Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The approximate composition of soda glass is : |
|
Answer» `SiO_2 75%, Na_2O 15%,CAO 8%,Al_2O_3 2%` |
|
| 2. |
Approximate atomic weight of an element is 26.89. If its equivalent weight is 8.9, the exact atomic weight of element would be |
|
Answer» 26.89 |
|
| 3. |
The appropriate reagent for the following transformation is |
|
Answer» ZN - Hg/HCl 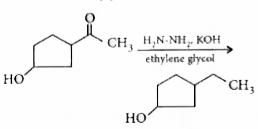 Both Zn- Hg/HCI and `NH_2 NH_2OH^(-)`can REDUCE  group but in case of zn-Hg/HCI , HCI will bring about substitution of `OH^(-)`by `CI^(-)`therefore , the most effective regent is `NH_2NH_2OH^(-)`. group but in case of zn-Hg/HCI , HCI will bring about substitution of `OH^(-)`by `CI^(-)`therefore , the most effective regent is `NH_2NH_2OH^(-)`.
|
|
| 4. |
The appropriate reagent for the following transformation |
|
Answer» `Zn(Hg),HCL` |
|
| 5. |
The appropriate formula ofdichloridobis(ethane-1, 2-diamine)platinum(IV) nitrate is : |
|
Answer» `[PtCl_(2)(en)_(2)](NO_(3))_(2)` |
|
| 6. |
The appreciable radioactivity of uranium minerals is mainly due to |
|
Answer» An uranium isotope of mass NUMBER 235 |
|
| 7. |
The appearance of colour in solid alkali halides is generally due to |
|
Answer» SCHOTTKY defect |
|
| 8. |
The apparatus used for measuring the heat changes of a reaction is called |
| Answer» Solution :barometer | |
| 9. |
The apparatus shown consits of three temperature jacketed 1 litre bulbs connected by stop cocks. Bulb A contains a mixture of H_(2)O(g) , CO_(2)(g)and N_(2)(g) at 27^(@)C and a total pressure of 547.2 mmHg. Bulb Bis empty and is held at a temperature - 23^(@)C. Bulb C is also empty and is held at a temperature of - 173^(@)C. The stopcocks are closed and the volumes of lines connecting the bulbs is zero. Givn : CO_(2)(g) converted into CO_(2)(s) at - 78^(@)C,N_(2)(g)converted into N_(2)(s) at - 196^(@)C &H_(2)O converted into H_(2)O at 0^(@)C. [Use R =0.08 atm-litre // mole.k ] (a) The stopcock between A &B is opened and the system is allowed to come to equilibrium. The pressure in A & B is now228 mmHg.What do bulbs A& B contain? ( b) How many moles of H_(2)O are in system ? ( c ) Both stopcocks are opened and the systemis again allowed to equilibrium.The pressure throughout the system is 45.6 m Hg. What do bulbs A,B and C contain? (d) How many moles of N_(2) are in the system ? |
|
Answer» Bulb `B : N_(2)(g) , CO_(2)(g) , H_(2)O(s)` (b) No. of mole of `H_(2) = 0.0025 ` ( c ) Bult `A : N_(2)(g)` Bulb B `: N_(2)(g) , H_(2)O(s)` Bulb C `: N_(2)(g) , CO_(2)(s)` ( d) TOTAL no of mole of `N_(2)` in system = 0.013 |
|
| 10. |
The apparatus generally used for measuring heat changes is: |
|
Answer» Voltameter |
|
| 11. |
The antisterility or anti reproductive vitamin is: |
|
Answer» B |
|
| 15. |
The antiseptic dettol is a mixture of _______and _______in a suitable solvent |
|
Answer» |
|
| 16. |
The antiseptic character of iodoform is due to: |
|
Answer» Its poisonous NATURE |
|
| 17. |
The antiseptic action of Dettol is due to |
|
Answer» CHLOROBENZENE |
|
| 18. |
The anticodon transfer RNA for the messenger RNA codon GCA is |
|
Answer» TGA |
|
| 19. |
The antibodies necessary to protect new born babies from infection are derived from: |
|
Answer» COW's milk |
|
| 20. |
The antibiotic streptomycin is specific against : |
|
Answer» TUBERCULOSIS |
|
| 21. |
The antibiotic which kills the has microrganism is /are |
|
Answer» oflaxacin |
|
| 22. |
The antibioticwhich is effective againstcertain strains of cancer cells |
|
Answer» DYSIDAZIRINE |
|
| 23. |
The antibiotic Streptomycin is obtained from |
|
Answer» TUBERCULOSIS |
|
| 24. |
The antibiotic having Beta-Lactam ring. |
|
Answer» Pencillin |
|
| 25. |
The anti oxidants for wine are |
|
Answer» `SO_2 & SO_3^(-2)` |
|
| 26. |
The answer to the correct scientific notation of the following given expression frac{2.320}{3.694} X 0.050 is, |
|
Answer» 0.03 |
|
| 27. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : In neutral or faintlyalkaline solution , 8 moles of permanganateanion quantitatively oxidize thiosulphate anios to produce X moles ofsulphur containing product . The magneticof X is |
|
Answer» SOLUTION :`8MnO_(4)^(-) + 3S_(2)O_(3)^(2-) +H_(2)O rarr 8 MnO_(2) + 6SO_(4)^(2-) + 2OH^(-)` `:. `Molesof `SO_(4)^(2-)` formed from 8 moles of `MnO_(4)^(-) = 6` |
|
| 28. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : Consider the following list of reagents : Acidified K_(2)Cr_(2)_(7),alkalineKMnO_(4), CuSO_(4), H_(2)O_(2), Cl_(2),O_(3), FeCl_(2) , HNO_(3) andNa_(2)S_(2)O_(3) . The total number of reagents that can oxidize aqueous iodide to iodine is |
|
Answer» Solution :`6KI+ K_(2)Cr_(2)O_(7) +7H_(2)SO_(4) rarr 4K_(2)SO_(4) + Cr_(2)(SO_(4))_(3)+ 3I_(2) + 7H_(2)O` `2CuSO_(4)+ 4KI rarr Cu_(2)I_(2)+ 2K_(2)SO_(4)+ I_(2)` `H_(2)O_(2) +2KI rarr I_(2)+ 2KOH` `Cl_(2)+ 2KI rarr 2KCl + I_(2)` `O_(3)+ 2KI + H_(2)O rarr 2KOH + I_(2) O_(2)` `2FeCl_(3) + 2KI rarr 2FeCl_(2)+ 2KCl + I_(2)` `4HNO_(3) + 2KI rarr 2KNO_(3) + 2NO_(2)+ I_(2)+ 2H_(2)O` |
|
| 29. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : The oxidation number of Mn in the porduct of alkanline oxidative fusion of MnO_(2) is |
|
Answer» Solution :`2MnO_(2)+ 4KOH + O_(2) RARR 2K_(2)MnO_(4) + 2H_(2)O` OX. Stateof Mn in `OVERSET(+1)(K_(2))overset(x)(M)noverset(-2)(O_(4)): 2+x-8=0` or `x= + 6` |
|
| 30. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : The number of metals collectivelycalled 'platinum metals ' in the transiton elements is |
| Answer» Solution :The ELEMENTS of 2nd and 3RD triads of GROUPS 8,9 and 10 ( i.e., RU , Rh , Pd, OS, Ir, Pt) are collectivelycalledplatinum metals. | |
| 31. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : Th enumber of electrons present in the 4f-subshell ofTerbium( atomic no. = 65) is |
| Answer» SOLUTION :Electronic configuration of TERBIUM (Z= 65) is `[Xe]^(54) 4f^(9) 5d^(0)6S^(2)` | |
| 32. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : The coefficient of H_(2)SO_(4) on balancing the equation K_(2)Cr_(2)O_(7) + H_(2)SO_(4) + KI rarrK_(2)SO_(4) + Cr_(2)(SO_(4))_(3) + H_(2)O+I_(2) is |
|
Answer» Solution :The balanced EQUATION is`:` `K_(2)Cr_(2)O_(7) + 7H_(2)SO_(4) +6KI rarr 4K_(2)SO_(4)+ Cr_(2)(SO_(4))_(3)+ 3I_(2)+ 7H_(2)O` |
|
| 33. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : Oxidation state of Cr in CrO_(5)is |
Answer» SOLUTION :`CrO_(5)` is aperoxo compound , `CrO(O_(2))_(2)` with the structure  `i.e., x+4 (-1)+ ( -2) = 0` or `x= +6` |
|
| 34. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side : The highest oxidationstate shown byany transition element is |
| Answer» Solution :The highest oxidation STATE SHOWN by the TRANSITION element is `+8` viz. Ru in 4DSERIES and Os in 5dseries. | |
| 35. |
The answer to each of the following questions is a single digit integer, ranging from 0 to 9. If the correct answers to the qustion numbers A,B,C and D (say) are 4,0,9 and 2 respectively, then the correct darkening of bubbles should be as shown on the side :The magnetic moment of a transition metal ion is foundto be 5.92 B.M. The number of unpairedelectrons present in the species is |
| Answer» SOLUTION :`MU = SQRT(N(n+2))` B.M.`mu5.92 ` B.M.Only when n=5 | |
| 36. |
The answer to each of the following question is a single digit integer, ranging from 0 to 9. if the correct answers to the question number A,B,C and D (say ) are 4,0,9 and 2 respectively. Then the correct correct darkening of bubbles should be as shownon the side. |
|
Answer» In MG-Al cell, the number of electrons involved in the cell reaction i s |
|
| 37. |
The anomalous boiling point of water is due to |
|
Answer» van derWaals'forces |
|
| 38. |
The anodic solution of standard " Al-Ag" voltaic dilute by 100 times. The potential of the cell is increased by '0.01x' volts what is x ? |
|
Answer» ` in_2 = E^(0) - (0.06)/(3) log ""(((C)/(100)))/([Ag^(+1)]^(3)), DeltaE = (in_2 - in_1) = - 0.02 log [(C)/(100) xx (1)/([Ag^(+)]^(3)) xx ([Ag^(+)]^(3))/(e^(-))]` `=-0.02 log 10^(-2) = - 0.02 xx (-2) log_(10)^(10) = 0.04 = 0.01 X = 0.04 , x =4 ` |
|
| 39. |
The anode mud obtained during electro refining of Cu contains: |
|
Answer» Ag |
|
| 40. |
The anode is a dry cell is |
|
Answer» graphite rod |
|
| 41. |
The anode half-reaction occurring during the discharge of a lead storage battery is |
|
Answer» `Pb_((s))+SO_(2)+O_(2)toPbSO_(4(s))` |
|
| 42. |
The anode and cathode used in Leclanche cell are …………….. respectively. |
|
Answer» Zinc , GRAPHITE rod with `MnO_2` |
|
| 43. |
The anions of A & B respectively are ______ |
|
Answer» Chlorite & HYPOCHLORITE |
|
| 44. |
The anionic sites occupied by unpaired electrons are called F-centres or colour centres . They impart __(X)__ colour to the crystals of NaCl. Excess of lithium makes LiCl crystals __(Y)__and excess of potassium makes KCl crystals _(Z)___. (X),(Y) and (Z) are |
|
Answer» yellow, GREEN and pink respectively |
|
| 45. |
The anion formed by aluminium with alkali is |
|
Answer» Aluminide ion |
|
| 46. |
The anhydride of pyrosulphuric acid is |
|
Answer» `SO_(2)` |
|
| 48. |
The anhydride of orthophosphoric acid is: |
|
Answer» `P_4 O_10` |
|
| 50. |
The anhydride of nitrous acid is: |
|
Answer» `N_2O_3` |
|