Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The bond between carbon atoms 1 and 2 in the compound : N-=overset(1)(C)-overset(2)(C)H=CH_(2) involves the hybridised carbon as : |
|
Answer» `SP^(2)` and `sp^(2)` |
|
| 2. |
The bond between carbon atom (1) and carbon atom (2) in compound N-=underset(1)(C)-underset(2)(CH)=CH_(2), involves the hybridization as |
|
Answer» `sp^(2)` and `sp^(2)` `'C_(1)'` FORMS TWO `SIGMA`-bonds, hence it is sp-hybridized `C_(2)` forms three `sigma`-bonds, hence, it is `sp^(2)`-hybridized. |
|
| 3. |
The bond between carbon atom (1) and carbon atom (2) in compound N-=C-CH=CH_2 involves the hybrid is: |
|
Answer» `SP^2` and `sp^2` |
|
| 4. |
The bond angles in molecules H_(2)O, NH_(3), CH_(4) and CO_(2) are in the order : |
|
Answer» `H_(2)O gt NH_(3)gt CH_(4)gt CO_(2)` |
|
| 5. |
The bond between atoms of two elements of atomic number 37 and 53 is : |
|
Answer» Covalent |
|
| 6. |
The bond angle O-S-O and hybridisation of sulphur in SO_2 are: |
|
Answer» `119.5^@, sp^3` |
|
| 7. |
The bond angle in XeO_3 molecule decreases from 109^@28^1 to 1030^@ due to |
|
Answer» GREATER repulsions AMONG lone PAIRS |
|
| 10. |
The bond angle in PH_3 is less than bond angle in PF_3 This is due to |
|
Answer» Enhanced replulsions due to the presence of double bond in `PF_3` 
|
|
| 11. |
Bond angle in "PH"_(4)^(" "+) is higher than that in "PH"_(3). Why ? |
| Answer» Solution :In `PH_(4)^(+)`, phosphorous undergo `SP^(3)` hybridization, but in `PH_(3)` phasphorous uses PURE p-oribitals for bonding. Hence BOND angle in `PH_(3)` is nearly `90^(@)`. | |
| 12. |
The bond angle in NF_(3) (102.3^(@)) is similar than NH_(3) (107.2^(@)). This is because of |
|
Answer» large size of F COMPARED to H 
|
|
| 14. |
The bond angle in H_2O (for H-S-H) is |
|
Answer» same as that of Cl-Be-Cl in `BeCl_2` WHEREAS that of 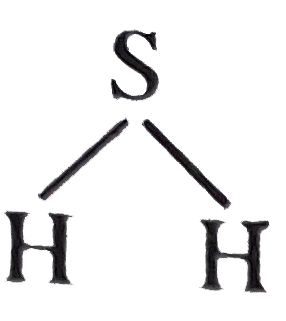 is close to `90^@` |
|
| 18. |
What is the bond angle in the molecule of ammonia ? |
|
Answer» `109^@28'` |
|
| 19. |
What is the bond angle in the molecules of ammonia ? |
| Answer» Answer :D | |
| 20. |
The bond angle C- OH in methanol is ………………….. . |
| Answer» SOLUTION :`108.09^@` | |
| 21. |
The bond angle and % of d- character is SF_(6) are |
|
Answer» `120^(@), 20%` % of d-character `=((2"no. of d-orbital"))/(6("total hybridised ORBITALS"))xx100=33%` BOND ANGLE `=90^(@)` |
|
| 22. |
The bolume of nitrogen gas v_(m) (measured at S.T.P.) required to cover a sample of silica gel with a mono-molecular layer is 129 cm ^(3) g^(-1) of gel. Calculate the surface area per gram of the gel if each nitrogen molecule occupies 16.2xx10^(-20)m^(2). |
|
Answer» Solution :`22400cm^(3)` or `N_(2)` at S.T.P. CONTAIN `=6.022xx10^(23)` molecules `THEREFORE 129 cm ^(3)` of `N_(2)` at S.T.P. will contain `=(6.022xx10^(23)xx129)/(22400)=3.468xx10^(21)` molecules Area occupied by a single molecule `=16.2xx10^(-20)m^(2)` `therefore` Area occupied by `6.468 xx10^(21)` molecules of `N_(2)=(16.2 xx10^(-20))XX(3.468xx10^(21))m^(2)=561.8m^(2)` `therefore` Surface area per gram of gel `=561.8m^(2).` |
|
| 23. |
The bond angle and O-O bond length in O_3 are respectively ________. |
|
Answer» `116.8^(@) and 1.28Å` |
|
| 24. |
The boliling point of 5% solution (W/W) of non-volatile solute in water is 100.45^(@)C. The boiling point of pure water is 100^(@)C.Calculate th molar mas of th solute (K_(b)"for water=0.52 K kg mol"^(-1)). |
|
Answer» `W_(B)=5G, W_(A)=100-5=95g, 0.095 KG, K_(b)=0.52" K kg mol"^(-1)` `M_(B)=(K_(b)xxW_(B))/(DeltaT_(b)xxW_(A))=((0.52K" kg mol"^(-1))XX(5g))/((0.45K)xx(0.095 kg))=60.8" g mol"^(-1)`. |
|
| 25. |
The boling point of benzone is 353.23K. When 1.80 g of a non-valatioe solute is mixed in 90 g of benzene, the boling point id raised to 354.11 Calculate the molar mass of the solute. Given that K_(b) benzene is 2.53 K kg mol^(-1) |
|
Answer» Solution :`M_(B)=(K_(b)xxW_(B))/(DeltaT_(b)xxW_(A))` `W_(B)=1.80 KG,k_(b)=2.53K Kg mol^(-1)` `DeltaT_(b)=(354.11-353.23)=0.88 K` `M_(B)=((1.80)XX(2.53 kg mol^(-1)))/((0.88)xx(0.090 kg))=9.5 g mol^(-1)` |
|
| 26. |
The boiling point of an azeotropic mixture of water and ethyl alcohol is less than that of the theoretical value of water and alcohol mixture. Hence the mixture shows |
|
Answer» that SOLUTION is HIGHLY saturated |
|
| 27. |
The boiling points of thio-ethers are____than those of ether: |
|
Answer» LESSER |
|
| 28. |
The boiling points of the alcohols are higher than the alkanes of corresponding molecular weight because . |
|
Answer» Alcohols can form H-bond(molecular ASSOCIATION) |
|
| 29. |
The boiling points of organohalogen compounds are comparatively ___________ than corresponding hydrocarbons. |
|
Answer» |
|
| 30. |
The boiling points of hydrides of group 16 elements are in the order : |
|
Answer» `H_(2)O GT H_(2)S gt H_(2)TE gt H_(2)Se` |
|
| 31. |
The boiling points of HF, HCI, HBr and HI follow the order |
|
Answer» `HF gt HCI gt HBr gt HI` (1) Stronge H-bonding that increase boiling point. (2) VAN der Waal.s force increase boiling point. Van der WAALS. force increases with increase in atomic radius. (3) Increase in the molecular weight increases boiling point. Factor (2) and (3) give us the following order: `HI > HBr > HCl` Factor (1) makes HF highest boiling point containing. |
|
| 32. |
The boiling points of H_2O, H_2S , H_2Se and H_2Te are in the following order |
|
Answer» `H_2O GT H_2S gt H_2Se gt H_2Te` |
|
| 33. |
The boiling points of halogens increase with increase in molecular weight, it is because: |
|
Answer» As the size increases molecules UNDERGO ASSOCIATION leading to HIGHER stability |
|
| 34. |
The boiling points of amines are____than those of alcohols of comparable molecular mass due to_______ |
| Answer» SOLUTION :LOWER, WEAKER H-bonding. | |
| 35. |
The boiling points for aqueous solutions of sucrose and urea are same at constant temperature. If 3 gm of urea is dissolved in its 1 litre solution, what is the weight of sucrose dissolved in its 1 litre solution ?[Urea - 60 gm / mole, sucrose = 342 gm / mole] |
|
Answer» 3.0 GRAM |
|
| 36. |
The boiling points of acetic anhydride (I), propionic anhydride (II) and butyric anhydride (III) follow the order: |
|
Answer» `I LT II lt III` |
|
| 37. |
The boiling points of aldehydes and ketones are lower than that of the corresponding alcohols and acids. Why? |
| Answer» SOLUTION :ALDEHYDES and ketones have lower boiling point than alcohols and carboxylic ACIDS because they are not associasted with intermolecular H-bonding WHEREAS alcohols and carboxylic acids are ASSOCIATED with intermolecular H-bonding. | |
| 38. |
The boiling points of aldehydes and ketones lie in between alkanes and alcohols of comparable masses because : |
|
Answer» Alkanes are polar |
|
| 39. |
The boiling point of water is exceptionally high because |
|
Answer» there is COVALENT BOND between H and O |
|
| 40. |
The boiling point of water is 100^(@)C and it becomes 100.52^(@)C if 3 g of a non-voltile is dissolved in 20 mL of it. Calculatethe molecular weight of the solute. (K_(b) for water is 0.52 K kg mol^(-1), density of water= 1 g mol^(-1)). |
|
Answer» `W_(B)= 3 g, W_(A)=20 g [20" mL of water= 20 g"], 0.02 kg, k_(b)=0.52" K kg mol"^(-1)` `M_(B)= ?` `M_(B)=(K_(b)xxW_(b))/(DeltaT_(b)xxW_(A))=((0.52" K kg mol"^(-1))xx(3G))/((0.52K)xx(0.02 kg))=150" g mol"^(-1)`. |
|
| 41. |
The boiling point of water ,ethyl alcohol and diethyl ether are 100°C , 78.5°C and 34.6°C respectively. Intermolecular forces will be in order of |
|
Answer» `WATER gt Ethyl ALCOHOL gt Diethyl ether` |
|
| 42. |
The boiling point of water becomes 100.52^(@)C if 1.5 g of a non-volalite solute is dissolved in 100 mL of it. Calculate the molecular weight of the solute. (K_(b) for water=0.6 K kg mol^(-1)). (density of waer-1 g moL^(-1)). |
|
Answer» `W_(B)=1.5g, W_(A)=100g=0.1 kg ""[1" mL of water"~~1G]` `K_(b)=0.6K//m, M_(B)=?` `M_(B)=(K_(b)xxW_(B))/(DeltaT_(b)xxW_(A))=((0.6" K kg MOL"^(-1))xx(1.5g))/((0.52 K)xx(0.1 kg))=17.31" g mol"^(-1)` |
|
| 43. |
The boiling point of water at high altitude is low, because |
|
Answer» the TEMPERATURE is low |
|
| 44. |
The boiling point of water at high altitude is low, because- |
|
Answer» the TEMPERATURE is LOW. |
|
| 45. |
The boiling point of water (100^@ C) becomes, 100.52^@ C if 3 g of non-volatile solute is dissolved is 200 ml of it: Calculate the molecular weight of the solid. (K_b = 0.6 K/m) |
|
Answer» Solution :`DELTA T_b = 100.52 -100=0.52 ^@ C K` ` W_B =3g` `W_A =200 ml` ` M_B=?` ` K_b = 0.6 K//m` ` DeltaT_b =K_b xx(W_b xx 1000 )/( M_B W_A)` ` 0.52 =( 0.6 xx 3xx 1000)/( 200xx 0.52 )` `=( 90 )/( 0.52)` `=173.07//Mol .` |
|
| 46. |
The boiling point of water at 760 mm pressure is 373 K. The vapour pressure of water at 298 K is 23 mm. If the vaporisation enthalpy is 40.656 KJ/mole, then what will be boiling point of water at 23 mm. pressure ? |
|
Answer» 250 K |
|
| 47. |
The boiling point of solution containing 68.4 g of sucrose ("molar mass" = 342 g mol^(-1)) in 100 g of water is (K_(b) "for water" = 0.512 K. kg mol^(-1)) |
|
Answer» `100.02^(@)C` `=0.512 xx 68.4/(342 xx 0.1) = 1.024 ^(@) C ` ` :. T_(b) = T_(b)^(o) + Delta T_(b) =100 ^(@) C + 1.024^(@)C=101.024^(@)C` |
|
| 48. |
The boiling point of p-nitrophenol is hgiher than that of o-nitrophenol because |
|
Answer» `NO_(2)` group at p-position behaves in a DIFFERENT way from that at o- position |
|
| 49. |
The boiling point of methanol is greater than that of Methyl thiol because |
|
Answer» there is intramolecular hydrogen BONDINGIN methanol and intermolecular hydrogen bondingin methyl thiol |
|
| 50. |
The boiling point of n-alkanes is …. Than the branched chain alkanes of the same molecule wieght |
|
Answer» More |
|