Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 2. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 3. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 4. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 5. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 6. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 7. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 8. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 9. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 10. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 11. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 12. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 13. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 14. |
Prove the buffer action of acetic acid and sodium acetate by the addition of 0.01 mol of solid sodium hydroxide. |
|
Answer» Solution :(i) Consider ONE litre to buffer solution containing 0.8 cm `CH_3COOH` and 0.8 cm `CH_3COONa`. Assume that the VOLUME changes due to the addition of 0.01 mol of solid NaOH is negligible. `K_a` for `CH_3COOH` is `1.8 times 10^-5` (ii) `Ch_3COOH(AQ) leftrightarrow^(H_2O(l)) CH_3COO^(-) (aq) +H_(aq)^+` 0.8-aq a a `CH_3COONa_(aq) to^(H_2O) CH_3COO^(-) (aq)+Na_(aq)^+` 0.8 0.8 0.8 (iii) The dissociation constant for `Ch_3COOH` is given by `K_a=([CH_3COO^-][H^+])/([CH_3COOH])` `[H^+]=(K_a[CH_3COOH])/([CH_3COO^-])` The above expression shows that the concentration of `H^+` is directly proportional to `([CH_3COOH])/([CH_3COO^-])` degree of dissociation of `CH_3COOH-a` (iv) `[CH_3COOH]=0.8-a and [CH_3COO^-]=a+0.8` `therefore [H^+]=K_a.([0.8-a])/([0.8+a])` `If a lt lt0.8,0.8-a approx0.8` `0.8+a approx0.8` `therefore [H^+]=(K_a.[0.8])/([0.8])=K_a` `[H^+]=K_a` (v) Given that `K_a` for `CH_3COOH` is `1.8 times 10^-5` `[H^+]=1.8 times10^-5` `pH=-log[H^+]` `=-log[1.8 times10^-5]` `=5-log 1.8` `=5-0.26` `pH=4.74` (vi) After adding 0.1 mol NaOH to 1 litre of buffer. Given that volume change due to the addition of NaOH is negligible `therefore [OH^-]=0.01M` The consumption of `OH^-` are represented by the following EQUATION `CH_3COOH_(aq) leftrightarrowCH_3COO_(aq)^(-) +H^+ (aq)` 0.8-aa a `CH_3COONa_(aq) toCH_3COO^(-)+Na^(+) (aq)` 0.8 0.8 0.8 `therefore[CH_3COOH]=0.8-a-0.01=0.79-a` `[CH_3COO^-]=a+0.8+0.01` `=0.81+aa lt lt 0.8` `0.79-a approx0.79 and 0.81+a approx 0.81` `therefore [H^+]=1.8 times 10^-5 times 0.79/0.81` `[H^+]=1.76 times10^-5` `therefore pH=-log(1.76 times10^-5)` `=5- log 1.76` `=5-0.25` `pH=4.75` (vii) The addition of a strong base (0.01M NaOH) increased the pH only slightly i.e., from 4.74to 4.75. SO the buffer action is verified. |
|
| 15. |
Provide the missing compound A-C, in the following synthesis |
Answer» Solution :The FIRST reaction is a chrmoic acid oxidation , leading to `C_(3)H_(10)O_(2)`, which is conistent with the carboxylic acid derived from 3-methyl-1-butano B must be a reagent by which we can prepare an acid chloride. The final PRODUCT is an amide, THUS C must be the appropriate amine. compounds A-C therefore, are so FOLLOWS: 
|
|
| 16. |
Provide structures for A and B. |
Answer» Solution :`H_(2)CrO_(2)` oxidizes a primary alcohol to a arboxylic acid which is CONSISTENT with the FORMULA provided for A. Because A is a `beta`-ketocarboxylic acid. It decarboxylates on heating to form B. 
|
|
| 17. |
Prove thet relative lowering in vapour pressure of a liquid on addition of a non-volatile solute is a colligative property. |
| Answer» SOLUTION :For ANSWER CONSULT SECTION 9. | |
| 18. |
Prove that the time required for the completion (3^(th))/(4) of the reaction of a fitst order is twice the time required for the completion of a half of the reaction. |
|
Answer» SOLUTION :`t_((3)/(4))=(2.303)/(K)"log"([R]_(0))/((1)/(4)[R]_(6))` `t_((3)/(4))=(2.303)/(K)log 4` `= (2.303xx0.6021)/(K)=(1.386)/(K)` `= 2xx(0.693)/(K)` `= 2 t_((1)/(2))` |
|
| 19. |
Provethat sucroseis (i) invertsugar (ii) non - reducingsugar. |
|
Answer» SOLUTION :(i) Sucroseis an invertsugar : SUCROSE `(+66.6^(@))` and glucose`(+52.5^(@))`are dextro rotatorycompounds whilefurcostein levo rotatory`(-92.4^(@))` . Duringhydrolysis of sucrose , theopticalrotationof thereactionmixturechanges fromdextroto levo. Hencesucrose is alsocalledinvertsugar. (ii) In sucrose`C_(1)`of `ALPHA ` - Dglucoseis joinedto `C_(2)`of `alpha`- Dfructose . Theglycosidicbondthusformedis called`alpha` - 1, 2 - glycosidic bond . Sinceboth thecarbonyl carbonscarbons(reducinggroups) are involvedin the glycosidicbonding , surcoseis a non - reducingsugar. |
|
| 20. |
Prove that the colloidal solutions are itermediate between true solutions and suspensions. Give one example each of lyophilic and lyophobic sols. |
| Answer» | |
| 21. |
Prove that sucrose is (i) invert sugar (ii) non-reducingsugar. |
|
Answer» |
|
| 22. |
Prove that relative lowering of vapour pressure of a solution containing non-volatile solute is equal to its mole fraction. |
| Answer» SOLUTION :For ANSWER CONSULT SECTION 9. | |
| 23. |
Prove that relative loweing in vapour pressure is a colligative property. |
| Answer» SOLUTION :For ANSWER CONSULT Table 5. | |
| 24. |
Prove that Potassium permanganate is an oxidising agent in neutral medium. |
|
Answer» SOLUTION :In neutral medium, `KMnO_(4)` it is reduced to `MnO_(2)` `MnO_(4)^(-) + 2H_(2)O + 3e^(-) toMnO_(2) + 4OH^(-)` Example: Potassium PERMANGANATE oxidises `H_(2)S` to SULPHUR in neutral medium. `2MnO_(4)^(-) + 3H_(2)S to 2MnO_(2) + 3S + 2OH^(-) + 2H_(2)O` |
|
| 25. |
Prove that H_(2)SO_(4) is strong dibasic acid. |
|
Answer» Solution :Sulphuric acid forms two TYPES of salts namely SULPHATES and bisulphates. `H_(2)SO_(4)+NAOH rarr underset("sodium bisulphate")(NaHSO_(4))+H_(2)O` `H_(2)SO_(4)+2NaOH rarr underset("sodium sulphate")(Na_(2)SO_(4))+2H_(2)O` `H_(2)SO_(4)+2NH_(3)rarr underset("AMMONIUM sulphate")((NH_(4))_(2)SO_(4))` |
|
| 26. |
Prove that H_2SO_4 is a strong dibasic acid. (ii) Distinguish tetrahedral and octahedral voids. (b) In an octahedral crystal field , draw the figure to show splitting of d orbital's. |
|
Answer» Solution :(a) (i) Sulphuric acid forms two types of salts namely sulphates and bisulphates . `H_2SO_4+NaOHrarrunderset("sodium bisulphate")(NaHSO_4)+H_2O` `H_2SO_4+2NaOHrarrunderset("sodium sulphate")(Na_2SO_4)+2H_2O` `H_2SO_4+2NH_3rarrunderset("Ammonium sulphate")((NH_4)_2SO_4)`  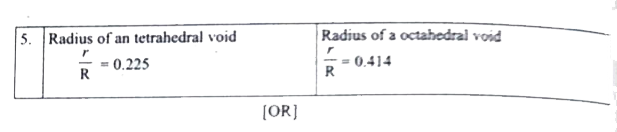 [OR] b. Step I : In an isolated gaseous state, all the five d orbital of the CENTRAL metal ion degenerate. Initially, the ligands from a spherical filed of negative charge around the metal, in this field the the energies of all the five orbitals willincrease due to the repulsion between the electrons of of the metal andthe ligand. Step 2: The ligands are approaching the metal atom in actual bond directions. To illustrate this letus consider and octahedral field, in which the central metal ionsis location at the origin and the six ligandsare COMING from the `+x,-x,+y,-y,+z and -z` direction as SHOWN below. . As shown in the figure,theorbitals lying along the AXES `dx^2-y^2 and dz^2` orbitals will experience strong repulsion and raise in energy to greater extent then the orbitals with lobes direction between the the axes `(d_(xy),d_(yz) and d_(zx))`Thus the degenerate d orbitals now split into two sets and the process is called crystal field splitting . 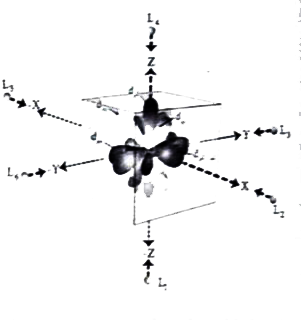 Step 3 : Up to this point the complex formation WOULD not be favoured. However , when the ligands approach further , there will be an attraction between the negatively charged electron and the positively charged metal ion, that results in a net decrease in energy.This decrease in energy is the driving force for the complex formation During crystal field splitting in octahedral field, in order to maintain the average energy of the orbitals (barycentre ) constant , the energy of the orbitals `d_(x^2-y^2)and d_(z^2)` (represented as `e_g` orbitals ) will increase by `3//5Delta_@`while that of the other three orbitals `d_(xy),d_(yz) and d_(zx)`( represented `t_(2g)`orbitals) decrease by `2//5Delta_@`Here, `Delta_@` represents the crystal field splitting energy in the octahedral field. 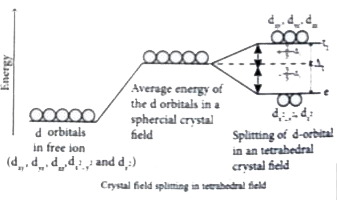
|
|
| 27. |
Prove that dipole moment of C_(6)H_(5)Cl and m-C_(6)H_(4)Cl_(2) are same |
| Answer» | |
| 28. |
Prove that acidified potassium dichromate is a powerful oxidising agent. |
|
Answer» Solution :`K_(2)Cr_(2)O_(7)` ACT as power oxidising agent in acidic medium. In the presence of `H^(+)` ions, the oxidation state of Cr froms `Cr^(6+)` is changed to `Cr^(3+)` `Cr_(2)O_(7)^(2-)+ 14H^(+) + 6E^(-) to2Cr^(3+) + 7H_(2)O` Example: ACIDIFIED `K_(2)Cr_(2)O_(7)` oxidises Ferrous salts to Ferric salts. `Cr_(2)O_(7)^(2-)+ 6Fe^(2+) + 14H^(+) to2Cr^(3+) + 6Fe^(3+) + 7H_(2)O` |
|
| 29. |
Prove that 1F=96500C. |
|
Answer» Solution :`"Charge of ONE ELECTRON"=1.6xx10^(-19)C` `"Charge one mole of electron "=6.023xx10^(23)xx1.602xx10^(-19)C` `=96488C` i.e., `1F~=96500C` |
|
| 30. |
Prove from the following resutls that mercury molecules are monoatomic: (a) 10g of mercury combine with 0.8g of oxygen to form an oxide. (b) 1000mL of vapour of Hg at NTP weigh 8.923g (c) The specific heat of the metal is 0.033. |
|
Answer» SOLUTION :(a) Equivalent MASS of Hg `=("Mass of metal")/("Mass of oxygen")xx8=(10)/(0.8)xx8=100` (b) Mass of 1000 mL of Hg vapour at NTP=8.923g Mass of 22400mL of Hg vapour at NTP=`(8.923)/(1000)xx22400` `=199.87g` Hence: molecular mass of Hg `=188.87g` (c) Approximate ATOMIC mass `=(6.4)/("Sp.heat")=(6.4)/(0.033)=193.93g` Valence of Hg `=(193.93)/(100)=2`(nearest whole number ) So, accurate atomic mass =Eq. mass `xx` Valence `=100xx2=200g` AUTOMICITY `=("Mol.mass")/("At.mass")=199.88)/(200)=1` Hence, mercury molecules are MONOATOMIC. |
|
| 31. |
Prouductof the followingreaction willbe CH_(3) - CH_(2) - CHO + CH_(3) - NO_(2) overset(alc. KOH) to |
|
Answer»
|
|
| 32. |
Prove ammonia act as a reducing agent? |
|
Answer» Solution :AMMONIA reduces the metal oxides to metal when passed over HEATED metallic oxide. `3PbO+2NH_(3)rarr 3Pb +N_(2)+3H_(2)O` |
|
| 33. |
Protons and neutrons are bound in a nucleus by the |
|
Answer» short range 'weak interaction' |
|
| 34. |
Protons accelerate the hydrolysis of esters. This is an example of : |
|
Answer» A HETEROGENEOUS catalysis |
|
| 35. |
Protons accelerate the hydrolysis of ester. This is an example of |
|
Answer» A NEGATIVE catalyst |
|
| 36. |
Proton bombardment on Th^(230) followed by emission of two alpha particles will produce: |
| Answer» Answer :C | |
| 37. |
Protocity of sulphuric acid is two. Explain? |
|
Answer» Solution :Sulphuric acid forms two SERIES of salts. Hence it is a dibasic acid. It forms two types of salts. `H_(2)SO_(4)+NaOHrarr H_(2)O+NaHSO_(4)` (acidic salt) `H_(2)SO_(4)+2NaOHrarr2H_(2)O+Na_(2)SO_(4)` (normal salt) |
|
| 38. |
Proteins which act as biological catalysts are called .................... and those which are important for communication systems are called .................... |
| Answer» SOLUTION :ENZYMES, RECEPTORS | |
| 39. |
Proteins when heated with cone. HNO_3give a yellowcolour. This is |
|
Answer» OXIDISING test Protein+ Conc. `HNO_3 to` Yellow colour |
|
| 40. |
Proteins when heated with conc. HNO_(3) give a yellow colour. This is |
|
Answer» Oxidising test [This test is given by a protein which consists of `alpha`-amino acid containing a BENZENE ring such as TYROSINE, phenylanine etc. the yellow colour is DUE to to nitration of benzene ring.] |
|
| 41. |
Proteins when heated with conc. HNO_3 give a yellow colour. This is |
|
Answer» OXIDIZING test |
|
| 42. |
Proteins such as. ………………….,………..act as structural back bones . |
| Answer» SOLUTION :KERATIN,COLLAGEN | |
| 43. |
Proteins on complete hydrolysis give : |
|
Answer» `alpha`-amino acids |
|
| 45. |
Proteins give a white precipitate with Millon's reagent,which is |
|
Answer» Mercurous and MERCURIC NITRATE in `HNO_3` |
|
| 46. |
Proteinsfulfil several functions in living system . An example of a protein which acts as a hormone is |
|
Answer» CASEIN |
|
| 47. |
Proteins found in a biological system witha uniquethree - dimensionalstructureand biologicalactivity is calleda nativeproteinin its nativeform , is subjected to a physical changelikechangein temperature or a chemical changein pH , denaturation ofprotein takes place. Explain the cause . |
| Answer» Solution :Due tophysicalor chemcial change, hydrogenbondsin PROTEINS are BROKEN , GLOBULES unfold and helix GETS uncoiled thereforeprotein loses itsbiologicalactivity. Thisis calleddenaturationof proteins. | |
| 48. |
Proteins give: |
|
Answer» A VIOLET COLOUR with ALKALINE |
|



