Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Silver chloride dissolves in excess of ammonium hydroxide solution. The cation present in the resulting solution is |
|
Answer» `[Ag(NH_(3))_(6)]^(+)` Ag exhibits a PRIMARY valency of 1. So, tentative secondary valency is 2. |
|
| 2. |
Silver chloride dissolves in a solution of ammonia but not in water because : |
|
Answer» Ammonia is a better solvent than WATER |
|
| 3. |
Silver can exhibit more than one oxidation states under a rare condition, e.g. when it combines with fluorine. Which of the following oxidation states of silver are possible when it comes with fluorine? |
|
Answer» ZERO |
|
| 4. |
Silver can be precipitated in hydrometallurgy by usingaluminium. Thisis because |
|
Answer» silver is more ELECTROPOSITIVE METAL |
|
| 5. |
Silver bromide reactswith hypo solution to give : |
|
Answer» `NA[AgS_2O_3]` |
|
| 6. |
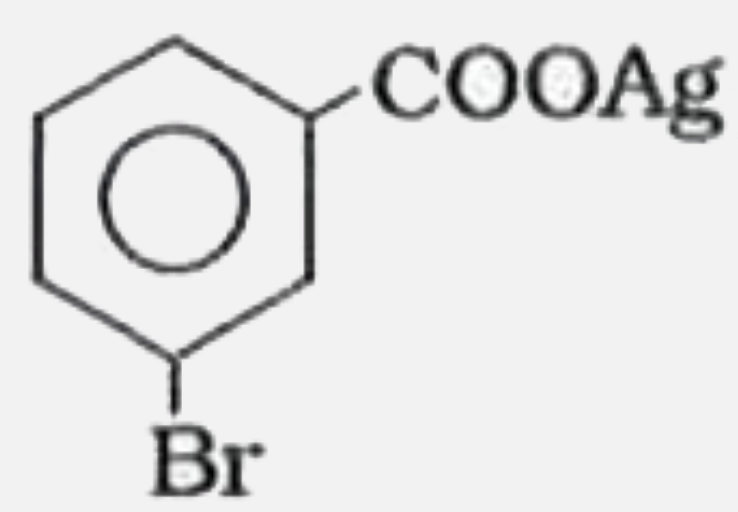
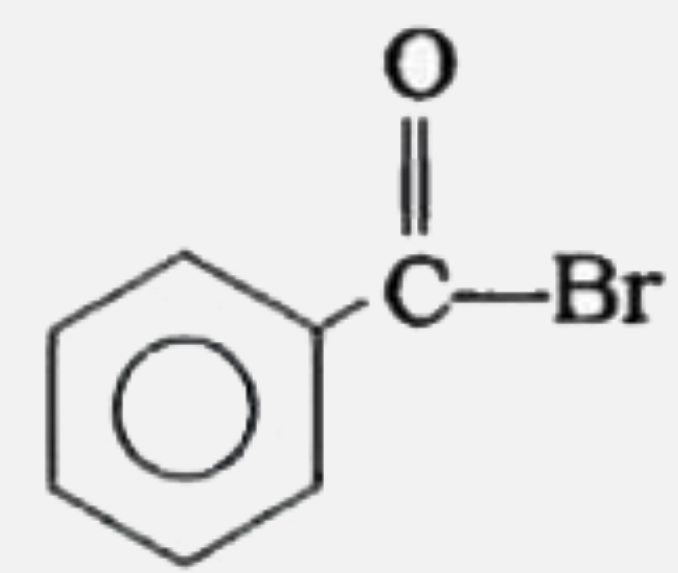
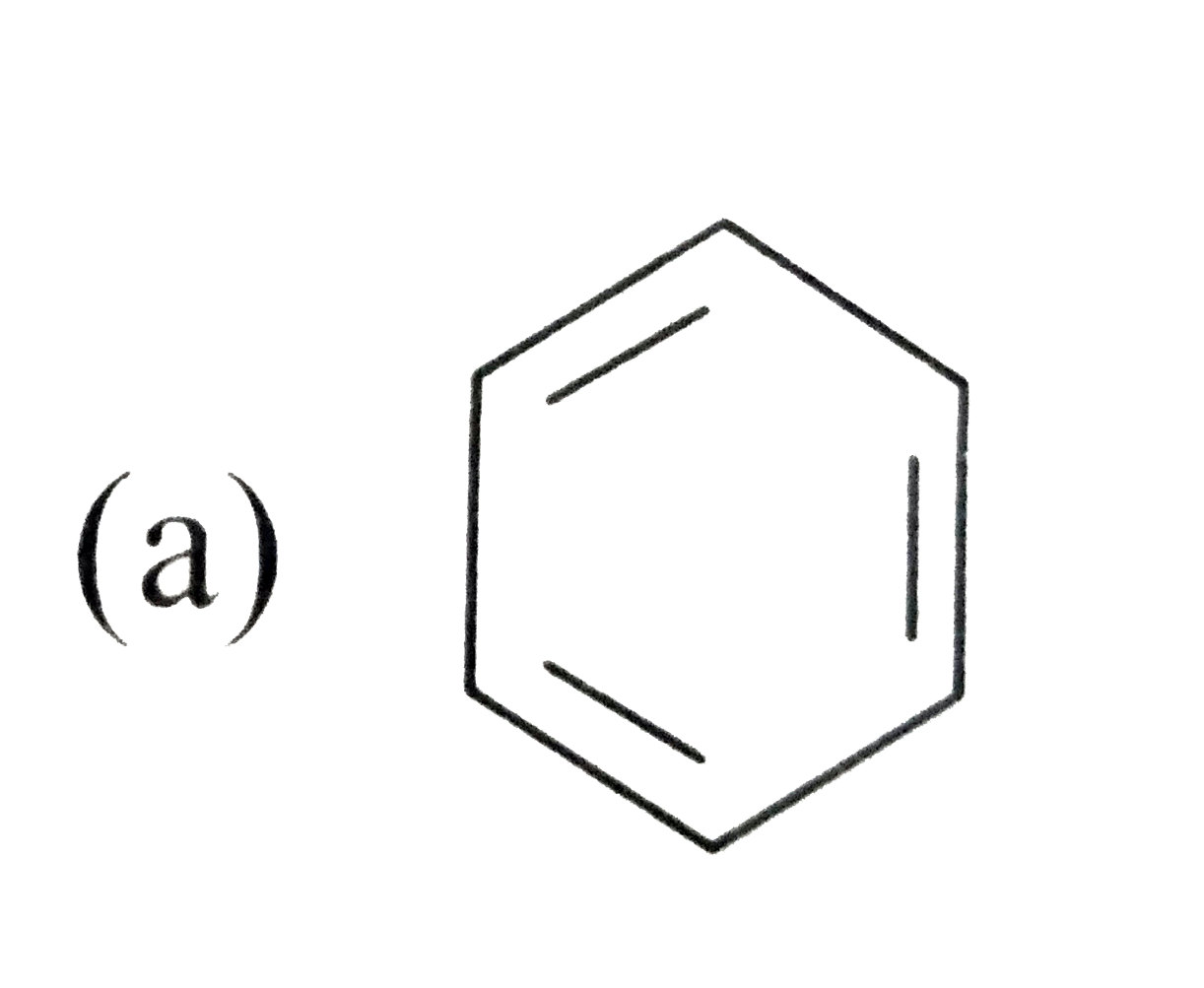
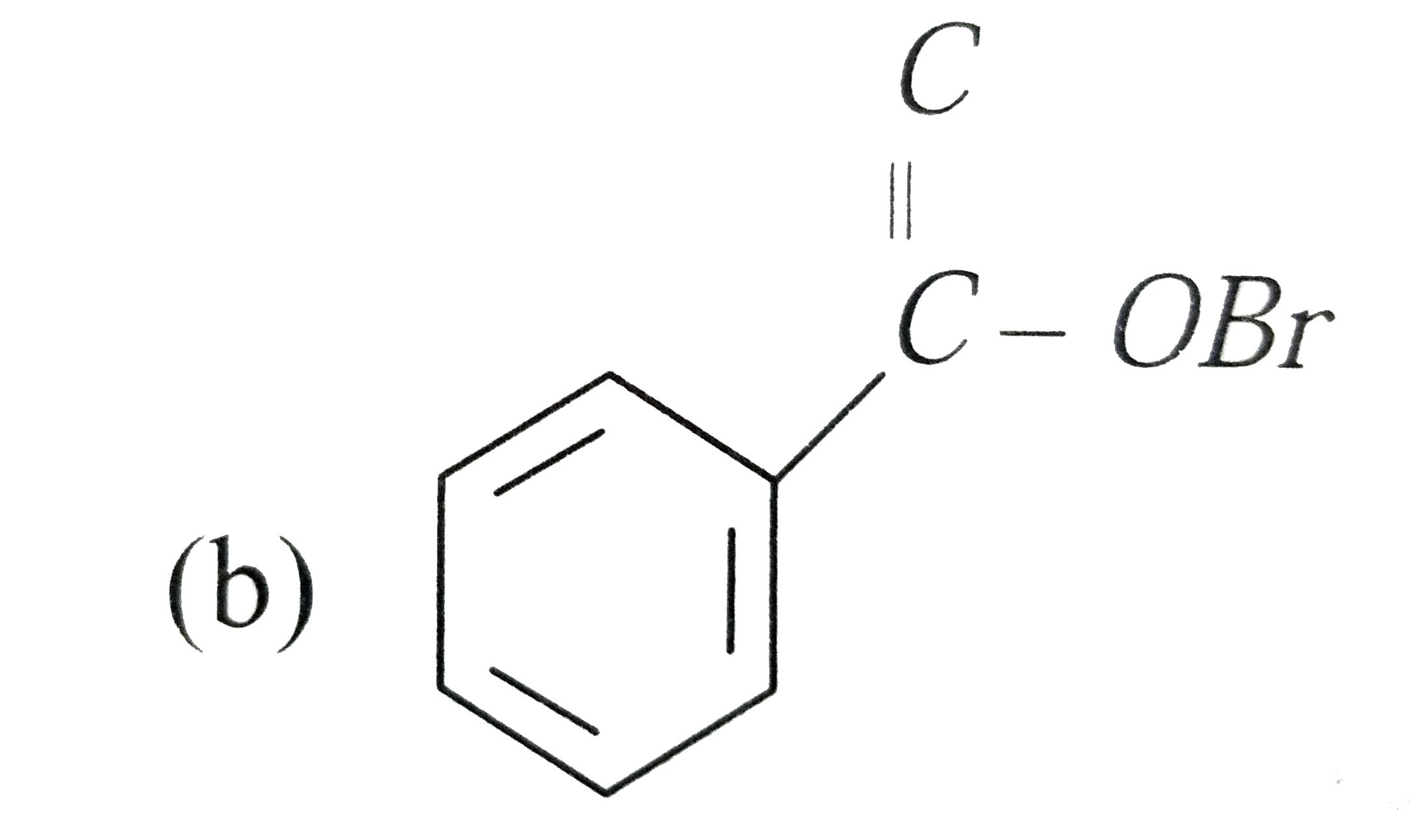
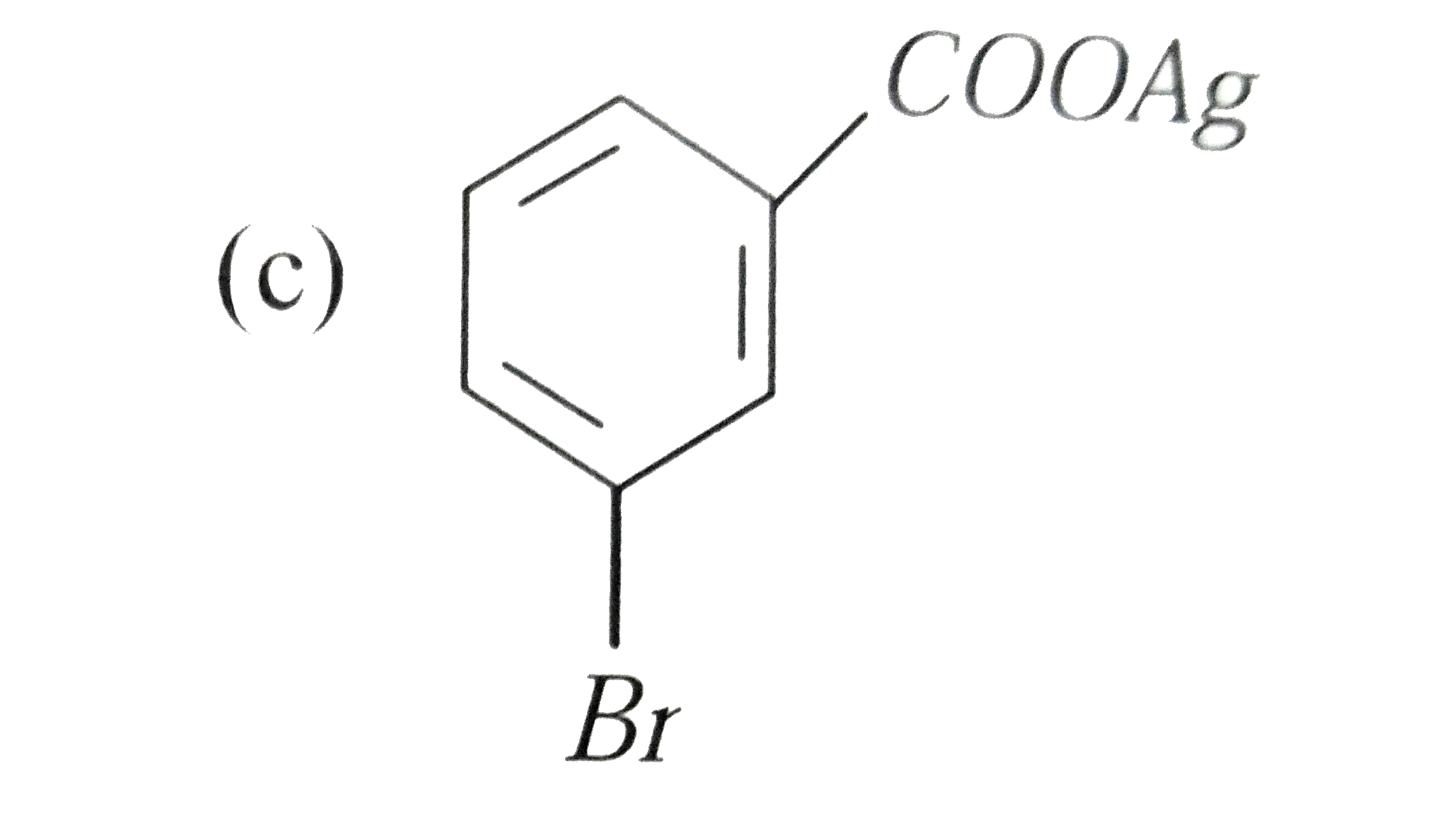
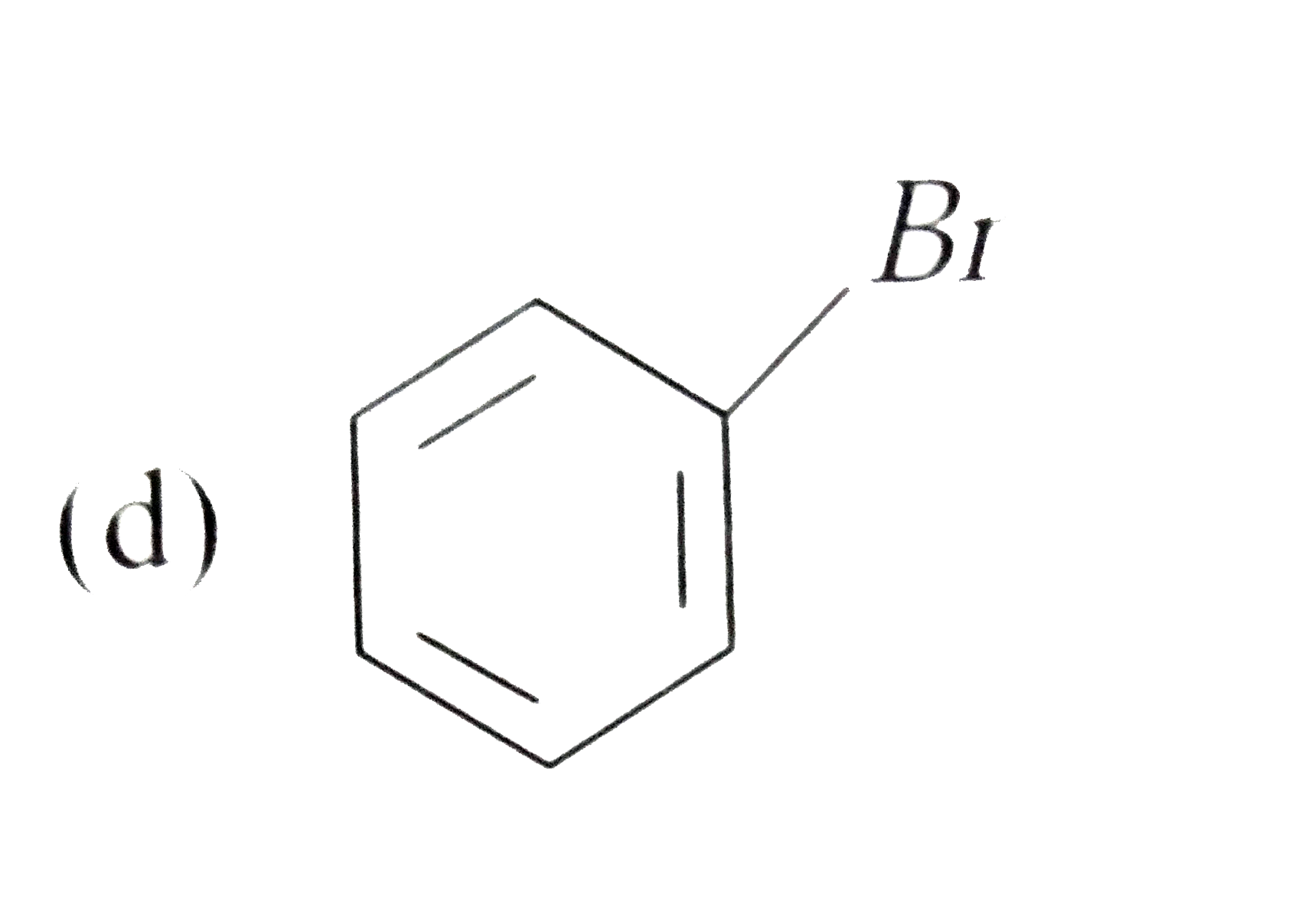
Silver benzoate will react with bromine in acetone to form : |
|
Answer»

|
|
| 7. |
Silver benzoate reacts with bromine to form |
|
Answer»
|
|
| 8. |
Silver benzoate reacts with bromine to form:- |
|
Answer»
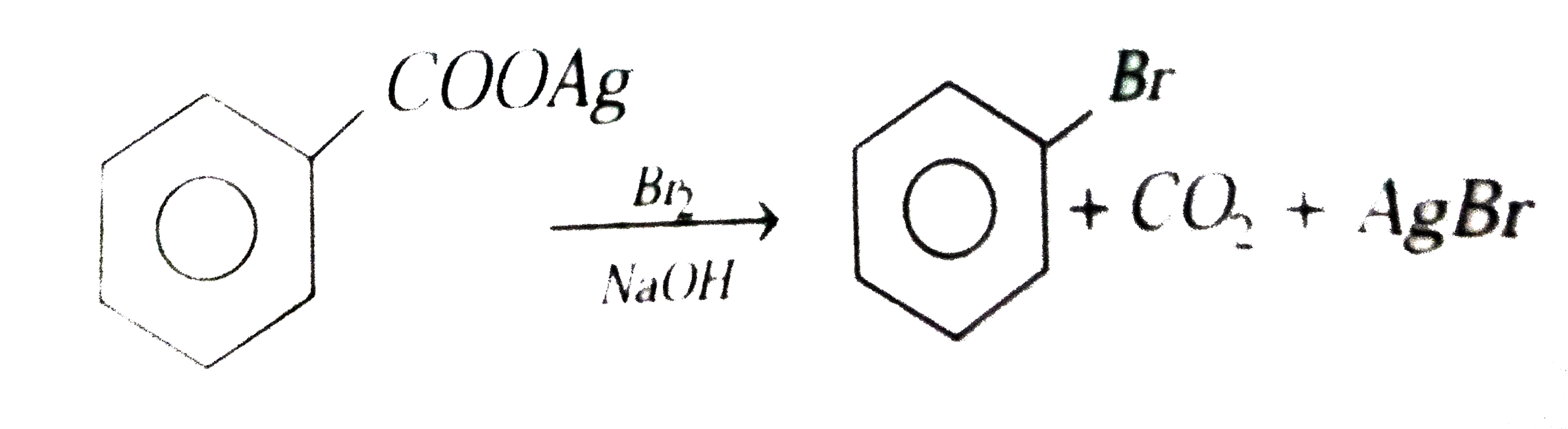
|
|
| 9. |
Silveratomhas completelyfilled d- orbitals(4d^(10)) in itsgroundstate . Howcan yousaythat it istransitionelement ? |
|
Answer» Solution :(i)Silverin its+1oxidationstateexhibits`4d^(10) 5S^(0)`configuration (ii) But in some compounds , it alsoshows +2oxidationstateso theconfigurationbecomes `4d^(9) 5s^(0)` (iii)Hered- orbitalis notcompletely filledThereforesilveris atransitionelement . |
|
| 10. |
Silver benzoate reacts with bromine in acetone to form - |
|
Answer»
|
|
| 11. |
Silver (Atomic weight =108g "mol"^(-1) has a density of 10.5g cm^(-3). The number of silver atoms on a surface area of 10^(-12) can be expressed in scientific notation as yxx10^(x). The value of x is: |
|
Answer» NUMBER of ATOMS `-(10.5)/(108)xx6.023xx10^(23)` Number of atoms in 1 cm`=[(10.5)/(108)xx6.023xx10^(23)]^(1//3)` Number of atoms in `1 cm^(2)=[(10.5)/(108)xx6.023xx10^(23)]^(2//3)` Number of atoms in `10^(-2)m^(2)` or `10^(-8)cm^(2)` `=[(10.5)/(108)xx6.023xx10^(23)]^(2//3)xx10^(-8)=1.5xx10^(7)`. |
|
| 12. |
Silver Benzoate on reaction with Bromine in Acetone form |
|
Answer»

|
|
| 13. |
Silver atom has completely filledd-orbitals ( 4d^(10)) in its ground state. How can you say that it is a transition element ? |
| Answer» Solution :The outer electronic CONFIGURATION of `AG(Z = 47)` is `4d^(10) 5s^(1)` .In ADDITION to `+1` , it shows an oxidation state of `+2` ( e.g., `AgO` and `AgF_(2)` exist ). In `+2` oxidation state, the configuration is `d^(9)` , i.e., the d-subshell is incompletely filled. Hence, it is a transition ELEMENT. | |
| 14. |
Silver atom has completely filled d-orbitals (4d^(10)) in its ground state. How can you say that it is a transition element ? |
| Answer» Solution :The outer electronic configuration of Ag (Z = 47) is `4d^(10)5S^(1)`. In addition to +1, it SHOWS an OXIDATION state of +2 also. In +2 oxidation state, the configuration is `d^(9)`, i.e., the d-subshell is incompletely filled. Hence, it is a transition ELEMENT. | |
| 15. |
Silver atom has completely-filled d-orbital(4d^105 s^1) in its ground state. How can you say- itis a transition element? |
| Answer» Solution :In addition to the common oxidation state of `+1`, silver ALSO SHOWS `+2` oxidation state in some of its COMPOUNDS. In the `+2` state, -the outer electronic configuration of Ag is `4d^2` and the cf-orbital is incompletely FILLED. ,So Ag is a TRANSITION metal. | |
| 16. |
Silver atom has completely filld d-orbitals (4^(10)) in its ground state. How can you say that it is a transition element? |
|
Answer» Solution :SILVER (Z=47) in (+2) oxidation state has incompletely filld 4d orbitals. Hence, it is a TRANSITION elements. `""_(47)Ag: [Kr] 4d^(10) 5s^(1) " " ""_(47)Ag^(+) : [Kr] 4d^(10) 5s^(0)` `""_(47)Ag^(2+): [Kr] 4d^(9) 5s^(0)` |
|
| 17. |
Silver and gold atoms have nearly the same atomic radii due to "…..........." |
|
Answer» |
|
| 18. |
Silver acetylide when heated with HCl gives |
|
Answer» `C_2H_4` |
|
| 19. |
Silver amalgam is used in: |
|
Answer» SILVERING of mirror |
|
| 20. |
Silver Acetate + Br_(2) overset(CS_(2)) to Main Product . The major product of this reaction is |
|
Answer» `CH_(3)-BR` |
|
| 22. |
Silicones are water repelling in nature because- |
|
Answer» they have highly COVALENT `Si-O-Si` linkages. |
|
| 23. |
Silicones are used for making waterproof fabrics. Give reason. |
| Answer» Solution :All SILICONES are water REPELLENT. This property arises due to the presence of organic side groups that surrounds the silicon which makes the molecule LOOKS LIKE an ALKANE. | |
| 24. |
Silicones are group of organosilicon polymers containing |
|
Answer» Si-O-Si LINKAGE |
|
| 25. |
Silicones are ____________ |
|
Answer» ortho silicates |
|
| 26. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. Draw the structure of the anion present in pyroxene (MgCaSi_(2)O_(6)). Silicones are important synthetic polymers which find extensive applications due to their chemical inertness and water repelling nature. Thet are produced via the following reactions. 2 C_(6)H_(5)Cl +Si underset("370 K")overset("Cu")to A A underset(-HCl)overset(H_(2)O)to B |
|
Answer» |
|
| 27. |
Silicon is usually found in: |
|
Answer» Sand |
|
| 28. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. Draw the structure of a cyclic silicate having structural formula of [Si_(6)O_(18)]^(n-).Also determine the value of n [Hint : SiO_(4)^(4-) canbe shown as unit] |
|
Answer» |
|
| 29. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. Identify A and the corresponding linear polymer B. |
|
Answer» |
|
| 30. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. The reactivities of C Cl_(4) and SiCl_(4) are different. For example SiCl_(4) can be easily hydrolysed and is prone to substitution reactions, whereas C Cl_(4) is inert. The observed difference is because (a)carbon atom has smaller size than hence substitution is not possible (b)carbon is more electronegative than silicon ( c)silicon has low lying unoccupied orbitals (d)C-Cl bond is stronger than Si-Cl bond Silicon has high affinity for oxygen to form silicates having SiO_(4)^(4-) units.Silicates can have chain or cyclic structures. |
|
Answer» CARBON atom has smaller size HENCE SUBSTITUTION is not possible |
|
| 31. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. The reason for the distinct difference in the properties of CO_(2) and SiO_(2) is : (a)carbon is more electronegative then O and in case of SiO_(2) oxygen is more electronegative than silicon (b)carbon has a small size and forms a pi bond with good overlap whereas silicon has larger size hence has a poor pi overlap ( c)carbon has only 'rho' orbitals and lacks 'd' orbitals whereas silicon has 'd' orbitals (d)first ionization potential of carbon is higher than that of silicon (1086 kJ mol^(-1) for C and 786 kJ mol^(-1) for Si). |
|
Answer» |
|
| 32. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. Glass, made of SiO_(2),Na_(2)SiO_(3) and CaSiO_(3) is attacked by hydrofluoric acid with formation of SiF_(6)^(2-) anion. The analogous CF_(6)^(2-) anion does not exist. The reason/s is/are : (a)carbon is more electronegative than silicon (b)silicon has larger atomic size than carbon (c)silicon has 3d orbitals which form an sp^(3)d^(2) hybrid orbitals (d)carbon and fluorine have comparable atomic sizes |
|
Answer» |
|
| 33. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. In zone refining, silicon is purified as (a)portions of Si rod are heated and cooled so that impurities are differentially evaporated (b)impurities are more soluble in liquid phase than in solid ( c)impurities are less soluble in liquid phase than in solid (d)impurities are insoluble in molten Si and can be separated |
|
Answer» PORTIONS of Si rod are heated and COOLED so that impurities are differentially evaporated |
|
| 34. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. In a silicon manufacturing unit, silicon is obtained by heating 100 kg of pure sand with 45 kg of high grade coke in an electric furnace. Write the balanced equation of the reaction. |
|
Answer» |
|
| 35. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. Silicon can be purified by converted it into volatile SiCl_(4)(b.p. 58^(@)C)which is purified by fractional distillation SiCl_(4) can then be converted into Si using molecular hydrogen. Write the balanced equations for the reactions involved. Silicon is widely used in semiconductor industry in which the required is of the order of 1 part per billion. This can be achieved using "zone refining technique". The technique involves moving a heater coil across an impure Si rod. |
|
Answer» |
|
| 36. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. The order in 1.4 can be explained on the basis of : (a)atomic size , (b)electronegativity (c )bond strength , (d)non-metallic character |
|
Answer» |
|
| 37. |
Silicon is the second most abundant element (~27.2%)in the earth's after oxygen (45.5 %) Carbon. Silicon, germanium, tin and lead constitute the group 14 of the periodic table. Chemistry of silicon is distinctly different from that of carbon. For example, under standard conditions CO_(2) is a gas whereas SiO_(2) is a covalent solid. Group 14 elements have tendency to catenate. The first three members C, Si and Ge show significant catenation. Arrange these elements in decreasing order of their catenation tendency. |
|
Answer» |
|
| 38. |
Silicon is second most abundant element occuring in earth crust it always found in combined state. It is obtained by reduction of silica. It exists in two allotropic forms amorphous and crystalline. Amorphous forms is obtained by heating dry powered silica with magnesium. Crystalline form is obtained by heating sand or quartz in an furnaces. Silicon is used in manufacture of certain alloys and polymers such as ferrosilicon, silicon benezne etc. The compounds of silicon are carborundum, silicons, silicates, glass etc. Silicons are organosilicon polymer and silicates are metal derivatives of salicic acid. While carborundum are obtained by heating mixture of sand carbon and salt. The ordinary formulae for a glass obtained from silica is: Where: R=Na or K and M=Ca, Ba, Zn or Pb |
|
Answer» RO.MO.`4SiO_(2)` |
|
| 39. |
Silicon is second most abundant element occuring in earth crust it always found in combined state. It is obtained by reduction of silica. It exists in two allotropic forms amorphous and crystalline. Amorphous forms is obtained by heating dry powered silica with magnesium. Crystalline form is obtained by heating sand or quartz in an furnaces. Silicon is used in manufacture of certain alloys and polymers such as ferrosilicon, silicon benezne etc. The compounds of silicon are carborundum, silicons, silicates, glass etc. Silicons are organosilicon polymer and silicates are metal derivatives of salicic acid. While carborundum are obtained by heating mixture of sand carbon and salt. (SiH_(3))_(3)N is weaker base than (CH_(3))_(3) N because: |
|
Answer» In `(SiH_(3))_(2)N` ions pair of electron on nitrogen is involve in `dpi-ppi` bonding. |
|
| 40. |
Silicon is second most abundant element occuring in earth crust it always found in combined state. It is obtained by reduction of silica. It exists in two allotropic forms amorphous and crystalline. Amorphous forms is obtained by heating dry powered silica with magnesium. Crystalline form is obtained by heating sand or quartz in an furnaces. Silicon is used in manufacture of certain alloys and polymers such as ferrosilicon, silicon benezne etc. The compounds of silicon are carborundum, silicons, silicates, glass etc. Silicons are organosilicon polymer and silicates are metal derivatives of salicic acid. While carborundum are obtained by heating mixture of sand carbon and salt. When two structural units of silicates join along a corner of containing oxygen atom called as |
|
Answer» Orthosilicates |
|
| 42. |
Silicon is second most abundant element occuring in earth crust it always found in combined state. It is obtained by reduction of silica. It exists in two allotropic forms amorphous and crystalline. Amorphous forms is obtained by heating dry powered silica with magnesium. Crystalline form is obtained by heating sand or quartz in an furnaces. Silicon is used in manufacture of certain alloys and polymers such as ferrosilicon, silicon benezne etc. The compounds of silicon are carborundum, silicons, silicates, glass etc. Silicons are organosilicon polymer and silicates are metal derivatives of salicic acid. While carborundum are obtained by heating mixture of sand carbon and salt. The structure unit of silicates is: |
|
Answer» `SiO_(2)` |
|
| 43. |
Silicon is second most abundant element occuring in earth crust it always found in combined state. It is obtained by reduction of silica. It exists in two allotropic forms amorphous and crystalline. Amorphous forms is obtained by heating dry powered silica with magnesium. Crystalline form is obtained by heating sand or quartz in an furnaces. Silicon is used in manufacture of certain alloys and polymers such as ferrosilicon, silicon benezne etc. The compounds of silicon are carborundum, silicons, silicates, glass etc. Silicons are organosilicon polymer and silicates are metal derivatives of salicic acid. While carborundum are obtained by heating mixture of sand carbon and salt. Which type of linkage present in silicones? |
|
Answer»
|
|
| 44. |
Silicon is found in nature in the form of |
|
Answer» Body centered CUBIC STRUCTURE |
|
| 45. |
Silicon is an important constituent of : |
|
Answer» Chlorophyll |
|
| 46. |
Silicon is main constituent of |
|
Answer» Alloys |
|
| 47. |
Silicon hydrides are named as : |
|
Answer» SILICONES |
|
| 48. |
Siliconhas4 elements in the outermost orbit. In forming the bonds, |
|
Answer» It gains electrons |
|
| 49. |
Silicon doped with electron-rich impurity forms |
|
Answer» P-type semiconductor |
|
| 50. |
silicon doped with arsenic is an example of : |
|
Answer» p-type conductor |
|