| 3. |
On vigorous oxidation by permanganate solution (CH_(3))_(2)C=CH-CH_(2)CH_(2)CH_(3) gives |
|
Answer» `CH_(3)-overset(OH)overset(|)underset(CH_(3))underset(|)(C)-overset(OH)overset(|)(CH)-CH_(2)CH_(5)` 
|
|
| 4. |
On vigorous oxidation by permangnate solution (CH_(3))_(2)=CHCH_(2)CHO gives |
|
Answer» `(CH_(3))_(2)CO` and `OHC CH_(2)OCHO` |
|
| 5. |
On visit to your native village, you find that lot of smoke, dust and some other gases are coming out of the chimney of a recently set up factory near the village. (i) As a chemistry student, what method you will suggest the factory owner to manage dust and smoke ? (ii) What is the basic principle involved in your suggestion ? (iii) What values are associated with your advice ? |
|
Answer» Solution :(i) Dust and smoke coming out of chimney can be controlled by installing cottrell smoke precipitator. (ii) Cottrell precipitator consists of two metal DISCS charged to a high electrical potential. The charge on colloidal dust and CARBON particles of smoke is neutralised and they get PRECIPITATED while free gases escape out of chimney. (iii) The knowledge of cottrell precipitator and management of dust and CORBON particles of smoke can help us to clean our environment and make it healthier. |
|
| 6. |
On treatment with cold water, an element (A) reacts readily liberating a colourless, odourless gas (B) and a solution (C). Lithium is reacted with (B) yeildng a solid product (D) which effervesce with water to give a strongely basic solution (E). When CO_(2) gas is bubbled through solution (C),a white ppt. (F) is formed but this redissolved forming solution (G) when more CO_(2) is passed. Precipitate (F) effervesced when moistened with conc. HCl and give deep red colouration to Bunsen burner flame. (F) on heating with excess of carbon at 2000^(@)C give (H). Answer the following question on the basis of above passage. Q.Solid (H) on hydrolysis gives a gas which on passing through ammoniated AgNO_(3) solution yields: |
|
Answer» WHITE ppt. |
|
| 7. |
On treatment with cold water, an element (A) reacted quietly, liberating a colourless, odourless gas (B) and a solution (C). Lithium reacted with (B) yielding a solid product (D) which effervesced with water to give a strongly basic solution (F). When CO_(2) (E) was bubbled through solution (C), an initial ppt. (G) was formed but this redissolved forming solution (H) when more CO_(2) was passed. Precipitate (G) effervesced when moistened with conc. HCl and gave a deep red coloration to a Bunsen burner flame. (G) on heating with excess of carbon at 2000^(@)C gave (I)Q(D) reacts with (CHO), B to form a selective reducing agent, which reduces |
|
Answer» Alkenes to alkanes. |
|
| 8. |
on treatment with Con. H_2SO_4, predominately gives ………………………… . |
|
Answer»
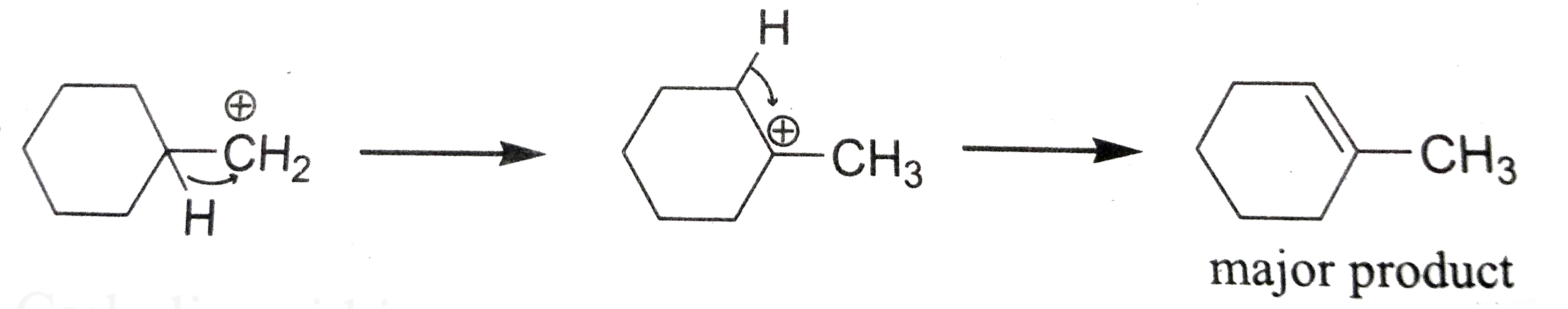
|
|
| 9. |
On treatment with cold water, an element (A) reacted quietly, liberating a colourless, odourless gas (B) and a solution (C). Lithium reacted with (B) yielding a solid product (D) which effervesced with water to give a strongly basic solution (F). When CO_(2) (E) was bubbled through solution (C), an initial ppt. (G) was formed but this redissolved forming solution (H) when more CO_(2) was passed. Precipitate (G) effervesced when moistened with conc. HCl and gave a deep red coloration to a Bunsen burner flame. (G) on heating with excess of carbon at 2000^(@)C gave (I)QSolution (H) contains a salt. Consider the following statements: (0) It causes permanent hardness of water (i) It can not be obtained in solid state (ili) It causes temporary hardness of water (iv) It can be obtained in solid state Select the correct statements, |
|
Answer» (i) and (II). |
|
| 10. |
List two similarity between Lithium and Magnesium. |
| Answer» | |
| 11. |
List two similarity between beryllium and aluminium. |
| Answer» | |
| 12. |
On treatment with cold water, an element (A) reacted quietly, liberating a colourless, odourless gas (B) and a solution (C). Lithium reacted with (B) yielding a solid product (D) which effervesced with water to give a strongly basic solution (F). When CO_(2) (E) was bubbled through solution (C), an initial ppt. (G) was formed but this redissolved forming solution (H) when more CO_(2) was passed. Precipitate (G) effervesced when moistened with conc. HCl and gave a deep red coloration to a Bunsen burner flame. (G) on heating with excess of carbon at 2000^(@)C gave (I)QMetal (A) may be |
|
Answer» Be |
|
| 13. |
On treatment of [Pt(NH_(3))_(4)]^(2+) with concentrated HCI, two compounds (P) and (Q) having the same formula, [Pt(NH_(3))_(2)CL_(2)] are obtained, (P) can be converted into (Q) by boiling with dilute HCI. A soution of (P) reacts with oxalic acid to form [Pt(NH_(3))_(2)(C_(2)O_(4))] whereas (Q) does not react. Point out the correct statement of the following : |
|
Answer» <P>(P) CIS, (Q) trans, both tetrahedral 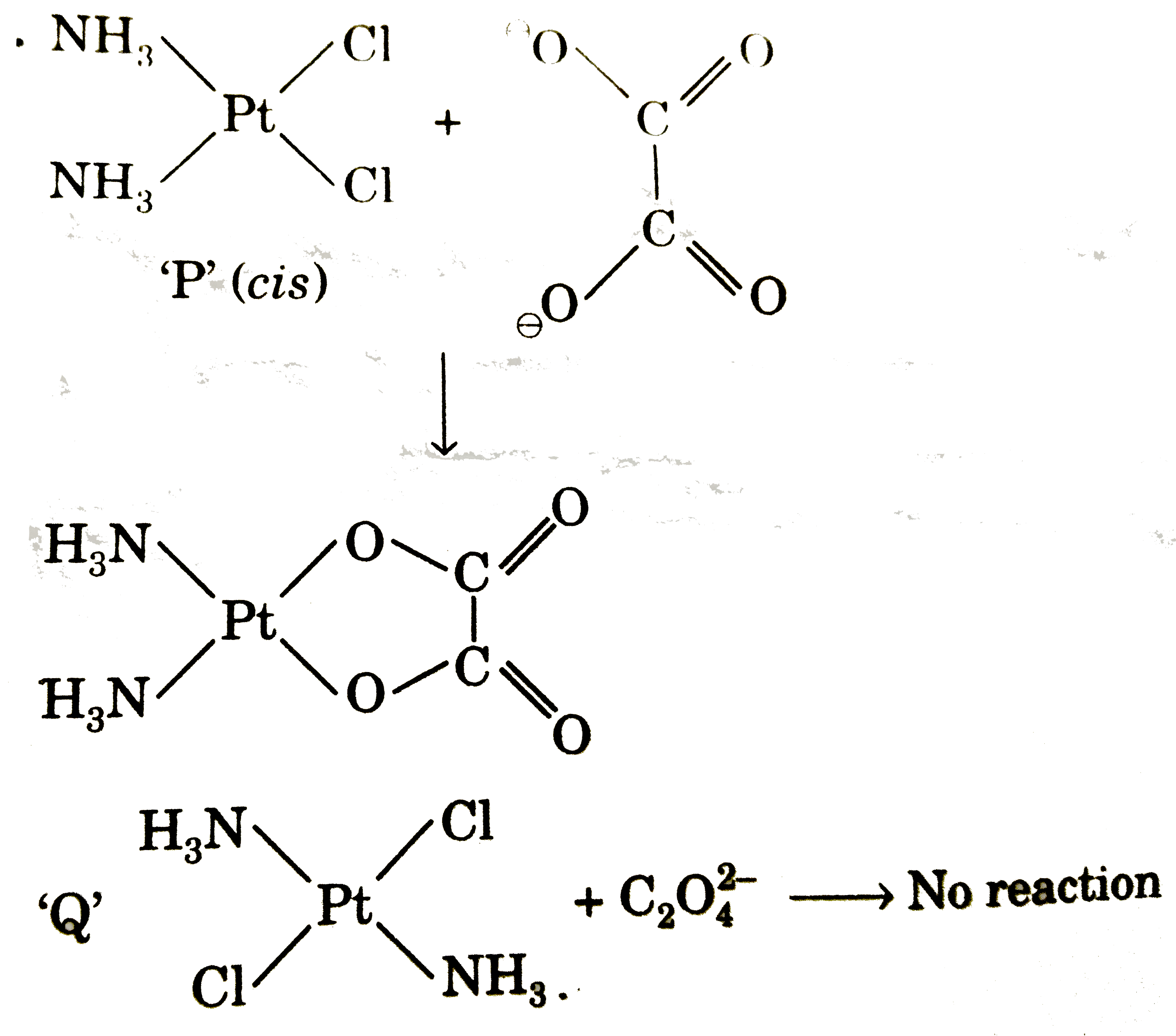 
|
|
| 14. |
On treatmentof [Ni(NH_(3))_(4)]^(+2) with conc.HC/ two compounds I and II having the same formula [Ni(NH_(3))_(2)Cl_(2)] are obtained, I can be converted into II by boiling with dil.HCI. A solution of I reacts with oxalic acid to form [Ni(NH_(3))_(2)(C_(2)O_(4))] where as II does not react. Point out the correct statement from the following |
|
Answer» I CIS, II TRANS, both tetrahedral |
|
| 15. |
On treatment with a clear solution of CrO_(3) in dilute H_(2)SO_(4), an aliphatic alcohol gives a greenish opaque solution within 2-3 seconds. The alcohol can be |
|
Answer» `CH_(3)CH_(2)OH` |
|
| 16. |
On treatment of citric acid with fuming H_2SO_4 which of the following is produced : |
|
Answer» ACETONE |
|
| 17. |
On treatment of 100mL of 0.1 M solution of CoCl_(3).6H_(2)O with excess AgNO_(3),1.2xx10^(22) ions are precipitated. The complex is : |
|
Answer» `[Co(H_(2)O)_(3)Cl_(3).3H_(2)O` |
|
| 18. |
On treatment of 100 mL of 0.1 M solution of CoCl_3.6H_2O with excess AgNO_3, 1.2 xx 10^(22) ions are precipitated. The complex is __________. |
|
Answer» `[CO(H_2O)_4Cl_2]Cl.2H_2O` |
|
| 19. |
On treatment of 100 ml of 0.1 M solution of CoCl_(3).6H_(2)O with axcess AgNO_(3), 1.2xx10^(22) ions are precipitated. The complex is |
|
Answer» `[Co(H_(2)O)_(6)]Cl_(3)` `=100xx0.1` millimole = 10 millimole = 0.01 mole As the IONS produced are precipitated by `AgNO_(3)`, the ions produced must be `Cl^(-)` ions. Thus, 0.01 mole of the complex give `Cl^(-)` ions `=1.2xx10^(22)` `=(1.2xx10^(22))/(6.0xx10^(23))` mole = 0.02 mole `therefore` 1 mole of the complex gives `Cl^(-)` ions = 2 moles (which are precipitated as AgCl) Remembering that COORDINATION number of Co is 6 and TWO `Cl^(-)` ions are present outside the coordination sphere, the formula of the complex will be `[Co(H_(2)O)_(5)Cl]Cl_(2).H_(2)O`. |
|
| 20. |
On treating phenol with chloroform in presence of aqueous sodium hydroxide at 340 followed by acid hydrolysis give the product. Give the name of the reactions. |
| Answer» SOLUTION :Reimer-Tiemann REACTION | |
| 21. |
On treating with dilute acidic solution sucrose gives |
|
Answer» GLUCOSE only |
|
| 22. |
On treating the organic compound with HIO_4 the products are CH_3COCH_3 and HCHO. Hence the compound should be ________. |
|
Answer» `(CH_3)_2underset(OH)UNDERSET(|)C-underset(OH)underset(|)CH-CH_3` |
|
| 23. |
On treating phenol with chloroform in presence of aqueous sodium hydroxide at 340 followed by acid hydrolysis give the product. Write the name of the product |
| Answer» SOLUTION :SALICYLALDEHYDE | |
| 24. |
On treating sodium fusion extract with sodium nitroprusside, a violet colour was observed. This indicates the presence of which element in the organic compound? |
|
Answer» Nitrogen |
|
| 25. |
On treating aniline with nitrous acid and HCl at 0-5^(@)C gives : |
|
Answer» An alcohol 
|
|
| 26. |
On the recently discovered10^(th)planet it has been found that the gases follow the relationship Pe^(V//2) =nCT where Cis constant other notation are as usual( V in lit.,P in atm andTinKelvin) . A curve is plottedbetween P and V at 500 K & 2 moles of gas as shown in figure. Find the slope of the curve plotted between P Vs T for closed container of volume 2 lit. having same moles of gas |
| Answer» Answer :D | |
| 27. |
On the recently discovered10^(th)planet it has been found that the gases follow the relationship Pe^(V//2) =nCT where Cis constant other notation are as usual( V in lit.,P in atm andTinKelvin) . A curve is plottedbetween P and V at 500 K & 2 moles of gas as shown in figure. If a closed container of volume 200 lit.of O_(2) gas ( ideal gas ) at 1 atm& 200K is taken to planet.Find the pressure of oxygen gas at the planet at 821 K in same container |
| Answer» Answer :A | |
| 28. |
On the surface of the earth at 1 atm pressure, a balloon filled with H_2gas occupies500 mL. This volume is 5/6 of its maximum stretching capacity. The balloon is left in air. It starts rising. Calculate the height above which the balloon will burst if the temperature of the atmosphere remains constant and the pressure decreases 1 mm for every 100-cm rise in height. |
| Answer» SOLUTION :126.67 m | |
| 29. |
On the velocity in a reversible reaction, the correct explanation of the effect catalyst is |
|
Answer» It provides a now reaction path of LOW activation energy |
|
| 30. |
On treating a mixture of two alkyl halides with sodium metal in dry ether, 2-methylpropane was obtained. The alkyl halides are |
|
Answer» 2-chloropropane and CHLOROMETHANE 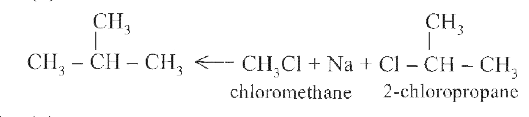
|
|
| 31. |
On the electrolysis for aqueous solution of sodium sulphate , on cathode we get |
| Answer» Solution :Out of `H^(+)` and `Na^(+)` IONS, `H^(+)` ions preferentially migrate at cathode. | |
| 32. |
On the electrolysis of aqueous solution of sodium sulphate, on cathode we get |
|
Answer» Na INSTEAD of `Na^(+) and SO_(4)^(2-)` Cathode: `2H_(2)O+2E^(-)toH_(2)+2OH^(-)` Anode: `H_(2)Oto2H^(+)+(1)/(2)O_(2)+2e^(-)`. |
|
| 34. |
On the basis of Werner's theory explain, why cobalt amine complex, CoCl_(3)4NH_(3) when treated with AgNO_(3) solution precipitates only one Cl^(-) ion even though there are three Cl^(-) ions. |
|
Answer» Solution :The complex, `CoCl_(3)4NH_(3)` with `AgNO_(3)` solution precipitates only one `CL^(-)` ion. `CoCl_(3)4NH_(3)OVERSET(AgNO_(3)"soln")rarrunderset("ppt")(AgCl)` This indicates that, on inonisation the complex GIVES only one `Cl^(-)` Hence according to Werner's theory, out of `3Cl^(-)`, there will be `2CL^(-)` in the coordination sphere as ligands and one `Cl^(-)` in the ionisation sphere or OUTER sphere. Hence formula of the complex must be `[Co(NH_(3))_(4)Cl_(2)]Cl.` `[Co(NH_(3))_(4)Cl_(2)]Clrarr [Co(NH_(3))_(4)Cl_(2)]^(+)+Cl^(-)` `AgNO_(3)+Cl^(-)rarr underset("ppt")(AgCl)+NO_(3)^(-)` |
|
| 35. |
On the basis of VSEPR theory, what geometry is predicted for the central sulphur atom in SOCl_(2)? |
|
Answer» TETRAHEDRAL 
|
|
| 36. |
On the basisof valence bondtheoryexplainthenatureof bondingin [CoF_(6)]^(3-) ion. |
|
Answer» Solution :Thenatureofbondingin `[CoF_(6)]_("ions")^(3-)`: (1) Hexafluorcobalt(III) ions, `[CoF_(6)]^(3-)` is an an anioniccomplex , thestate of cobaltis +3 and thecoordinationnumnber is 6. (2) Electronicconfiguration : `""_(27)Co[Ar]^(18) 3d^(7)4s^(2) 4p^(0)4d^(0)` Electronicconfiguratin `:Co^(3+)[Ar]^(18) 3d^(6) 4s^(0) 4p^(0) 4d^(0)` `Co^(3+)` (Groundstate) 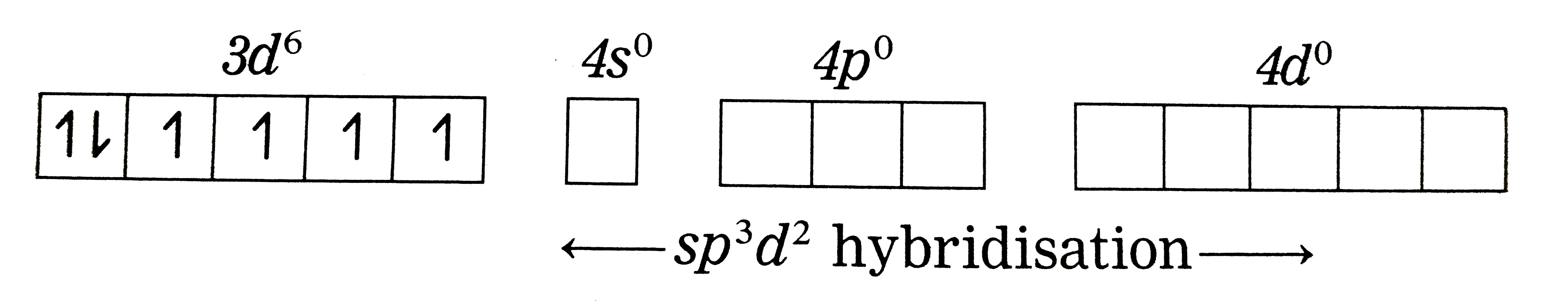 (3) Since`F^(-)` is a weakligand, there is nospin pairingeffect and`Co^(3+)`possesses 4 unpaired ELECTRONS. (3) Since`F^(-)` is a weakligand, there is nospin pairingeffect and`Co^(3+)`possesses 4 unpaired ELECTRONS. (4) Sincethe coordinationnumberis 6,the`Co^(3+)`ion getssix vacantorbitalsby hybridisationof one 4s orbitalsgivingoctahedral geometry. 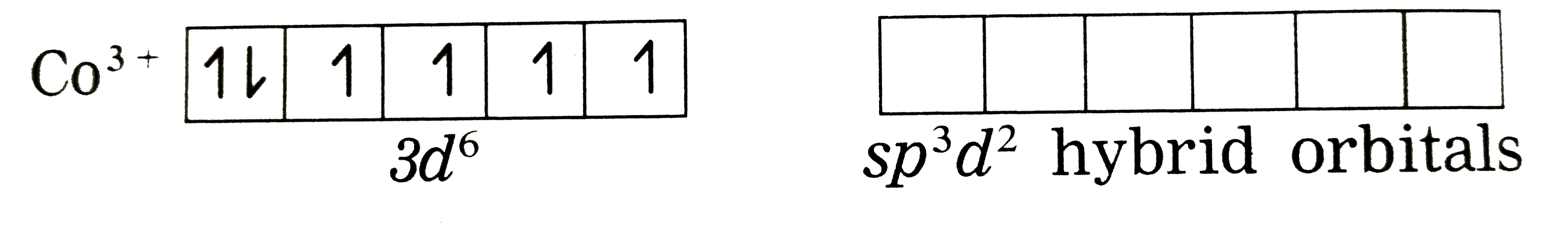 (5)6 lonepairsof electrons FORM `6F^(-)`LIGANDS are accommodated in the sixvacant `sp^(3)d^(2)` hydrib oribitalsof `F^(-)`forming 6 coordinatebondsgivingoctahedral geometryto thecomplex. It isan outercomplex. 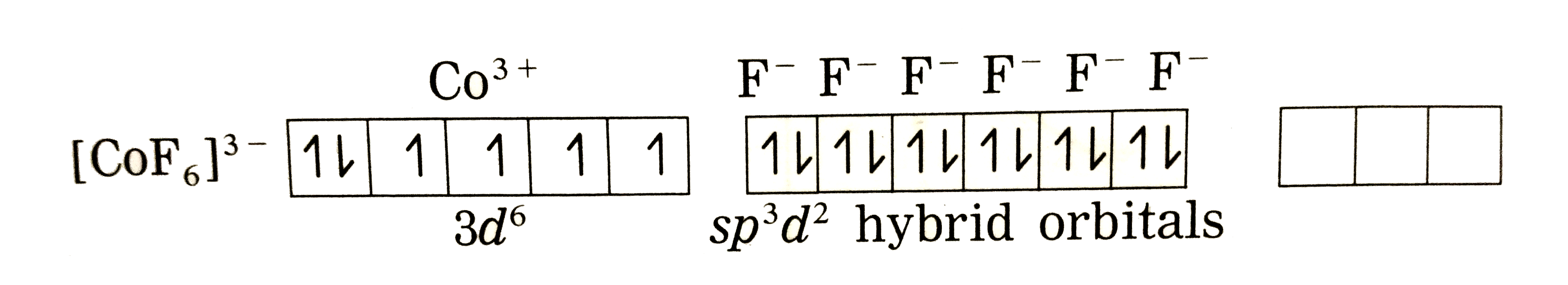 As the complexhas 4 unpariedelectrons it isparamagentic. |
|
| 37. |
On the basis of VB theory explain the nature of bonding in [Co(C_(2)O_(4))_(3)]^(3-). |
|
Answer» Solution :In the complex entity `[Co(C_(2)O_(4))_(3)]^(3-)`, the `Co` is in `+3` oxidation state . The outer electronic configuration of `Co^(3+)` is `3d^(6)` . The oxalato ligand is FAIRLY strong FIELD ligand. So it faces the `3d` electrons in `Co^(3+)` to pair up and make two of the `3d` orbitals available for bonding. As a result, `Co3^(+)` shows `d^(2)sp^(2)` hybridisation. Electronic configuration of `Co` atom Electronic configuration of `Co^(3+)` ion Hybridisation and FORMATION of `[Co(C_(2)O_(4))_(3)]^(-3)` There is no unpaired electron in `[Co(C_(2)O_(4))_(3)]^(3-)`. Thus `[Co(C_(2)O_(4))_(3)]^(3-)` is diamagnetic in nature. During the formation of `[Co(C_(2)O_(4))_(3)]^(3-)`, two of the `3d`-orbitals are used in bonding. Therefore it is an inner ORBITAL (low SPIN)complex. The `[Co(C_(2)O_(4))_(3)]^(3-)` has the octahedral geometry. 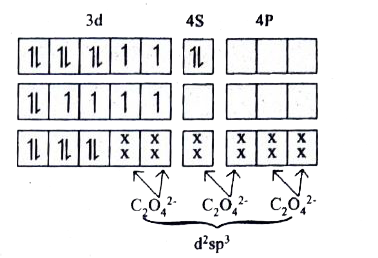
|
|
| 38. |
On the basis of valence bond theory, explain geometry, nature of hybridisation, magnetic property and optical isomerism of [Co(o x)_(3)]^(3-) and [CoF_(6)]^(3-). |
|
Answer» SOLUTION :`[CO(o x)_(3)]^(3-)`: Geometry = octahedral, Hybridisation = `sp^(3)d^(2)`, Magnetic properties = PARAMAGNETIC. Two optical isomers `[CoF_(6)]^(3-)` : Geometry = octahedral, Hybridisation = `sp^(3)d^(2)`, Magnetic properties = paramagnetic. No optical isomerism |
|
| 39. |
On the basis of Valence bond theory account for the hybridization, shape and magnetic property of cuprammonium ion. |
Answer» Solution :`Cu^(2+) ………….. [AR] 3d^94s^0`  Under the influence of approaching `NH_3` ligand an `1e^-` from 3d gets excited to 4P.  The four empty orbitals undergo dsp-hybridization to form four HYBRID orbitals of equal energy. The orbitals of the ligand which CONTAIN lone pair of electrons overlap with hybrid orbitals `Cu^(2+)` ION to form co-ordinate bond. 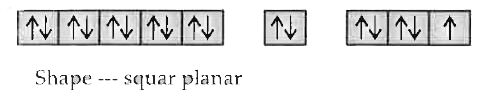 Shape ........ squar planar Or 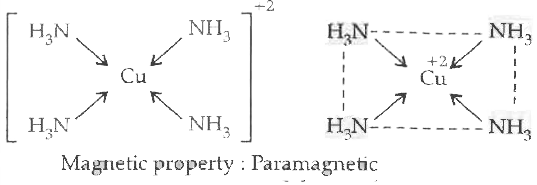
|
|
| 40. |
on the basis of thermochemical equation a,b and c, find out which of the algebraic relationships given in option a , to d . Is correct ? a. C(graphite)+ O_(2)(g) to CO_(2)(g), Delta_(c)H = x kJ mol^(-1) b. C(graphite)+ 1/2O_(2)(g) to CO_(2)(g), Delta_(c)H = y kJ mol^(-1) Co(g) + 1//2 O_(2)(g) to CO_(2)(g), Delta_(c)H = zkJ mol^(-1) |
|
Answer» z=x+y C (graphite) `+1/2O_(2)(g)to CO(g),Delta_(r)H= y KJ mol^(-1).....(ii)` `CO(g)+1/2O_(2)(g)toCO_(2)(g),Delta_(r)H=zkJ mol^(-1) ...... (iii)` ltbr gt `(i)=(ii)+(iii)` x=y+z |
|
| 41. |
On the basis of the standard electrode potential values stated for acid solution, predict whether Ti^(4+) species may be used to oxidise Fe^(II)to Fe^(III) . Reaction{:(E^(@)//V),(Ti^(3+) + e^(-) to Ti^(3+), +0.01),(Fe^(3+) + e^(-) to Fe^(2+), +0.77):} |
|
Answer» Solution : We shall find out the EMF of the cell obtained by the combination of the two. `E_("cell")^(@) = E_("FE^(3+)//Fe^(2+))^(@) - E_(Ti^(4++)//Ti^(3++))^(@) = +0.77 V - 0.01 V = +0.76 V` As the emf of cell is positive therefore, `Ti^(4+)` can be USED to oxidise `Fe^(2+)` to `Fe^(3+)` ion. `Ti^(4+) + Fe^(2+) to Ti^(3+) + Fe^(3+)`. |
|
| 42. |
On the basis of thermochemical equation (a), (b) and (c ), which of the algebric relationship is correct. ( a) C("graphite") + O_(2)(g) rarr CO_(2)(g) , Delta_(r)H = x kJ mol^(-1) (b) C("graphite") + (1)/(2)O_(2)(g) rarr CO(g) , Delta_(r)H = y kJ mol^(-1) ( c) CO(g) + (1)/(2)O_(2)(g) rarr CO_(2)(g) , Delta_(r)H = z kJ mol^(-1) |
| Answer» Answer :C | |
| 43. |
On the basis of the information available from the reaction 4/3 Al + O_(2) rarr 2/3 Al_(2) O_(3). Delta G^(o) = - 827 " kJmol "^(1) " of "O_(2), The minimum emf required to carry out an electrolysis of Al_(2) O_(3) is : |
|
Answer» `8.56` V |
|
| 44. |
On the basis of the following thermochemical data :(DeltafG^(@)H_((aq))^(+)=0)H_(2)O(l)rarrH^(+)(aq)+OH^(-)(aq), DeltaH=57.32 kJH_(2)(g)+(1)/(2)O_(2)(g)rarrH_(2)O(l), DeltaH=-286.20 kJThe value of enthalpy of formation of OH^(-) ion at 25^(@)C is |
|
Answer» `-22.88` kJ `DeltaH_(r)=DeltaH_(f)(H_(2)O,l)-DeltaH_(f)(H_(2),g)-(1)/(2)DeltaH_(f)(O_(2),g)` `-286.20=DeltaH_(f)(H_(2)O(l))` so, `DeltaH_(f)(H_(2)O,l)=-286.20` `H_(2)O(l)rarrH^(+)(AQ)+OH^(-)(aq),DeltaH=57.32 kJ` `DeltaH_(r)=DeltaH_(f)^(@)(H^(+),aq)+DeltaH_(f)^(@)(OH^(-),aq)-DeltaH_(f)^(@)(H_(2)O,l)` `57.32=0+DeltaH_(f)^(@)(OH^(-),aq)-(-286.20)` `DeltaH_(f)^(@)(OH^(-),aq)=57.32-286.20=-228.88 kJ` |
|
| 45. |
On the basis of the given equivalent conductivity lamda_(oo)(NH_(4)Cl)=130 lamda_(oo)(OH^(-))=174 lamda_(oo)(Cl^(-))=66 The value of lamda_(oo)(NH_(4)OH) will be |
|
Answer» 304 `130+174tox+66` `304tox+66` `304-66tox` `x=238` |
|
| 46. |
On the basis of the following thermochemical data: DeltaH_(f)^(@)(H_(aq)^(+))=0, H_(2)O(l)toH^(+)(aq)+OH^(-)(aq)," "DeltaH^(@)=57.32kJ H_(2)(g)+1/2O_(2)(g)toH_(2)O(l)," "DeltaH^(@)=-286.20kJ The value of enthalpy of formation of OH^(-) ions at 25^(@)C is |
|
Answer» `-22.88` KJ |
|
| 47. |
On the basis of the following thermochemical data [(Delta_(f)G^(@)H^(+) (aq) = 0] H_(2)O (l) rarr H^(+) (aq), Delta H = 57.32 kJ H_(2) (g) + (1)/(2)O_(2) (g) rarr H_(2)O (l), Delta H = - 286 kJ The value of enthalpy of formation of OH^(-) at 25^(@)C is |
|
Answer» `- 22.88 kJ` `H_(2)O (L) rarr H^(+) (aq) + OH^(-) (aq), Delta H_(2) = 57.32 kJ` …..(i) `H_(2) (g) + (1)/(2)O_(2)(g) rarr H_(2)O (l), Delta H_(3) = - 286.2 kJ` …. (ii) `Delta H_(1)` will be obtained by adding (i) and (ii) `= 57.32 + (- 286.2) = - 228.88 kJ` |
|
| 48. |
On the basis of the following thermochemical data (Delta f G^(@)H_((aq))^(+)=0) H_(2)O(l) rarr H^(+)(aq)+OH^(-)(aq), DeltaH=57.32 kJ H_(2)(g)+1/2 O_(2)(g) rarr H_(2)O(l), DeltaH=-286.20 kJ The value of enthalpy of formation of OH^(-) ion at 25^(@)C is - |
|
Answer» `-228.88 kJ` `H_(2)(g) +1/2 O_(2)(g) rarr H_(2)O(l), DeltaH=-286.20 KJ""`.....(ii) For the reaction `H_(2)(g)+1/2 O_(2) (g) rarr H^(+)(aq) +OH^(-)(aq)""DeltaH=?` By ADDING equation (i) and (ii) `H_(2)(g)+1/2 O_(2)(g) rarr H^(+) (aq) +OH^(-) (aq)""DeltaH=57.32 kJ -286.20 kJ` `DeltaH=-280.88" kJ/mol"` |
|
| 49. |
On the basis of the following observations made with aqueous solutions, assign secondary valencies to metals in the following compounds : Formula Moles of AgCl precipitated per mole of the compound with excess of AgNO_(3) {:((i),PdCl_(2).4NH_(3),2),((ii),NiCl_(2).6H_(2)O,2),((iii),PtCl_(4).2HCl,0),((iv),CoCl_(3).4NH_(3),1),((v),PtCl_(2).2NH_(3),0):} |
|
Answer» SOLUTION :(i) Formula = `[Pd(NH_(3))_(4)]Cl_(2)`, Secondary valencies = 4 (ii) Formula = `[Ni(H_(2)O)_(6)]Cl_(2)`, Secondary valencies = 6 (iii) Formula = `H_(2)[PtCl_(6)]`, Secondary valencies = 6 (IV) Formula = `[CO(NH_(3))_(4)Cl_(2)]Cl`, Secondary valencies = 6 (V) Formula = `[Pt(NH_(3))_(2)Cl_(2)]`, Secondary valencies = 4 |
|
| 50. |
On the basis of the following thermochemical data (Delta H_(f)^(@))_(H^(+)) = 0 H_(2)O (l) to H^(+) (aq) + OH^(-) (aq) , Delta H = 57.32 kJ H_(2) (g) + (1)/(2) O_(2) (g) to H_(2) O (l) , Delta H= -286.20 kJ The value of enthalpy of formation of OH^(-)ion at 25^(@) C is - |
|
Answer» `-228.88 KJ ` |
|