Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The reaction of WCl_6with Al at about 400^@Cgives black crystals of a compound that contains only tungsten and chlorine. A sample of this compound, when reduced with hydrogen gives 0.2232 g of tungsten metal and hydrogen chloride, which is absorbed in water. Titration of the hydrochloric acid thus produced required 46.2 mL of 0.1051-M NaOH to reach the end point. What is the empirical formula of the black tungsten chloride? |
| Answer» SOLUTION :`WCl_4` | |
| 2. |
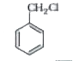
The reaction of toluene with CLl_(2) in presence of FeCl_(3) gives X and reaction in presence of light gives Y. thus, X and Y are: |
|
Answer» X=benzyl CHLORIDE, Y=m-chlorotoluene  THUS, OPTION (d) is CORRECT. |
|
| 3. |
The reaction of water with sodium and potassium is: |
|
Answer» Reversible |
|
| 4. |
The reaction of toulene with Cl_2 in presence of FeCl_3 gives predominently |
|
Answer» <P>m-Chloro TOLUENE |
|
| 5. |
Toluene reacts with Cl_2 in presence of FeCl_3 giving |
|
Answer» <P>m-chlorotoluene |
|
| 6. |
The raction of toluene with CI_(2) in presence of FeCI_(3) gives X and reaction in presence of light gives Y Thus X and Y are . |
|
Answer» <P>X=benzyl CHLORIDE , Y=m-CHLOROTOLUENE 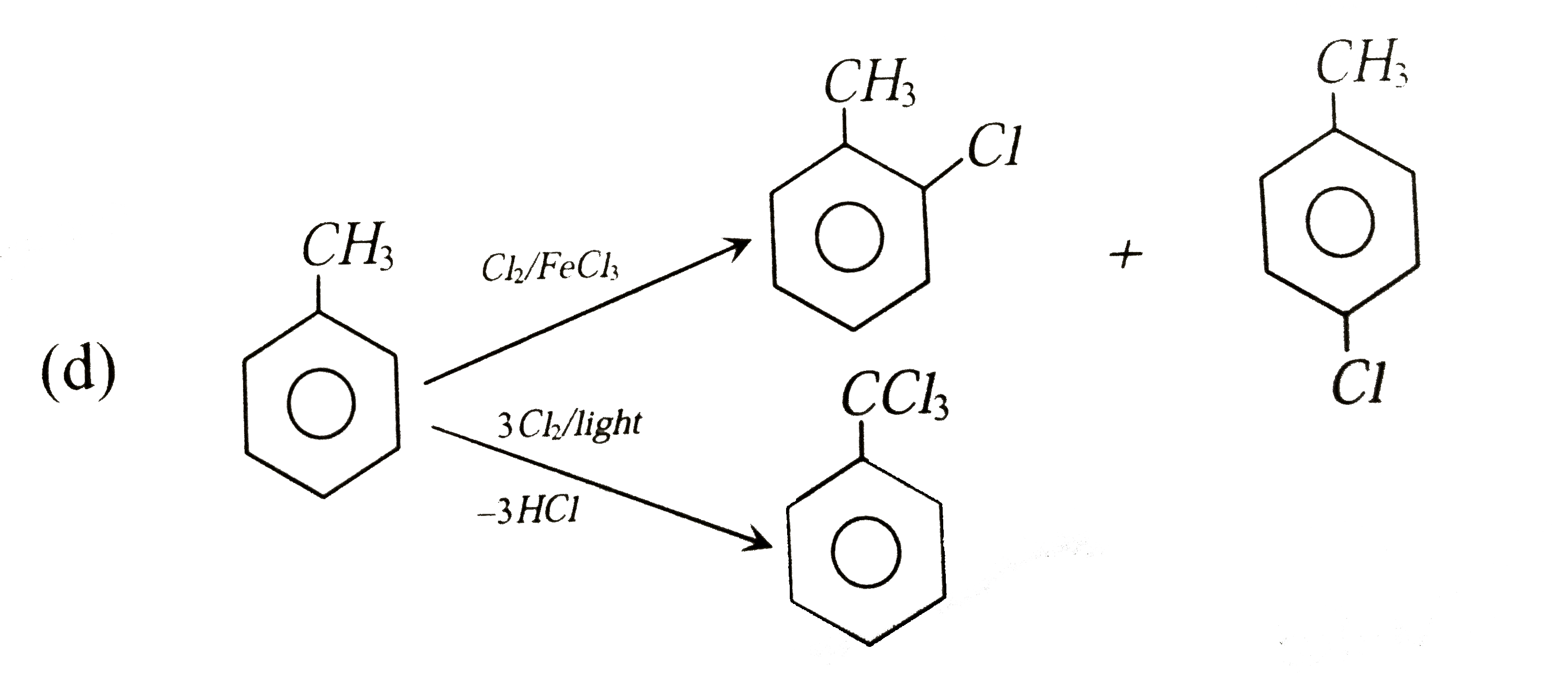
|
|
| 7. |
The reaction of toluene with Cl_(2) in presence ofFeCl_(3) gives predominantly. |
|
Answer» <P>m-CHLOROTOLUENE |
|
| 8. |
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields______ |
| Answer» Solution :CL is o, p-directing, i.e., OPTION (d) is correct. | |
| 9. |
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields …….. . |
Answer» SOLUTION :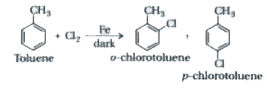
|
|
| 10. |
The reaction of toluene with Cl_(2) in presence of FeCl_(3) gives predominantly : |
|
Answer» BENZYL CHLORIDE 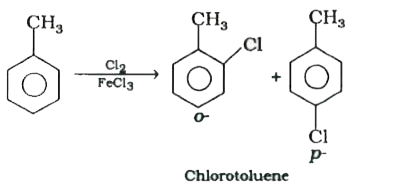
|
|
| 11. |
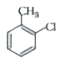
the reaction of toluence with Cl_(2) in presence of FeCl_(3) gives 'X' and reaction in presence of light gives 'Y'. Thus, 'X' and 'Y' are: |
|
Answer» <P>X= Benzal CHLORIDE, Y = o-Chlorotoluene 
|
|
| 12. |
The reaction of toluene with chlorine in the presence of FeCl_(3) gives predominantly |
|
Answer» BENZYL CHLORIDE |
|
| 13. |
The reaction of toluene with chlorine in the presence of ferric chloride gives mainly |
|
Answer» <P>BENZOYL CHLORIDE 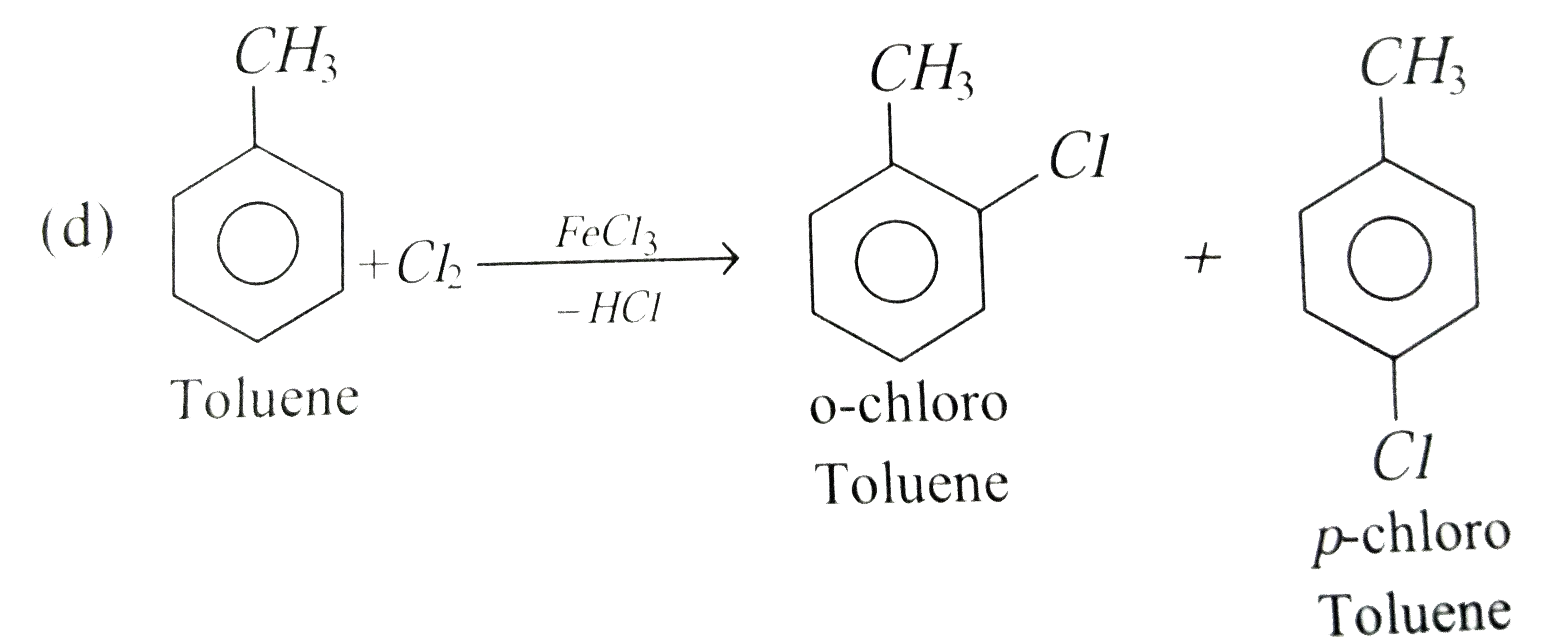
|
|
| 14. |
The reaction oftoluene withchlorine in thepresence of ferricchloride givesprodominantly |
|
Answer» BENZYL CHLORIDE |
|
| 15. |
The reaction of the diene shown below with dry HCl can lead to four products. Provide structural formula of all the products. |
|
Answer» |
|
| 16. |
The reaction of t-Butylbromide with sodium methoxide produces mainly |
|
Answer» Isobutane 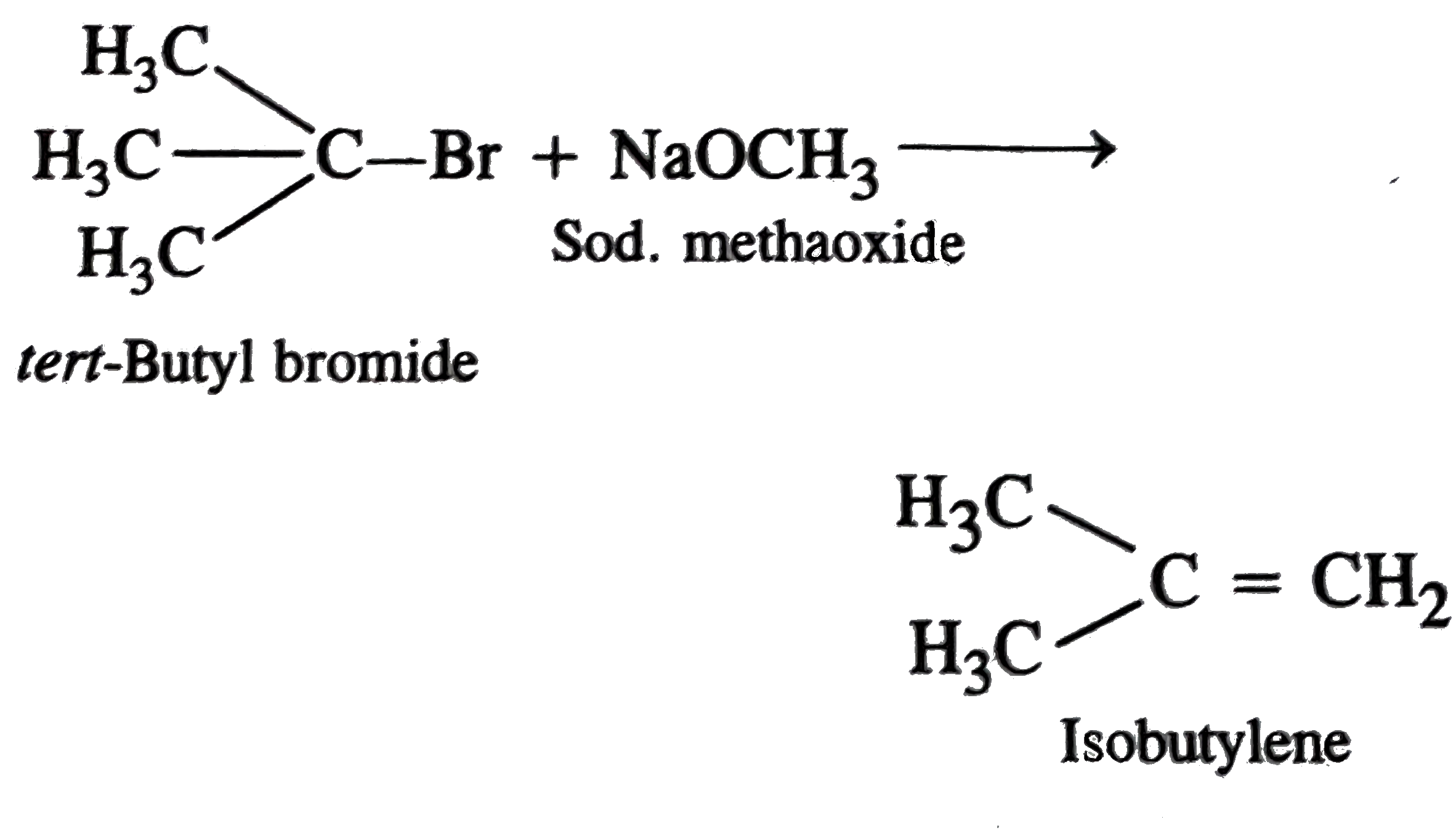
|
|
| 17. |
The reaction of tert, butyl bromide with sodium methaoxide produces mainly : |
|
Answer» Isobutane |
|
| 18. |
The reaction of t-butyl bromide with aq. KOH us represented as (CH_(3))_(3)-C-Brunderset("StepI")to(CH_(3))_(3)C^(delta+).........Br^(delta-)underset("StepII")overset(-Br^(-))to(CH_(3))_(3)C^(+)underset("StepIII")overset(OH^(-))to(CH_(3))_(3)C-OH In above reaction which is the fast step? |
|
Answer» Step -I |
|
| 19. |
The reaction of sulphur powder with phenylmaggnesium bromide followed by hydrolysis gives_____. |
| Answer» Solution :`C_(6)H_(5)SH` (THIOPHENOL) | |
| 20. |
The reaction of t-butyl chloride and sodium ethoxide gies mainly : |
|
Answer» t-butyl ethyl ether `CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-Cl+C_(2)H_(5)barONa^(+)rarrunderset("2-Methylprop-1-ene")(CH_(3)-underset(CH_(3))underset(|)(C)=CH_(2))+CH_(3)CH_(2)OH+NaCl` |
|
| 21. |
The reaction of sunflower oil with NaOH can be called___. |
|
Answer» cracking |
|
| 22. |
The reaction of sodium with water is highly exothermic. The rate of reaction is lowered by: |
|
Answer» LOWERING the temperature |
|
| 23. |
The reaction of sodium methoxide with ethyl bromide follows …………… . |
|
Answer» `SN^1` MECHANISM |
|
| 24. |
The reaction of sodium sulphate with I_(2) gives |
|
Answer» Sodium Iodide |
|
| 25. |
The reaction of sodium thiosulphate with I_2 gives: |
|
Answer» SODIUM sulphide |
|
| 26. |
The reactionof sodiumacetateandsodalimegives |
| Answer» Solution : `UNDERSET ("sodiumacetate")( CH_3COOH )+underset ( "sodalime")( NaOH)to underset ( "methane ") (CH_ 4 )+underset ( "SODIUMCARBONATE")( Na _2CO _ 3) ` | |
| 27. |
The reaction of sodium ethoxide with ethyl iodide to form diethyl ether is termed |
|
Answer» ELECTROPHILIC substitution |
|
| 28. |
The reactionof RMgX with a ketone, followed by treatment with H_(3)O^(+) forms |
|
Answer» `1^(0)` ALCOHOL |
|
| 29. |
Thereactionof RCONH_(2)withamixtureof Br_(2)and KOHgivesR - NH_(2)as a product . Theintermediatesinvolvedin this reactionare . |
| Answer» Answer :A::B | |
| 30. |
The reaction of replacement of a hydrogen atom in benzene by alkyl group can be brought about with the following reagents: |
|
Answer» ALKYL chloride and `AlCl_(3)` |
|
| 31. |
The reaction of R-overset(O)overset(||)(C)-NH_(2) With a mixture of Br_(2) and KOH gives R-NH_(2) as the main product. The intermediates (s) involved in this reaction is/are |
|
Answer»
|
|
| 32. |
The reaction of RCN with RMgX following by hydrolysis gives |
|
Answer» an aldehydes |
|
| 33. |
The reaction of propene with HOCl(Cl_2+H_2O) proceeds through the intermediate |
|
Answer» `CH_3-CH^(+)-CH_2-Cl` |
|
| 34. |
The reaction of propene with HOCl(Cl_2+H_2O) proceedes through the intermediate- |
Answer» SOLUTION :
|
|
| 35. |
The reaction of propene with HOCI proceeds via the addition of: |
|
Answer» `H^+` in the FIRST step |
|
| 36. |
The reaction of primary alkyl halide with nitrile salt produces both RNO_(2) and RONO. Account for this behaviour. |
Answer» Solution : NITRITE ion is an ambident nucleophile `[O^(-)-overset(. .)(N)=O]` tehrefore, it can attack as nucleophile through both oxygen and NITROGEN atoms and LEADS to the formation of two compounds. |
|
| 37. |
The reaction of phenol with air. The product is |
|
Answer» Anthraquinone |
|
| 38. |
The reaction of phenyl methanal and ethanal in the presence of dilute NaOH is known as |
|
Answer» CANNIZARO reaction |
|
| 39. |
The reaction of phenol with a diazonium salt in weakly alkaline medium is called_____. |
| Answer» SOLUTION :COUPLING REACTION | |
| 40. |
The reaction of p-HOC_(6)H_(4)COOH with excess Br_(2) forms |
|
Answer»
|
|
| 41. |
The reaction of p-aminophenol with one mole of acetyl chloride in presence of pyridine gives |
|
Answer»

|
|
| 42. |
The reaction of ozone with oxygen atoms in the presence of chlorine atoms can occur by two step process shown below, O_(3)(g)+Cl(g) to O_(2)(g)+ClO(g)… (i) [K_(i)=5.2times10^(9) L*mol^(-1)*s^(-1) ClO(g)+O_(2)(g) to O_(2)(g)+Cl(g)…. (ii) [K_(ii)=2.6times10^(10)l*mol^(-1)*s^(-1)] The closet rate constant for the overall reaction O_(3)(g)+O(g) to 2O_(2)(g) is: |
|
Answer» `1.4times10^(20)l*mol^(-1)*s^(-1)` Hence overall rate CONSTANT=`K_(i)K_(n)=5.2times10^(9)times2.6times10^(10)=1.4times10^(2)mol^(-1)Ls^(-1)` 
|
|
| 43. |
The reaction of O_(2) and CO with haemoglobin gives |
|
Answer» only OXYGEN - HAEME COMPLEX |
|
| 44. |
The reaction of O_(2) and CO with hemoglobin gives |
|
Answer» only oxygen - HEME complex |
|
| 45. |
The reaction of o-chloroaniline with a mixture of HCl and NaNO_2 followed by cuprous bromide will give |
|
Answer»

|
|
| 46. |
The reaction of nitroprusside anion with sulphide ion gives purple colouration due to the formation of |
|
Answer» the tetranionic complex of IRON (II) coordinating to ONE `NOS^(-)` ION `[overset(+2)(Fe) (C overset(-5)(N))_(5) NOS]^(-4) implies` Tetra-anionic complex of iron (II) coordinating to one `NOS^(-)` ion. |
|
| 47. |
The reaction of nitrogen with hydrogen to make ammonia has Delta H= -92 kJ. N_(2)(g)+3H_(2)(g)rarr2NH_(3)(g) What is the value of Delta U ( in kJ) if the raction of correct out at a constant pressure of 40 bar and the volume changes is 1.25 litre. |
|
Answer» |
|
| 48. |
The reaction of nitrocompounds to primary amines occurs through the intermediate formation of nitroso compound and hydroxylamine catalytic hydrogenation of reduction of nitro compound with an active metal and concentrated hydrochloric acid always gives the corresponding primary amines. In the neutral medium, the nitro compounds are reduced to corresponding hydroxylamines. However, in the basic medium, bimolecular reduction products are obtained which redult through initial condensation of nitroso and hydroxyl amine intermediates followed by reduction to give different products depending upon the nature of the reducing agent used In the reduction of nitrobenzene, which of the following is the intermediate |
|
Answer» `C_(6)H_(5)-N=O` |
|
| 49. |
The reaction of nitrocompounds to primary amines occurs through the intermediate formation of nitroso compound and hydroxylamine catalytic hydrogenation of reduction of nitro compound with an active metal and concentrated hydrochloric acid always gives the corresponding primary amines. In the neutral medium, the nitro compounds are reduced to corresponding hydroxylamines. However, in the basic medium, bimolecular reduction products are obtained which redult through initial condensation of nitroso and hydroxyl amine intermediates followed by reduction to give different products depending upon the nature of the reducing agent used The reagent that reacts with nitromethane to form methyl hydroxylamine is ? |
|
Answer» `Zn//HCl` |
|
| 50. |
The reaction of nitrobenzene with zinc and alkali results in the formation of |
|
Answer» Aniline |
|