Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Photochemical smog occursis warm dry and sunnyclimate.Oneof thefollowingis notamongstthe components of photochemicalsmogidentifyit. |
|
Answer» `NO_(2)` |
|
| 2. |
Photochemical smog is related to the pollution of |
|
Answer» Noise |
|
| 3. |
Photochemical smog occors at temperature. |
| Answer» | |
| 4. |
Photochemical smog is formed in : |
|
Answer» SUMMER during MORNING time |
|
| 6. |
Photochemical smog is a mixture of oxidising pollutants. |
|
Answer» |
|
| 7. |
Photochemical smog consists of excessive ammmt of X, in addition to aldehydes, ketones, peroxyacetyl nitrate (PAN), and so forth, X is: |
|
Answer» `CO` |
|
| 8. |
Photochemical smog formed in congested metropolitan cities mainly consists of |
|
Answer» HYDROCARBONS, `SO_2` and `CO_2` |
|
| 9. |
Photochemical smog consists of excessive amount of X, in addition to aldehydes, ketones, peroxyacetyl nitrate (PAN), and so forth X is: |
| Answer» Answer :D | |
| 10. |
Photochemical bromination of n-butane produces |
|
Answer» 1-BROMOBUTANE as the major product Thus 2-bromobutane is the major product |
|
| 11. |
Photochemical chlorination of alkanes is initiated by a process of |
|
Answer» Pyrolysis |
|
| 13. |
Photochemical chlorination of alkane is initiated by a process of: |
|
Answer» Pyrolysis |
|
| 14. |
Phosphorus pentachloride dissociates as follows, ina closed reaction vessel, PC1_(5(g))hArrPC1_(3(g)) + C1_(2(g)) If total pressure at equilibrium of the reaction mixture is P and degree of dissociation of PC1_(5) is x, the partial pressure of PC1_(3) will be: |
|
Answer» `((x)/(x-1))P` `therefore"Partial pressure of" PCl_(3)=((x)/(x+1))P` |
|
| 15. |
Phosphate ester of X is called a nucleotide. X is |
|
Answer» adenine |
|
| 16. |
Phospodiester linkages are present between |
|
Answer» `C-3,` of ONE NUCLEOTIDE and C-5, of NEXT nucleotide |
|
| 17. |
Phosphorus when dissolved in alkali, the products formed are |
|
Answer» <P>`PH_(3)+NaH_(2)PO_(2)` |
|
| 18. |
Phosphorylation of glucose is increased by |
|
Answer» AUXINS |
|
| 19. |
Natuaral reservoir of phosphorus is : |
| Answer» Answer :D | |
| 20. |
Phosphorus when exposed to air burns spontaneously because: |
|
Answer» The REACTION is endothermic |
|
| 21. |
Phosphorus pentoxide reacts with nitric acid to form. |
| Answer» SOLUTION :`N_2O_5` | |
| 22. |
Phosphorus pentoxide is : |
|
Answer» OXIDISING AGENT |
|
| 23. |
Phosphorus pentachloride reacts with sulfur dioxide to form |
|
Answer» Chlorosulfonic ACID |
|
| 24. |
Phosphorus pentoxide cannot be used to dry: |
|
Answer» Nitrogen |
|
| 25. |
Phosphorus pentachloride reacts with acetone to give POCl_(3) and : |
|
Answer» `CH_(3)-CHCl-CH_(3)` |
|
| 26. |
Phosphorus pentachloride dissociates as follows in a closed vesselmPCl_(5)(g)hArr PCl_(3)(g)+Cl_(2)(g).If total pressure at equilibrium of the reaction mixture is P and degree of dissociation of PCl_(5) is x, the partial pressure of PCl_(3) will be : |
|
Answer» `((2x)/(1-X))P` Total moles `=1-x+x+x=1+x` PARTIAL pressure of `PCl_(3)=((x)/(1+x))P`. |
|
| 27. |
Phosphorus occurs in nature in the form of stable phosphates. (Animal bones also contain calcuim phosphate (58%)).The important minerals are: (i)Phosphorite, Ca_3(PO_4)_2(ii)Chloraptite,Ca_3(PO_4)_2CaCl_2 (iii)Fluoraptite, Ca_3(PO_4)_2CaF_2 There are some depoists of nitrates and phosphates in earth's crust.Nitrates are more soluble in water Nitrates are difficult to reduce under the laboratory conditions but microbes do it easily.Compounds of phosphorus and nitrogen are used as fertilizer. Amongs the following select the correct statement : |
|
Answer» Regular use of Superphosphate of lime fertilizers increases the acidity of soil Polyphosphate SEPARATE `Ca^(2+)` and `Mg^(2+)` (present in hard water ) and forms soluble complex with `Ca^(2+)` and `Mg^(2+)` which do not precipitate with `CO_3^(2-)` and soap. |
|
| 28. |
Phosphorus occurs in nature in the form of stable phosphates. (Animal bones also contain calcuim phosphate (58%)).The important minerals are: (i)Phosphorite, Ca_3(PO_4)_2(ii)Chloraptite,Ca_3(PO_4)_2CaCl_2 (iii)Fluoraptite, Ca_3(PO_4)_2CaF_2 There are some depoists of nitrates and phosphates in earth's crust.Nitrates are more soluble in water Nitrates are difficult to reduce under the laboratory conditions but microbes do it easily.Compounds of phosphorus and nitrogen are used as fertilizer. Ca_3(PO_4)_2CaF_2+H_2SO_4to(X)+HF X is - |
|
Answer» Triple SUPER PHOSPHATE `H_3PO_4` is USED to avoid the FORMATION of the insoluble `CaSO_4` (waste PRODUCT). |
|
| 29. |
Phosphorus occurs in nature in the form of stable phosphates. (Animal bones also contain calcuim phosphate (58%)).The important minerals are: (i)Phosphorite, Ca_3(PO_4)_2(ii)Chloraptite,Ca_3(PO_4)_2CaCl_2 (iii)Fluoraptite, Ca_3(PO_4)_2CaF_2 There are some depoists of nitrates and phosphates in earth's crust.Nitrates are more soluble in water Nitrates are difficult to reduce under the laboratory conditions but microbes do it easily.Compounds of phosphorus and nitrogen are used as fertilizer. Amongs the following which is not member of apatite family ? |
|
Answer» `Ca_9(PO_4)_6. CaCl_2` |
|
| 30. |
Phosphorus occus in free state. |
| Answer» Solution :PHOSPHORUS DOCS not CCUR in the FREE state. | |
| 31. |
Phosphorus is kept in : |
|
Answer» COLD water |
|
| 32. |
Phosphorus is manufactured by heating____in a furnace. |
|
Answer» Bone-ash, SODIUM CHLORIDE and COKE |
|
| 33. |
Phosphorus has three allotropic forms : (i) white phosphorus (ii) red phosphorus and (iii) black phosphorus. Write the difference between white and red phosphorus on the |
Answer» SOLUTION :
|
|
| 34. |
Phosphorus in +1 oxidation state is found in: |
|
Answer» `H_3PO_3` |
|
| 35. |
Phosphorus has the oxidation state of + 3 in |
|
Answer» orthophosphoric acid |
|
| 36. |
Phosphorus forms five bonds due to sp^(3)d hydridisation but nitrogen does not form pentahalides due to : |
|
Answer» small SIZE of N atom |
|
| 37. |
Phosphorus forms a number of oxoacids. Out of these oxoacids, phosphinic acid has strong reducing property. Write its structure and also write a reaction showing its reducing behaviour. |
|
Answer» SOLUTION :The structure of phosphinic acid (hypophosphorus acid) is given on the R.H.S. Since it contains two P-H bonds, it acts as a strong reducing agent. For EXAMPLE, it reduces `AgNO_(3)` to metallic SILVER. `4 AgNO_(3) + H_(3)PO_(2) + 2H_(2)O rarr 4 Ag darr + H_(3)PO_(4) + 4 HNO_(3)` 
|
|
| 38. |
Phosphorus forms a number of oxoacids. Out of these oxoacids phosphinic acid has strong reducing property. Write its structure and also write a reaction showing its reducing behaviour. |
Answer» Solution :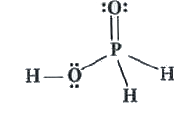 Phosphinic ACID acts as a good reducing agent because of presence of TWO P-H bonds. `4AgNO_(3) + 2H_(2)O + H_(3)PO_(2) to 4Ag + 4HNO_(3) + H_(3)PO_(4)` |
|
| 39. |
Phosphorus compound used as drying agent and desicating agent is: |
|
Answer» `PCl_3` |
|
| 40. |
Phosphorus can expand its valency. Why? |
| Answer» Solution :In its VALENCY SHELL, phosphorus has vacant d-orbitals, which can be used for bonding. Hence, phosphorus can expand its valency upto 6, as in `PF_(6)^(-)`. | |
| 41. |
Phosphorus and chlorine form two compounds. The first contains 22.54% by mass of phosphorus and the second 14.88% of phosphorus. Show that these data are consistent with law of multiple proportions. |
| Answer» Solution :The ratio of the masses of chlrorine which combines with a fixed mass of PHOSPHORUS in two COMPOUNDS is 3:5 which is simple whole NUMBER ratio. Thus, the data illustrate law multiple proportions. | |
| 42. |
Phosphorous acid on heating gives the following products: 4H_(3)PO_(3)overset(Delta)rarr3H_(3)PO_(4)+PH_(3) The above reaction is an example of |
|
Answer» OXIDATION |
|
| 43. |
Phosphorous is usually extracted from |
|
Answer» Phosphorite |
|
| 44. |
Phosphorium bromide is obtained by reacting phosphine with |
| Answer» ANSWER :A | |
| 46. |
Phosphoric acid on reaction with sufficient quantity of NaOH gives: |
|
Answer» `Na_3PO_4` |
|
| 47. |
Phosphoric acid is the most important of the phosphorus oxyacids. Industrially, phosphoric acid is prepared by |
|
Answer» the Ostwald process |
|
| 48. |
Phosphoric acid is syrupy liquid due to |
|
Answer» Strong COVALENT bond |
|
| 49. |
Phosphoric acid is prepared by |
|
Answer» HYDROLYSIS of `P_2O_3` |
|
| 50. |
Phosphoric acid ionises as :H_(3)PO_(4)overset(K_(1))hArr H^(+)+H_(2)PO_(4)^(-)H_(2)PO_(4)^(-)overset(K_(2))hArr H^(+)+H_PO_(2)^(-)H_(2)PO_(4)^(2-)overset(K_(3))hArr H^(+)+PO_(3)^(-)The equilibrium constant, K for the reaction :H_(3)PO_(4)hArr 3H^(+)+PO_(4)^(3-) is : |
|
Answer» `K_(1)//K_(2) K_(3)` |
|