Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The boiling and melting points of carboxylic acids depend on : |
| Answer» Answer :A | |
| 2. |
The Bohr orbit radius for the H-atom (Z=1) is approximately 0.53Å. The radius for the first excited state orbit is: |
|
Answer» `0.13Å` For H-atom Z=1 `r prop n^(2)` `(r_(1))/(r_(2))=(n_(1))/(n_(2))` `0.530/(r_(2))=(1^(2))/(2^(2))` `:.r_(2)=0.530xx4=2.12Å` |
|
| 3. |
The blue print process involves the use of |
|
Answer» Indigo dyes |
|
| 4. |
The blue coloured mineral 'Lapis Lazuli' which is used as a semi-precious stone is a mineral of the following class |
|
Answer» SODIUM aluminosilicates |
|
| 5. |
The blue complex ion formad on addition of conc. NH_4OH solution to a Cu^(2+) salt solution is : |
|
Answer» `[CU(NH_4)_2]^(2+)` |
|
| 6. |
The blue coloured mineral 'Lapis Lazuli' used as semi precious stone is : |
|
Answer» PRUSSIAN blue |
|
| 7. |
The blue colour produced on adding hydrogen peroxide to acidified K_2Cr_2O_7 is due to the formation of |
|
Answer» `CrO_5` |
|
| 9. |
The blue colour produced on adding H_2O_2 to acidified K_2Cr_2O_7 is due to the formation of : |
|
Answer» `CrO_5` |
|
| 10. |
The blue colour of the sky is due to ....... |
|
Answer» COAGULATION |
|
| 11. |
The blue colour obtained by the addition of excess dil. NH_(4)OH to an aqueous solution of a salt containing copper is due to the presence of : |
|
Answer» `CuSO_(4)` |
|
| 12. |
The blue colour of liquid ammonia solutions of alkali metals is mainly due to : |
|
Answer» COLOURED cations |
|
| 13. |
The blue colour is obtained when a copper wire is immersed in AgNO_(3) solution, it is due to formation of |
|
Answer» `Ag^(+) ` ions |
|
| 14. |
The blood red colour obtained in the detection of nitrogen and sulphur together in an organic compound in Lassaigne's test is due to |
|
Answer» `[FE(CNS)]^(+)` |
|
| 15. |
The blocks of Mg metal are often strapped to the steel hulls of ocean going ships in order to |
|
Answer» PROVIDE CATHODIC protection |
|
| 16. |
The blood buffers are most often involed in stabilizing the pH in presence of metabolically produced: |
|
Answer» Acids |
|
| 17. |
The bleeding from a wound is stopped by the application of ferric chloride as |
|
Answer» Blood starts FLOWING in the opposite DIRECTION |
|
| 18. |
The blistered appearance of Cu obtained from the reverberatory furnace is due to evolution of |
|
Answer» `CO_(2)` gas |
|
| 19. |
The bleaching action of the Bleaching powder is due to liberation of |
|
Answer» NASCENT CHLORINE |
|
| 20. |
The bleaching action of SO_(2) is temporary but that of Cl_(2) is permanent-why? |
|
Answer» Solution :In the presence of water, `SO_(2)` bleaches by reduction. `SO_(2)+2H_(2)OtoH_(2)SO_(4)+2[H]`(NASCENT) Coloured ORGANIC matter `underset("Reduction")overset([H])to`Bleached organic matter It is not permanent bleaching because the bleached matter regains its original colour if it is kept in air. On the other hand, in the presence of water, `Cl_(2)` bleaches by oxidation. `Cl_(2)+H_(2)Oto2HCl+[O]`(nascent) Coloured organic matter `underset("Oxidation")overset([O])to`Bleached organic matter It is permanent bleaching because if the bleached substance is exposed to air, there is no possibility of its reduction. Thus, its original colour cannot be restored. |
|
| 21. |
Bleaching action of H_2O_2 is due to its ---- character . |
|
Answer» REDUCING properties |
|
| 22. |
The bleaching action of bleaching powder is due to the formation of |
|
Answer» `CaCl_(2)` |
|
| 23. |
The bleaching action ofbleaching powder is due to the formation of: |
|
Answer» `CaCl_2` |
|
| 24. |
The black oxide of copper (CuO) in attacked by moisture and carbon dioxide in air and forms a green film of : |
|
Answer» `Cu(OH)_(2) and C` |
|
| 25. |
The bivalent metal ion having maximum paramagnetic behaviour among the first transition series elements is |
|
Answer» `Mn^(2+)` `Cu^(2+) (3d^9 4s^0) to 1 ` unpaired electron `Sc^(2+) (3d^1 4s^0) to 1 `unpaired electron `Cu^+ (3d^(10) 4s^0 ) to 0 ` unpaired electron HENCE , `Mn^(2+)` has MAXIMUM NUMBER of unpaired THUS, it shows maximum paramagnetic behaviour |
|
| 26. |
The biodegradable polymer is …………. |
|
Answer» Nylon-6,6 |
|
| 27. |
The bite of honey - bee inject into our body |
|
Answer» ACETIC ACID |
|
| 28. |
The bivalent metalion having maximum paramagnetic behavior among the first transition series element is |
|
Answer» `Mn^(2+)` `CU^(2+)(3d^(9)4s^(0))rightarrow1` unpaired electron `Sc^(2)(3d^(9)4S^(0))rightarrow` unpaired electron `Cu^(+)(3d^(10)4S^(0))rightarrow0`unpaired electron Hence , `Mn^(2+)` has MAXIMUM number of unpaired electrons THUS , it shows maximum paramagnetic behaviour |
|
| 29. |
The biodegradable polymer among the following is |
|
Answer» nylon-6 |
|
| 30. |
The biochemical reaction by using urease enzyme is .. |
|
Answer» hydrolysis of urea. |
|
| 31. |
The binding energy of an element is 64 MeV . If BE/nucleon is 6.4 the number of nucleons are : |
| Answer» Solution :`ABE = (B.E)/(A) = (64)/(A) = 6.4` (where A = 10 ) | |
| 32. |
The binding energy of ._(8)O^(16) is 127 MeV. Its binding energy per nucleon is |
|
Answer» 0.794 MeV |
|
| 33. |
The binaphthol (Bnp) is: |
|
Answer» an opticlly ACTIVE compound having chiral centre 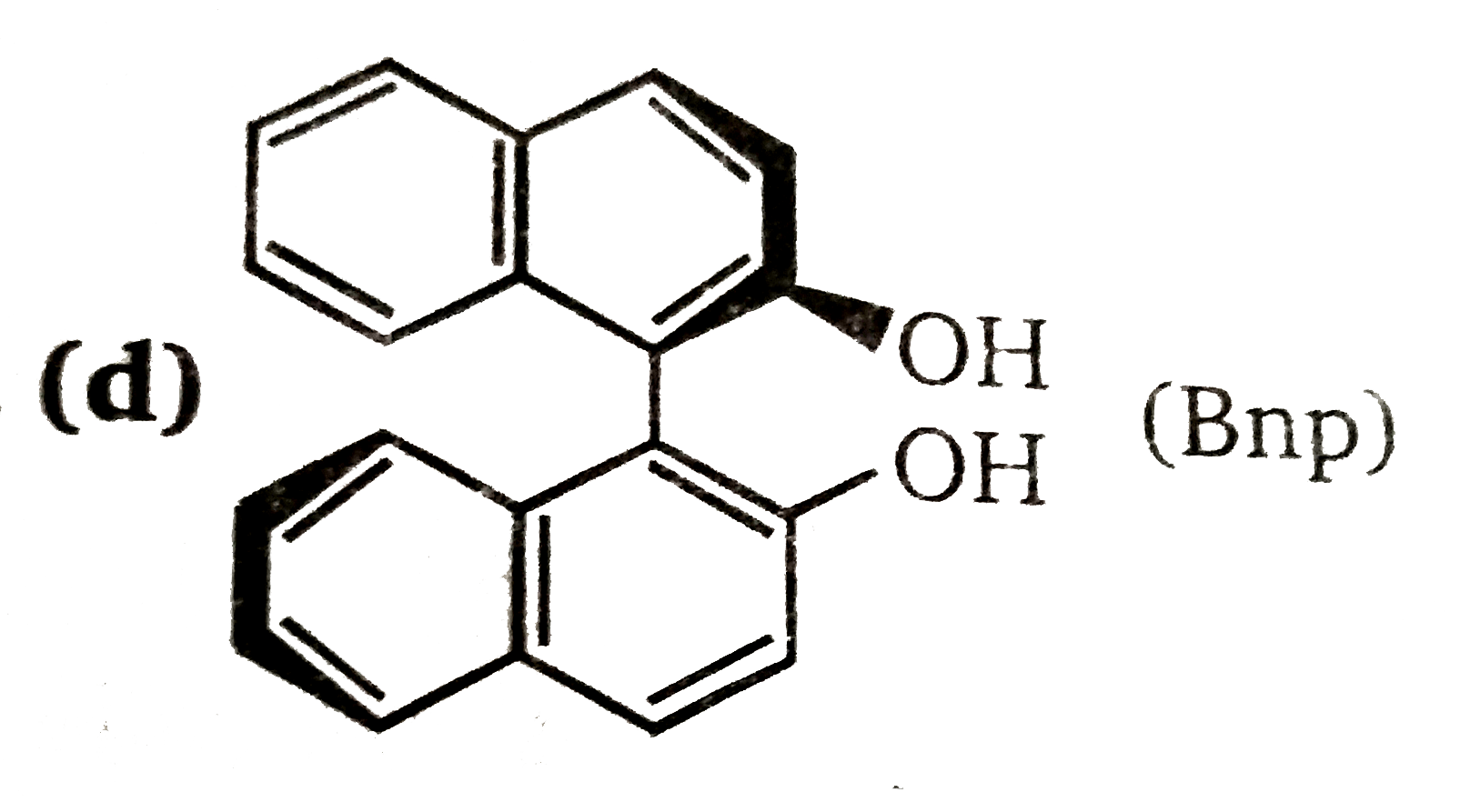 Compound give Attopi isomerism and is optically active due to absence of plane and centre of symmetry. |
|
| 34. |
The bimolecular reaction is represented by : |
|
Answer» <P>(I)(II)(R) 
|
|
| 35. |
The bicabonates of group 2 metals are |
|
Answer» EASILY oxidized by air |
|
| 37. |
The beta- decay of a radioactive element results in formation of its : |
|
Answer» isotope |
|
| 38. |
The beta and alpha glucose have different specific rotations. When either is dissolved in water , their rotation changes until the same fixed value results. This is called |
|
Answer» epimerization |
|
| 39. |
The beta and alpha glucose have different specific rotations. When either is dissolved in water, their rotation changes until the same fixed value results. This is called |
|
Answer» Epimerisation 
|
|
| 40. |
The best yield of product 'X' can be obtained by using which one of the following sequence of reagents and reactants X=CH_3-C-=C-oversetoverset(OH)(|)CH-CH(CH_3)_2 |
|
Answer» `CH_3-C=Choverset(NaNH_2)tooverset((CH_3)_2CHCHO)tooverset(H_3O^+)to` 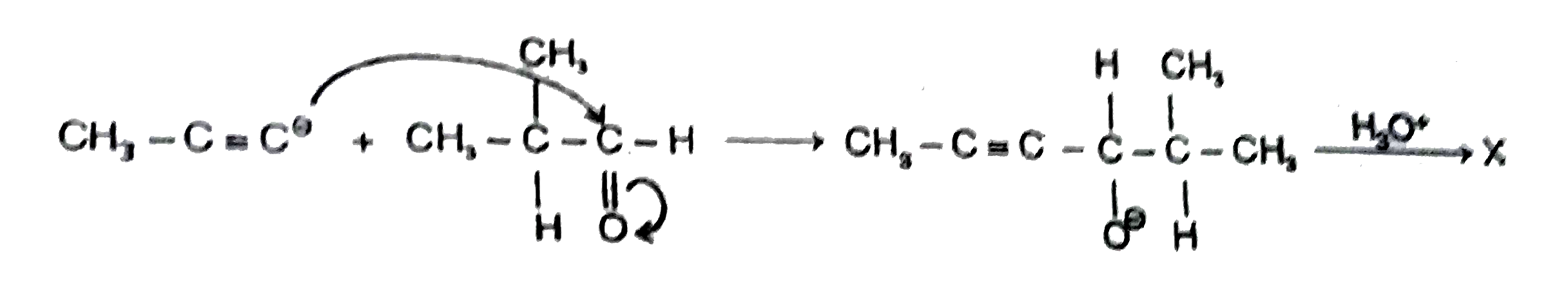
|
|
| 41. |
The beta-decay of""_(11)^(24)Na produces in isotope of |
|
Answer» Al |
|
| 42. |
The best source of vitamin A is |
|
Answer» beans |
|
| 43. |
The best source of vitamin C is |
|
Answer» COD LIVER oil |
|
| 44. |
…...... is the precursor of vitamin-A. |
|
Answer» Beans |
|
| 45. |
Vitamin C is a : |
|
Answer» Beans |
|
| 46. |
The best source of vitamin A is : |
|
Answer» Wheat |
|
| 47. |
The best reagent to convert pent-3-en-2-ol into pent-3-en-2-oneis |
|
Answer» ACIDIC permanganate |
|
| 48. |
The best reagent for the preparation of pure C_2H_5Cl from ethanol is |
|
Answer» LUCAS reagent |
|
| 49. |
The best reagent for converting 2-phenylpropanamide into 2-phenylpropanamine is _____________ . |
|
Answer» excess `H_2` |
|
| 50. |
The best reagent for converting 2-phenylpropanamide into 2-phenylpropanamine is |
|
Answer» excess `H_2` |
|