Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The basicity of phosphorus acid is : |
|
Answer» Two |
|
| 2. |
The basicity of orthophosphoric acid is: |
|
Answer» 2 |
|
| 3. |
The basicity of hydroxides of lanthanides ........... with increase in atomic member due to ............... . |
| Answer» SOLUTION :DECREASES , LANTHANOID CONTRATION. | |
| 4. |
The basicity of HClO_(4) is 'X'. What is the value of 'X' ? |
Answer»  NUrepleacable .H. is .I. NUrepleacable .H. is .I.
|
|
| 5. |
The basicity of aniline is less than that of cyclohexylamine. This is due to |
|
Answer» `+R` - effect of `-NH_(2)` group 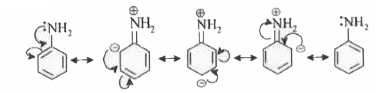
|
|
| 6. |
What is the basicity of H_3PO_2 and why ? |
|
Answer» 1 |
|
| 7. |
The basicity of aromatic (aryl) amines follows the order : |
|
Answer» `3^@gt 2^@ gt 1^@ gt` NH_3` |
|
| 8. |
The basicstruturalunit of silicates is |
|
Answer» `(SiO_(3))^(2-)` |
|
| 9. |
The basic structural unit of silicates is ………… |
|
Answer» `(SiO_(3))^(2-)` |
|
| 10. |
The basic structural unit of silicates is : |
|
Answer» `SiO_(3)^(2-)` |
|
| 11. |
Basic unit fo zeolite, mica, asbestos and feldpar is |
|
Answer» `(--overset(R)overset(|)underset(R)underset(|)Si-O--)_(n)(R=Me)` |
|
| 12. |
The basic repeating structural unit of a crystalline solid is called a……………… |
| Answer» SOLUTION :UNIT CELL | |
| 13. |
The basic strength "…................" as we proceed fromLa(OH)_(3) to Lu(OH)_(3) ( decreases or increases or remains constant). |
|
Answer» |
|
| 14. |
The basic sterength of amine is in the order of…………. |
|
Answer» `NH_3 GT CH_3 NH_2 gt (CH_3)_2NH` |
|
| 15. |
The basic principle of Cottrell 's precipitator is |
|
Answer» neutralisation of charge on colloidal particles |
|
| 16. |
The basic oxide among the following is |
| Answer» Answer :4 | |
| 17. |
Explain the principle behind the Hydrogen bomb. |
|
Answer» NUCLEAR fusion |
|
| 18. |
The basic character of the transition metal monoxides follows the order (AtomicNos.,Ti = 22, V=23, Cr= 24, Fe=26) |
|
Answer» TiOgt VOgtCrOgt Fe |
|
| 19. |
The basic nature of the Lanthanide hydroxides decreases from Ce(OH)_(3), to Lu(OH)_(3)This is due to |
|
Answer» DECREASE in IONIC radius |
|
| 20. |
The basic character of the transition metal oxides follows the order… |
|
Answer» `CRO GT VO gt FEO gt TIO` |
|
| 21. |
The basic character of transition metal monoxides follow the order : |
|
Answer» `VO gt CrO gt TiO gt FeO` |
|
| 22. |
The basic character of the transition metal monoxides follows the order. (Atomic Nos, Ti = 22,V = 23, Cr=24,Fe = 26) |
|
Answer» `Ti O gt VO gt CrO gt FeO ` |
|
| 23. |
The basic character fo the transition metal monoxides follows the order (Atomic no's. Ti = 22, V = 23, Cr = 24 , Fe = 26) |
|
Answer» TiO GT VO gt CrO gt FeO |
|
| 24. |
The basic character of hydrides of the VB group elements decreases in the order: |
|
Answer» `SbH_3 GT PH_3 gt AsH_3 gt NH_3` |
|
| 25. |
The basic character of methylamins in vapour phase is : |
|
Answer» `3^@ GT 2^@ gt 1^@ gt NH_3` |
|
| 26. |
The basic character of the transition metal monoxides follows the order |
|
Answer» `VO gt CRO gt TiO gt FEO` |
|
| 27. |
The basic character of hydrides of the 15th-group elements decreases in the order |
|
Answer» `SbH_(3) gt PH_(3) gt AsH_(3) gt NH_(3)` On MOVING down the group atomic size increases and AVAILABILITY of lone pair decreases. Hence, basic CHARACTER decreases, |
|
| 28. |
The basic character of an amine is due to presence of _________on nitrogen atom. |
| Answer» SOLUTION :A LONE PAIR of ELECTRONS. | |
| 29. |
The basic character of hydrides of nitrogen family............ on moving down the group. |
| Answer» SOLUTION :DECREASE | |
| 30. |
The basic character of amines is due to |
|
Answer» presence of NITROGEN atom |
|
| 31. |
The basic character of amines is due to : |
|
Answer» PRESENCE of nitrogen atom |
|
| 32. |
The basic character of amines is because |
|
Answer» They produce `OH^(-)` ions when treated with water |
|
| 33. |
The basic character of amine can be explained : |
|
Answer» In terms of lewis and Arrhenius CONCEPT |
|
| 34. |
The basic character of amines can be explained |
|
Answer» only in TERMS of Lowry-Bronsted concepT |
|
| 36. |
The bases that are common in both RNA and DNA are |
|
Answer» ADENINE, GUANINE, Thymine |
|
| 37. |
The base present only in RNA and not in DNA is : |
|
Answer» URACIL |
|
| 38. |
The base present in nucleic acids _______ . |
|
Answer» purine base |
|
| 39. |
The base present in DNA, but not in RNA is |
|
Answer» uracil |
|
| 40. |
The base present in DNA but not in RNA is………. . |
|
Answer» GUANINE |
|
| 41. |
The base present in Cytidine : |
|
Answer»
|
|
| 42. |
The base adenine occurs in |
| Answer» Solution :DNA and RNA both. | |
| 44. |
The base pairing occurs in double helix of DNA is |
|
Answer» A to T and G to C |
|
| 45. |
The bapour density of completely dosscisted NH_(4)Cl would be |
|
Answer» Singht LESS then half the of `NH_(4)Cl` `NH_(4)ClhArrNH_(3)+HCl` `becausealpha=therefore"Experimental Molecular WT"=("nor. mol. wt.")/(2)` |
|
| 46. |
The balanced chemical equation for the reaction of potassium permanganate and sulphuric acid with ferrous sulphate to produce ferric sulphate , potassium sulphate , maganese sulphate and water is : |
|
Answer» `2KMnO_(4)+16H_(2)SO_(4)+10FeSO_(4)toK_(2)SO_(4)+2MnSO_(4)+5Fe_(2)(SO_(4))_(3)+16H_(2)O` |
|
| 47. |
The balance chemical equation for the reaction Cu+HNO_(3)toCu(NO_(3))_(2)+NO+H_(2)Oinvolves |
|
Answer» `6HNO_(3)` |
|
| 48. |
The balanced chemical equation for the reaction : MnO_(4)^(-)+AsO_(3)^(3-)+H^(+)toMn^(2+)+AsO_(4)^(3-)+H_(2)Oinvolves |
|
Answer» `4 AsO_(3)^(3-)` |
|
| 49. |
Balance the following equation by oxidation number method.K2 Cr2 O7 +HCl→KCl+CrCl3 +H2 O+Cl2 . |
|
Answer» `K_(2)Cr_(2)O_(7)+8HCL to2CrCl_(3)+2Cl_(2)+2KCl+4H_(2)O` |
|



