Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement-1: Specific conductivity of an electrolytic solution decreases with dilution whereas molar conductivity. Statement-2: Specific conductivity is the conductance of a specific amount of the electrolyte whereas molar conductivity is for 1 mole of the electrolyte. |
|
Answer» Statement-1 is True, statement-2 is true, statement-2 is a correct EXPLANATION of statement-1 |
|
| 2. |
Statement-1: Solubility on n-alcohol in water decreases with increse in molecular weight. Statement-2: The hydropbobic nature of alkyl chain increases. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-7 |
|
| 3. |
STATEMENT-1: Sodium thiosulphate is used in photography and STATEMENT-2: Sodium thiosulphate is photosensitive. |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is a correct explanation for STATEMENT-1. |
|
| 4. |
Statement-1: Sodium chloride is added during electrolysis of fused anhdrous magnesium chloride. Statement-2: Anhydous magnesium chloride is obtained by heating hydrated magnesium chloride, MgCl_(2).6H_(2)O |
|
Answer» Statement -1 is TRUE, Statement-2 are true and Statement-2 is the correct EXPLANATION of Statement -1 |
|
| 5. |
Statement 1: Snow does not melt at monutains easily. Statement 2: A decrease in pressure leads to an increase in freezing point of water. |
|
Answer» Statement 1 is true, STETEMENT 2 is true,statement 2 is a correct explanation for statement 2 |
|
| 6. |
STATEMENT-1 S_(N)2 reaction is carried out in the presence of polar aprotic solents and . STATEMENT-2: Polar aprotic solvent doesnot contain acidic hydrogen |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 |
|
| 7. |
Statement-1. Small quantity of soap is used to prepare a stable emulsion. Statement-2. Soap lowers the interfacial tension between oil and water. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION of Statement-1. |
|
| 8. |
STATEMENT-1 S_(N)1 reaction is comied out in the presence of polar protic solvent and STATEMENT-2 : Polar protic solvent increases stability of carboncation due to the solvation. |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 9. |
Statement 1: Schiff's regent is a dilute solution of rosaniline hydrochloride in water whoce magneta colour is discharged with aqueous SO_(2) or H_(2)SO_(3). Statement 2: Schiff's reagent oxidies benzaldehyde to benzoic acid. |
|
Answer» a.Statement 1 and Statement 2 are true and Statement 2 is the correct explanation of Statement 1. |
|
| 10. |
Statement-1: Silica is added as a flux in reverberatory furnance, in the extraction of copper from copper pyrites. Statement-2: Silica decreases the melting point of the ore and remove the impurity of lead sulphide as PbSiO_(3) |
|
Answer» Statement -1 is TRUE, Statement-2 are true and Statement-2 is the CORRECT explanation of Statement -1 |
|
| 11. |
Statement1: Ring test for nitrates is performed from the Na_(2)CO_(3) extract of the salt solution. Statement 2: All nitrates are soluble in water. |
|
Answer» STATEMENT -1is True, Statement -2 is True , Statement -2 is a correct EXPLANATION for Statement -1. |
|
| 12. |
Statement-1: Sb_(2)S_(3) is not soluble in yellow ammonium sulphide. Statement-2: the common ion effect due to S^(2-) ions reduces the solubility of SbS_(3). |
|
Answer» If both the statement are TRUE and Statement -2 is the correct explanation of Statement-1: |
|
| 13. |
Statement -1 : Salts of ClO_(3)^(-) and ClO_(4)^(-) are well known but those of FO_(3)^(-) and FO_(4)^(-) are unknown. Satement - 2 : F is more electronegative than O while Cl is less electronegative than O. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct explanation for Statement-1. |
|
| 14. |
STATEMENT - 1 : Result of victor Meyer test 1^(@) ROH-Red colour 2^(@) ROH - Blue colour 3^(@) ROH - white or no colour STATEMENT - 2 : Victor Meyer test is a method for separtions of 1^(@),2^(@) and 3^(@) alcohol. |
|
Answer» Statement - 1 is TRUE, Statement - 2 is Ture, Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 15. |
Statement-1 : Rate of nitration is C_(6)H_(6) cong C_(6) D_(6) cong C_(6)T_(6) Statement-2 : Formation of wheland intermediate is rate determining step in nitration of benzene, not the breaking of C–H or C–D bond. |
|
Answer» Statement-1 is TRUE, statement-2 is true and statement-2 is CORRECT explanation for statement-1 |
|
| 16. |
STATEMENT-1 Reaction between sodium tertbutoxide and ethyllodide doesnot produce ether and STATEMENT-2 : Sodium tert butoxide is very strong base but it is not a nucleophile |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 17. |
Statement-1: Reaction of disodium hydrogen phosphate with magnesium sulphate in presence of ammonium hydroxide gives a white crystalline precipitate. Statement-2:Insoluble magnesium ammonium phosphate is formed. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 `MgSO_4 + Na_2HPO_4 + NH_4OH to Mg(NH_4)PO_4 darr + Na_2SO_4+H_2O` |
|
| 18. |
Statement-1: Reaction of t-butyl chloride on Wurtz reaction gives alkene. Statement-2: t-butyl chloride on Wurtz reaction gives alkene. |
|
Answer» If both the statement are TRUE and Statement -2 is the CORRECT EXPLANATION of Statement-1: |
|
| 19. |
STATEMENT - 1 : Rate of reaction with Lucas reagent is C-overset(OH)overset(|)C-CgtC-C-OHgtC-OH STATEMENT - 2 : Lucas reagent reacts with all alcohol byS_(N)1mechanism and rate prop stability of carbocation. |
|
Answer» STATEMENT - 1 is true, Statement - 2 is TURE, Statement-2 is a CORRECT explanation for Statement-1. `"rate"prop(1)/("steric hindrance")` |
|
| 20. |
Statement-1 : rate of dehydration. Statement-2: Rate of dehydration is directly proportional to stability of carbocation |
|
Answer» STATEMENT -I is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 21. |
Statement-1 : Rate of a chemical reaction increases as the temperature is increased. Statement-2 : As the temperature is increased fraction of molecules occupying E_(T) or more increases. |
| Answer» ANSWER :B | |
| 22. |
Statement-1. Rate constant of a zero order reaction has same units as the rate of reaction. Statement-2. Rate constant of a zero order reaction does not depend upon the units of concentration. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-1 is a correct explanation of Statement-1. |
|
| 23. |
Statement-1. Ranitidine is used to treat hyperacidity while brompheniramine is used to treat hypersensitivity. Statement-2. Both the drugs are antihistamines. |
|
Answer» If both ASSERTION and REASON are true, and reason is the true explanation of the assertion. |
|
| 24. |
which is/are statement is correct.Statement-1: R_(2)SiCl_(2) is the starting material for linear polymer of silicone. Statement-2: R_(2)SiCl_(2) is the starting material for 3-D polymer of silicone. Statement-3: R_(3)SiCl is the starging material for 3-D polymerr of silicone. |
|
Answer» |
|
| 25. |
Statement 1: R-CH=CH_(2)overset(C Cl_(3)Br)underset("Peroxide")tooverset(Br)overset(|)(RCH)-CH_(2)C Cl_(3) Statement-2: The addition obey free radical addition on alkenes in presence of peroxide. |
|
Answer» If both the STATEMENT are TRUE and Statement -2 is the correct EXPLANATION of Statement-1: |
|
| 26. |
Statement- 1Pyridiniumchlorochromate canconvertprimaryalcoholsintocorrespondingaldehyde Statement -2MnO_(2)can oxidizebenzylicalcoholintocorrespondingcarboxylicacid Statement- 3 :OsO_(4) canconvert alkeneintotrans1,2- diol |
| Answer» Solution :NA | |
| 27. |
STATEMENT-1: Quenched steel is very hard and brittle. and STATEMENT-2: During quenching, steel is heated to a temperature well below red hot. |
|
Answer» STATEMENT - 1 is TRUE, STATEMENT-2 is True, STATEMENT -2 is correct explanation for STATEMENT-1 |
|
| 28. |
Statement 1 : Proton -proton electrostatic repulsions begin to overcome attractive forces involving protons and neutrons in heavier nuclides Statement 2 : The plot of atomic number (y-axis) veruse number of neutrons (x-axis) for stable nuclei shows a curvature towards x-axis from the line of 45^(@) slope as the atomic number is increased. |
|
Answer» Statement 1 is true, statement 2 is true , statement 2 is a correct explanation for statement 1 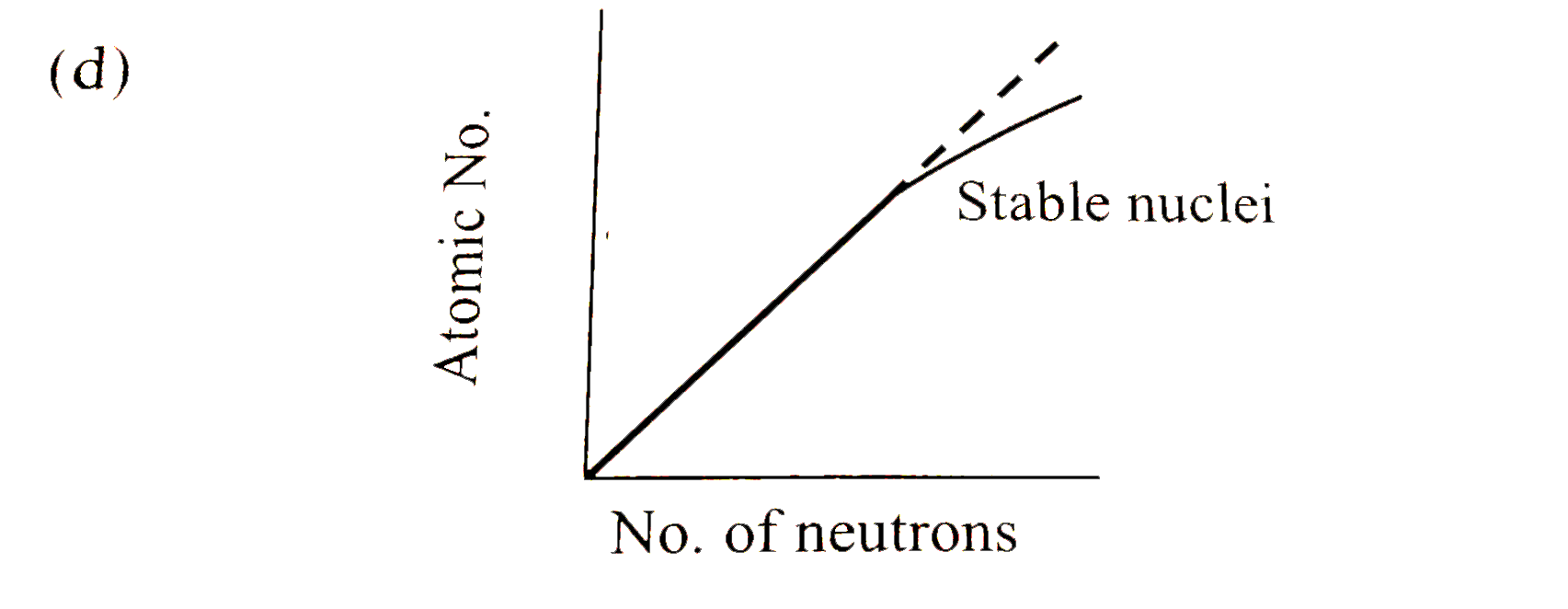 With INCREASE in atomic no. `(Z gt 20)`, no. of NEUTRONS increases more than th eno. of protons. So a curvature is observed as SHOWN. Hence, Statement -1 is correct. The STABILITY of heavier nuclides is due to the fact that attractive FORCES between protons and neutrons overcome the proton-proton electrostatic repulsions. Hence, Statement -2 is false |
|
| 29. |
STATEMENT-1: Pressure exerted by a real gas is lesser than that exerted bý an ideal gas under similar conditions. and STATEMENT-2: At high pressure repulsive forces dominate in real gases. |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is CORRECT EXPLANATION for STATEMENT-1 |
|
| 30. |
STATEMENT - 1 : Propan -2, 2-diol is unstable. STATEMENT - 2 : Repusion between lone pairs of electron of two OH groups makes its unstable. |
|
Answer» Statement - 1 is TRUE, Statement - 2 is TURE, Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 31. |
Statement-1: Potassium ferrocyanide is diamagnetic where as potassium ferricyanide is paramagnetic . Statement-2: Crystal field spletting in ferrocyanide ion is greater than that of ferricyanide ion. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 32. |
Statement-1. Polyester is a copolymer. Statement-2 The repeating structural unit of polyester is derived from two types of monomer units , ethylene glycol and terephthalic acid. |
|
Answer» STATEMENT -1 is True , Statement -2 is True , Statement-2 is a CORRECT explanation for Statement-1. |
|
| 33. |
Statement -1: Potassium can not obtained by the electrolysis of fused KCL.And Statement -2: Potassium has high reactivity at the melting point of KCl. |
|
Answer» Statement -1 is true, Statement -2 is true, Statement -2 is CORRECT EXPLANATION for statement -1. |
|
| 34. |
Statement -1: PMMA is used for making lenses and light covers Statement -2: It has excellent light transmission properties. |
|
Answer» Statement 1 is true, statement-2 is true , statmenent -2 is a correct EXPLANATION for statement -1 |
|
| 35. |
Statement - 1 : Platinum and gold occur in native state in nature. And Statement - 2 : Platinum and gold are noble metals. |
|
Answer» Statement - 1 is TRUE, Statement - 2 is True, Statement - 2 is correct EXPLANATION for statement - 1. |
|
| 36. |
Statement - 1 : Pine oil is fused as a foaming agent in froth floatating process. And Statement - 2 : Adsorption principle is involved in froth floatating process. |
|
Answer» STATEMENT - 1 is TRUE, Statement - 2 is True, Statement - 2 is CORRECT explanation for statement - 1. |
|
| 37. |
Statement 1: Phenol is more reactive than benzene towards electrophilic substitution reaction. Statement 2: In the case of phenol, the intermediate carbocation is more resonance stabilized. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 is a CORRECT explanation for statement 1 |
|
| 38. |
Statement 1: Peracides are stronger acids than corresponding carboxylic acids. Statement 2: The anion of carboxylic acids is stabilized by resonance but not that of peracids. |
|
Answer» Statement-1 is True, statement-2 is true,statement-2 is a CORRECT explanation for statement-1 |
|
| 39. |
STATEMENT-1: Phenol is more reactive than benzene towards electrophilic substitution reaction. and STATEMENT-2: OH group of phenol is electron donating due to resonance. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-6 |
|
| 40. |
Statement 1: Peracids are stronger acids than corresponsing carboxylic acids. Statement 2: The anion of carboxylic acids is stabilized by resonance but not that of peracids. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 is a correct explanation for statement 1 |
|
| 41. |
Statement-1: Pentaamminethiocyanato-N-chromium(III) tetrachloridozincate(II) is a coloured compound and is an example of ionisation isomerism. Statement-2: The compound is paramagnetic and therfore, d-d transition is possible. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct EXPLANATION for Statement-1. 3 unpaired electron in `Cr^(3+)`. |
|
| 42. |
Statement-1 : PbO_(2) is an oxidising agent and reduced to PbO. Statement-2 : Stability of Pb (II) lt Pb (IV)on account of inert pair effect. |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 43. |
Statement 1: Pb^(4+) compounds are stronger oxidizing agents than Sn^(4+) compounds. statement 2: The higher oxidation states for the group 14 elements are more stable for the heavier members of the group due to 'inert pair effect'. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 si a correct EXPLANATION for statement 11 |
|
| 44. |
Statement 1: p-hydroxybenzoic acid has a lower boiling point than o-hydroxybenzoic acid. Statement 2: o-hydroxybenzoic acid has intramolecular hydrogen bonding. |
|
Answer» STATEMENT 1 is true, statement 2 is true, statement 2 is a correct explanation for statement 1 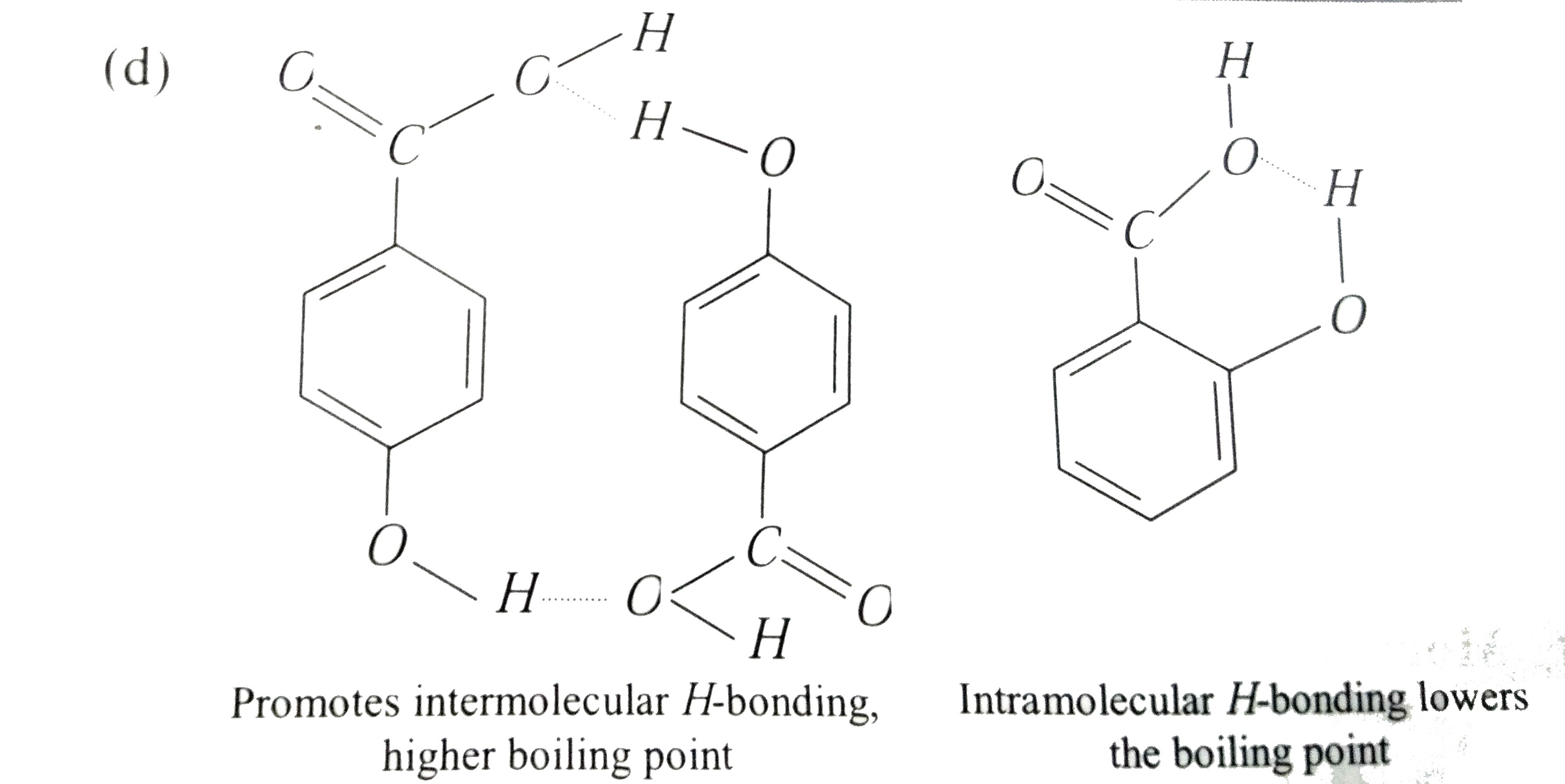 . .
|
|
| 45. |
Statement-1: p-Nitroaniline is more polar than nitrobenzene and Statement-2: Nitro group has -M effect. |
|
Answer» |
|
| 46. |
Statement-1 : Ortho boric acid crystals are hard and cannot be broken easily into the powder form. Statement-2 : In the solid state B(OH)_(3) units are hydrogen bonded together into two dimensional sheets. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 47. |
STATEMENT-1 : Out of all actinoids , Th has highest melting point . STATEMENT-2: Th has largest size among actinoids . |
|
Answer» Statement-1 is TRUE , Statement-2 is True , Statement-2 is a correct explanation for Statement-1 |
|
| 48. |
STATEMENT-1 : Order of reaction can never be fractional for an elementary reaction. and STATEMENT-2 : Elementary reactions take place by one step mechanism |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is CORRECT EXPLANATION for STATEMENT-1 |
|
| 49. |
Statement-1: Optically active 2-iodobutane on treatment with NaI in acetone undergoes racemization. Statement-2. Reaction involves repeated Walden inversion. |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION of statement-1. |
|
| 50. |
Statement -1: Only one Aldehyde 'X' responds positively with all the tests of carbonyl compunds like Tollen's tests, Fehling test, 2,4,DNP test, as well as iodoform test. Statement-2 All aldehydes respnds all the forur test given in assertion. |
|
Answer» Statement -1 is True,Statement -2 is True, Statement -2 is a correct explanation for statement -1. REASON: False, all aldehydes to not give iodoform test. |
|