Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct order of reactivity towards the electrophilic substitution of the compounds aniline (I) benzene (II) and nitrobenzene (III) is: |
|
Answer» `IgtIIgtIII` |
|
| 2. |
The correct order of reactivity towards electrophilic substitution of the compounds : aniline (I), benzene (II) and nitro benzene (III) is : |
|
Answer» `III GT II gt I` |
|
| 3. |
The correct order of reactivity of the following compounds towards aqueous NaCN will be III. CH_(2) = CH - CH_(2)- Cl III. CH_(3) - overset(Cl)overset(|)(CH) - CH_(3)"" IV. CH_(2) = CH -CH_(2) - Br |
|
Answer» I `GT` II `gt` III `gt` IV |
|
| 4. |
The correct order of reactivity of PhMgBr with underset((I))(Ph-overset(O)overset(||)(C)-Ph), underset((II))(CH_(3)-overset(O)overset(||)(C)-H) and underset((III))(CH_(3)-overset(O)overset(||)(C)-CH_(3)) |
|
Answer» `I gt II gt III` |
|
| 5. |
The correct order of reactivity of the following compounds towards S_N1 reaction is I) CH_3CHX ""II) C_6H_5CH_2X III) (CH_3)_3CX ""IV) (CH_3)_2CHX |
|
Answer» I GT IV gt III gt II |
|
| 6. |
The correctorder of reactivity of hydrogen halides with ethyl alcohol is |
|
Answer» `HF gt HCI gt HBR gt HI` |
|
| 7. |
The correct order of reactivity of >C=O group in given compound is : |
|
Answer»
|
|
| 8. |
The correct order of reactivity of following compounds in nucleophilic substitution reaction is (i) CH_(3)Br(ii)CH_(3)Cl |
|
Answer» `(i) GT (ii) gt (III) gt (IV)` |
|
| 9. |
The correct order of reactivity of alkyl halides: CH_3CH_2Cl, CH_3CHClCH and (CH_3)_3 C Cl towards dehydrohalogenation ? |
|
Answer» ` CH_ 3 CH_2 CL gt CH_3 CHCICH_3 gt (CH_3) _3 CCL` |
|
| 10. |
The correct order of reactivity of aldehydes and ketones towards hydrogen cyanide is |
|
Answer» `CH_(3)COCH_(3) GT CH_(3)CHO gt HCHO` `HCHO gt CH_(3)CHOgt CH_(3)COCH_(3)` |
|
| 11. |
The correct order of reactivity of alcohols with haloacids is ……. |
|
Answer» `1^(@)gt 2^(@)gt 3^(@)` |
|
| 12. |
The correct order of reactivity in S_(N^(1)) reaction for the following compounds is (i). CH_(3)-underset(Cl)underset(|)(C)H-CH_(2)-CH_(3) (ii). Ph-underset(Cl)underset(|)(C)H-CH_(2)-CH_(3) (iii). CH_(3)-underset(OC H_(3))underset(|)overset(Cl)overset(|)(C)-CH_(3) (iv). CH_(3)-CH_(2)-CH_(2)-Cl |
|
Answer» `(i)GT(II)gt(iii)gt(iv)` IIIgtIIgtIgtIV |
|
| 13. |
The correct order of reactivity in S_(N)1 reaction for the following compounds is- underset((I))underset(Cl)underset(|)(CH_(3)-CH-CH_(2)-CH_(3)), underset((II))(Ph-underset(Cl)underset(|)(C)H-CH_(2)-CH_(3)) underset((III))(CH_(3)-underset(OCH_3)underset(|)overset(Cl)overset(|)(C)-CH_(3)) , CH_(3)-CH_(2)-CH_(2)-Cl |
|
Answer» I GT II gt III gtIV 
|
|
| 14. |
The correct order of reactivity of alcohol during dehydration is |
|
Answer» primary `gt` SECONDARY `gt` TERTIARY |
|
| 15. |
The correct order of reactivity is : |
|
Answer» `CH_(3)COClgtCH_(3)COOC_(2)H_(5)gtCH_(3)CONH_(2)` |
|
| 16. |
The correct order of reactivity for the addition reaction of the following carbonyl compounds with ethyl magnesium iodide is |
|
Answer» `I gt III gt II gt IV` 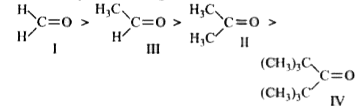
|
|
| 17. |
The correct order of radiii is/are: |
|
Answer» `Pb gt Pb^(2+) gt Pb^(4+)` |
|
| 18. |
The correct order of pseudohalide, polyhalide and interhalogen are |
|
Answer» `BrI_(2)^(-),OCN^(-),IF_(5)` |
|
| 19. |
The correct order of pi-acid nature is : |
|
Answer» `PF_(3)gtCOgtPCl_(3)gtPPh_(3)` |
|
| 20. |
The correct order of Paramagnetic moment is… |
|
Answer» `Cr^(3+)ltMn^(3+)ltFe^(3+)` |
|
| 21. |
The correct order of oxidizing power is .......... |
|
Answer» `CrO_(4)^(2-) GT MnO_(4)^(2-) gt FeO_(4)^(2-)` |
|
| 22. |
The correct order of pK_b value of …...... |
|
Answer» `overlineFgtOoverlineHgtNoverlineH_2gtCoverlineH_3` |
|
| 23. |
The correct order of oxidizing nature of Ocl^(-),Obr^(-) and Ol^(-)ions are............ |
|
Answer» `Ocl^(-) GT Obr^(-) gt OL^(-)` |
|
| 24. |
The correct order of O-O bond length in O_(2),H_(2)O_(2) and O_(3) is |
|
Answer» `O_(2)gtO_(3)gtH_(2)O` 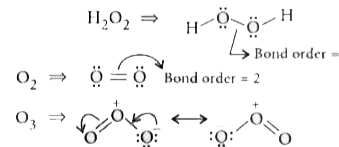 BOND ORDER = 1.5 `:.` Order is `O_(2)gtO_(3)gtH_(2)O_(2)` Bond length is `H_(2)O_(2)gtO_(3)gtO_(2)` |
|
| 25. |
The correct order of oxidising property of Ln ^(4+) ion is |
|
Answer» `DY gt Tb gt Nd gt PR gt Ce ` |
|
| 26. |
The correct order of nucleophilic strength is ……. |
|
Answer» `""^(Θ)OH gt CH_(3)COO^(Θ)gt CH_(3)O^(Θ)gt C_(6)H_(5)O^(Θ)` |
|
| 27. |
The correct order of negative electron gain enthalpy of the elements of oxygen family in the periodic table is : |
|
Answer» `OgtSgtSe` |
|
| 28. |
The correct order of N-O bond lengths in NO, NO_2^- , NO_3^- and N_2O_4 is |
|
Answer» `N_2O_4 gt NO_2^(-) gt NO_3^(-) gt NO` `underset"1.33"(NO_3^-) gt underset"1.5"(NO_2^-)gt underset"1.5"(N_2O_4)gt underset"2.5"(NO)` |
|
| 29. |
The correct order of the mobility of alkali metal ions in aqueous solution is : |
|
Answer» `Na^(+)GTK^(+)gtRb^(+)GTLI^(+)` |
|
| 30. |
The correct order of melting points of group-15 trifluorides is ........... |
|
Answer» `PF_(3) ltAsF_(3) lt SbF_(3) lt BiF_(3)` |
|
| 31. |
The correct order of melting and boiling points of primary (1^(@)), secondary and tertiary (3^(@)) alkyl halides is : |
|
Answer» `1^(@)GT2^(@)gt3^(@)` |
|
| 32. |
The correct order of melting and boiling points of the primary (1^@C) secondary (2^@C) and tertiary (3^@C) alkyl halides: |
|
Answer» <P>`P GT S gt T` |
|
| 33. |
The correct order of melting and boiling points of the primary (1^@)C, secondary (2^@)C and tertiary (3^@)C alkyl halides is : |
|
Answer» <P>`P GT S gt T` |
|
| 34. |
The correct order of magnetic moments (spin values in BM) among is: |
|
Answer» `[Fe(CN)_(6)]^(4-) gt [ClCl_(4)]^(2-) gt [MnCl_(4)]^(2-)` |
|
| 35. |
The correct order of magentic moments (spin only values is B.M ) among is |
|
Answer» `[Fe(CN)_(6)]^(4-) gt [MnCl_4]^(2-) gt [CoCl_(4)]^(2-)` 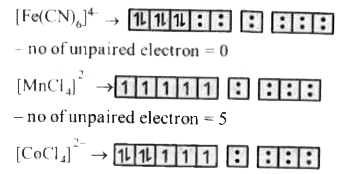 no fo UNPAIRED ELECTRON =3 The greater the number of unpaired electrons, greater the magnitude of magnetic moment. Hence the CORRECT order will be `[MnCl_(4)]^(2-) gt [CoCl_4]^(2-) gt [Fe (CN)_6]^(4-)` |
|
| 36. |
The correct order of magnetic moments (only spin value in BM) among is : |
|
Answer» `[Fe(CN)_(6)]^(4-) gt [CoCl_(4)]^(2-) gt [MnCl_(4)]^(2-)` |
|
| 37. |
The correct order of leaving group tendency is …… |
|
Answer» `I^(-)gt Br^(-)gt CL^(-)gt F^(-)` |
|
| 38. |
The correct order of leaving group ability in a nucleophilic substitution reaction is |
|
Answer» `Br^(-) gt CL^(-)gt CH_3CO_2^(-) gt H^(-)` |
|
| 39. |
The correct order of I^(st) ionisation potential among following elements Be, B, C, N, O is |
|
Answer» B lt Be lt C lt O lt N |
|
| 40. |
The correct order of ionisation energy is : |
|
Answer» CU GT AG gt AU |
|
| 41. |
The correct order of ionic size of N^(3-), Na^(+) , F^(-) ,Mg^(2+) and O^(-) is: |
|
Answer» `Mg^(2+) GT Na^(+) gt F^(-) gt O^(2-) gt N^(3-)` |
|
| 42. |
The correct order of ionisation enthalpy of C, N, O, F is |
|
Answer» `FltNltCltO` `CltNlt OltF` However, N has stable `2P^(3)` electronic conficutation, as a result of which, its ionisation energy is greater than that of oxygen and thus, the correct order is `CltOltNltF`. |
|
| 43. |
The correct order of ionic radius of Yb ^(3+), La ^(3+) , Eu ^(3+), Lu ^(3+) is |
|
Answer» `Yb lt LU lt EU lt LA` |
|
| 44. |
The correct order of ionic radii of Y^(3+) ,La^(3+), Eu^(3+)and Lu^(3+) is |
|
Answer» `Y^(3+) LT LA^(3+) lt EU^(3+) ltLu^(3+)`  Thus, `Lu^(3+) lt Y^(3+) lt Eu^(3+) LTLA^(3+)` |
|
| 45. |
The correct order of ionic radii of Y^(3+), La^(3+), Eu^(3+) and Lu^(3+) is (Atomic nos. Y=39 , La = 57, Eu = 63, Lu = 71) |
|
Answer» `Y^(3+) LT LA^(3+) lt Eu^(3+) lt LU^(3+)` |
|
| 46. |
The correct order of ionic radii of Y^(3+), La^(3+),Eu^(3+) and Lu^(3+) is |
|
Answer» `Y^(3+) LT La^(3+) lt EU^(3+)ltLu^(3+)` |
|
| 47. |
The correct order of ionic radii of Y^(3+), La^(3+), Eu^(3+) and Lu^(3+) is : |
|
Answer» `Y^(3+) lt La^(3+) lt Eu^(3+) lt LU^(3+)` `Y^(3+) lt Lu^(3+) lt Eu^(3+) lt La^(3+)` |
|
| 48. |
The correct order of ionic radii of Y^(3+), La ^(3+), Eu ^(3+) and Lu ^(3+) is |
|
Answer» `Y^(3+)lt Lu ^(3+) lt EU ^(3+) lt La ^(3+)` |
|
| 49. |
The correct order of ionic radii of the ion is |
|
Answer» `LA^(3+) lt EU^(3+)lt Lu^(3+) lt Yb ^(3+)` |
|



