Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Pearl white is: |
|
Answer» BiOCl |
|
| 2. |
Pearl ash and caustic potash are chemically: |
|
Answer» `K_2CO_3` and NAOH |
|
| 3. |
[PdCl_(2)(PM e_(3))_(2)] is a diamagnetic complex of Pd(II). How many total isomers are possible of analogous paramagnetic complex of Ni(II)? |
|
Answer» zero `Ni^(2+)` in `[NiCl_(2)(PM e_(3))_(2)]` 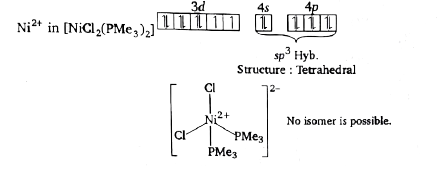
|
|
| 4. |
PDI for natural polymers is generally close to: |
|
Answer» Zero |
|
| 5. |
[PdCl_(2)(PM e_(3))_(2)] is a diamagnetic complex of Pd (II). How many unpaired electrons are presennt in analogous complex of Ni (II)? |
|
Answer» Zero |
|
| 6. |
[PdBr_4]^(2-) is a sqaure planar complex. The hybridisation of Pd^(2+)is |
|
Answer» `sp^3` |
|
| 7. |
Pd has exceptional outer electronic configuration as 4d^(10) 5 s^(0). It belongs to |
|
Answer» `4^(th)` period, group 11 |
|
| 8. |
Pd can adsorb 900 times its volume of hydrogen. This is called: |
|
Answer» Absorption |
|
| 9. |
PCl_(5) +SO_(2) rarr A+B A overset(3H_(2)O)rarrHCl C overset("Red hot")rarrD +H_(2)O Compound 'D'is |
|
Answer» `P_4O_6` `underset(C)(2H_3PO_3)overset("RED HOT")rarrunderset(P_4O_6)underset(darr"Dimerises")(P_2O_3)+3H_2O" "2PCl_(5)hArrPCl_4^(+)+PCl_6^(-)` |
|
| 10. |
PCl_5was found to dissociate to the extent of 42% at 227^@Cand 1 atm. Find the equilibrium constant at (i) constant pressure, and (ii) constant volume. |
| Answer» SOLUTION :0.214, 0.005 | |
| 11. |
PCl_(5) +SO_(2) rarr A+B A overset(3H_(2)O)rarrHCl C overset("Red hot")rarrD +H_(2)O Compound 'B' is |
|
Answer» `SOCl_2` `underset(C)(2H_3PO_3)OVERSET("Red hot")rarrunderset(P_4O_6)underset(DARR"Dimerises")(P_2O_3)+3H_2O" "2PCl_(5)hArrPCl_4^(+)+PCl_6^(-)` |
|
| 12. |
PCl_(5) reactws with ethanol to form chloroethane. However, with phenol, it does not give chlorobenzene but gives triphenylphoshate. Explain. |
Answer» SOLUTION :To begin with both ethanol and phenol react with `PCl_(5)` to give the corresponding chlorophosphate ESTERS. 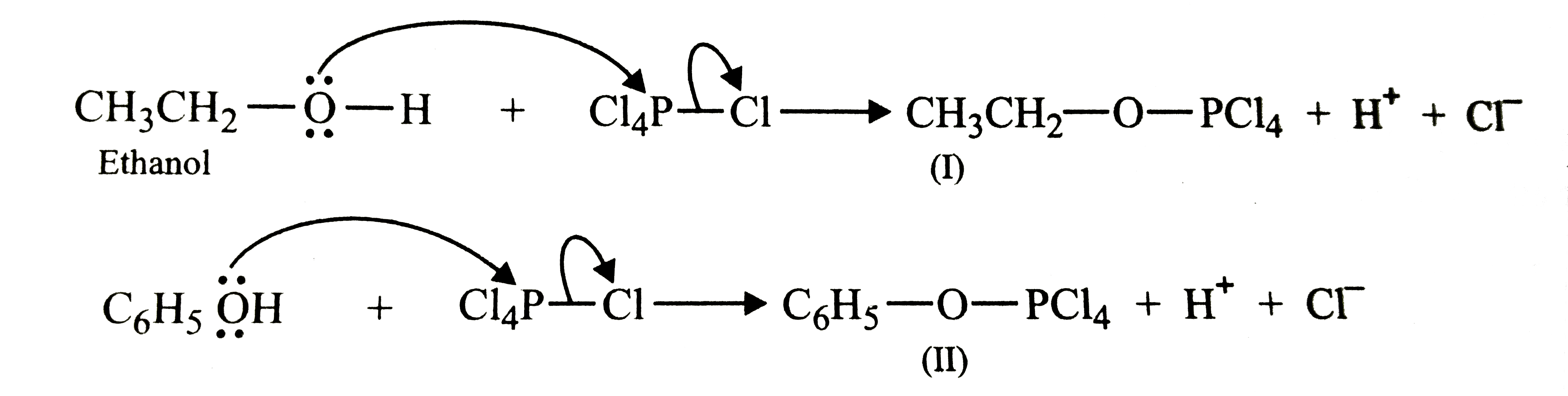 Since in ethanol, C-O bond has only single bond CHARACTER, therefore, nucleophilic ATTACK by `Cl^(-)` ion occurs easily on chlorophosphate ester (I) to give chloroethane.  however, in case of phenol, C-O bond has some double bond character due to resonance and hence difficult to break. instead nucleophilic attack by `C_(6)H_(5)OH` occurs twice on the chlorophosphate ester (II) to form intermediate (III). further, nucleophilic attack by `C_(6)H_(5)OH` on III does not occur due to steric hindrance. Instead (III) undergoes hydrolysis by water to form triphenylphosphate. 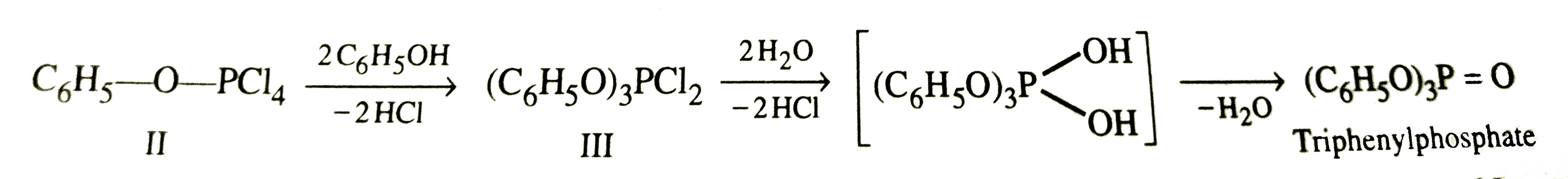 . .
|
|
| 13. |
PCl_(5) reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous NH_3 solution. Write the reactions involved to explain what happens. |
|
Answer» Solution :`PCl_5` will oxidise AG to AgCl. AgCl forms a soluble COMPLEX with ammonia when added in EXCESS. `PCl_(5) + 2Ag to 2AgCl + PCl_(3)` `underset("WHITE")(AgCl) + 2NH_(3) to underset("Soluble complex")([Ag(NH_(3))_(2)]Cl)` |
|
| 14. |
PCl_5 reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous NH_3 solution. Write the reactions involved to explain what happens. |
|
Answer» Solution :`PCl_5 + 2AG to 2AgCl + PCl_5` `AGCL + 2NH_3 (AQ) to underset("Soluble COMPLEX")([Ag(NH_3)_2]^(+) Cl^(-)` |
|
| 15. |
PCl_5 reacts with propanone, to give: |
|
Answer» Gem-dichloride |
|
| 16. |
Pcl_(5) reacts with a compoundcontaining |
|
Answer» `-SO_(3)` GROUP |
|
| 17. |
PCl_(5), PCl_(3) and Cl_(2) are at equilibrium at 500 K in a closed container and their concentrations are 0.8xx10^(-3) mol L^(-1), 1.2xx10^(-3)mol L^(-1) respectively. The value of K_(c ) for the reactionPCl_(5)(g)hArr PCl_(3)(g)+Cl_(2)(g)will be |
|
Answer» `1.8xx10^(-3) mol L^(-1)` `=1.8xx10^(-3) mol L^(-1)`. |
|
| 18. |
PCI_(5) on hydrolysis gives |
| Answer» Answer :2 | |
| 19. |
PCl_5 molecule has |
|
Answer» THREE FOLD AXIS of symmetry 
|
|
| 20. |
PCl_(5) molecule has the geometry : |
|
Answer» TRIGONAL bipyramidal |
|
| 21. |
PCl_5 is prepared by the action of Cl_2 on: |
|
Answer» `P_2O_3` |
|
| 22. |
PCl_5 is kept in well stoppered bottle because: |
|
Answer» It is HIGHLY volatile |
|
| 23. |
PCl_(5) is ionic in nature in the solid state. Give reasons. |
|
Answer» Solution :`PCl_(5)` conducts electricity in the molten state. This MEANS that in the solid state it CONSISTS of ions. `2PCl_(5)rarr [PCl_(4)]^(+)[PCl_(6)]^(-)`. On melting, these ions BECOMES free to move and hence `PCl_(3)` conducts electricity in the molten state. |
|
| 24. |
PCl_(5) is an example of a molecule having sp^(3)d-hybridisation. Three out of the orbitals in sp_(x)p_(y)-hybridisation while remaining two have p_(x)d_(x^(2))-hybridisation. If P-atom is attached to substitutuents differ in electronegativity, as in PCl_(x)F_(5-x), then it is has been experimently observed that the more electronegativity substituent occupies the axial position of t.b.p geometry. Q. The incorrect statement regarding PCl_(2)F_(2) molecule will be: |
|
Answer» given compound is polar |
|
| 25. |
PCl_(5) is an example of a molecule having sp^(3)d-hybridisation. Three out of the orbitals in sp_(x)p_(y)-hybridisation while remaining two have p_(x)d_(x^(2))-hybridisation. If P-atom is attached to substitutuents differ in electronegativity, as in PCl_(x)F_(5-x), then it is has been experimently observed that the more electronegativity substituent occupies the axial position of t.b.p geometry. Q.The correct statement is : |
|
Answer» in `CH_(2)F_(2)` the `F-C-F` BOND angle is larger `109^(@)28'` |
|
| 26. |
PCl_(5) in solid state exists as PCl_(4)^(+) and PCl_(6)^(-) and also in some solvents it undergoes dissociation as 2PCl_(5) hArr PCl_(4)^(+) +PCl_(6)^(-) The geometry and hybridisation of PCl_(6)^(-) is |
|
Answer» Octahedral , `sp^3` 
|
|
| 27. |
PCl_(5) in solid state exists as PCl_(4)^(+) and PCl_(6)^(-) and also in some solvents it undergoes dissociation as 2PCl_(5) hArr PCl_(4)^(+) +PCl_(6)^(-) The geometry and hydridisation of PCl_(4)^(+) is |
|
Answer» Tetrahderal , `sp^3` 
|
|
| 28. |
PCl_(5) exists but NCl_(5) does not due to |
|
Answer» INERTNESS of `N_(2)` |
|
| 29. |
PCl_5 exists but NCl_3 does not because: |
|
Answer» NITROGEN has no VACANT .d. ORBITALS |
|
| 30. |
PCl_(5) exist but NCl_(5) doesnot because :- |
|
Answer» NITROGEN ATOM is much smaller than P |
|
| 32. |
PCl_(5) causes cleavage of ether linkage R-O-R' forming RCl, R'Cl and POCl_(3), C_(5)H_(12)O on reaction with PCl_(5) forms 2 - chloropropane and 1 - chloroethane as main compound. Thus, C_(5)H_(12)O is named as |
|
Answer» 1 - ethoxypropane |
|
| 33. |
PCl_5 acts as an oxidising agent. Justify . |
|
Answer» Solution : `PCI_5` acts as an oxidising agent because its oxidation NUMBER is DECREASED in the reaction as under : `2AG + PCl_5 to 2Ag CL + PCl_3` |
|
| 34. |
PCl_3 under goes hydrolysis to produce an oxoacid.It has formula In solid PCl, exist as |
|
Answer» `HPO_(3)` |
|
| 35. |
PCl_3 reacts with organic compounds containing_____group. |
|
Answer» `-SO_3` |
|
| 36. |
PCl_(3) on reacting with HCOOH gives |
|
Answer» Chlorobenzene |
|
| 37. |
PCl_3 dissociates into PCl_3 and Cl_2 . If the total pressure of the system in equilibrium is p at a density, d, and temperature, T, show thatalpha = (pM)/(dRT) - 1where alphais the degree of dissociation, M is the relative molar mass of PCl_3. If the vapour density has the value 62 when the temperature is 230^@C, what is the value of p/d? |
| Answer» SOLUTION :`0.333L ATM G^(-1)` | |
| 38. |
PCI_(3) on hydrolysis gives |
|
Answer» `H_(3)PO_(2)` |
|
| 39. |
PCl_3 fumes in moisture because |
|
Answer» it UNDERGOES HYDROLYSIS giving FUMES of HCl |
|
| 40. |
PCI_5(g) at an initial pressure of 1 atm is placed in a closed vessel containing chlorine at a pressure of 0.2 atm. if PCI_5 dissociation to the extent of 20% then match the following : {:("","List-I","List_II",""),((P),"Partial pressure of" PCI_5,(1),0.4 atm),((Q),"Partial pressure of" PCI_3,(2),0.8 atm),((R ),"Partial pressure of"CI_2,(3),0.10 atm),((S),"Equilibrium constant" ("In terms of partial pressures" ) "for dissociation of "PCI_5,(4),0.2 atm):} |
|
Answer» <P>`{:(P,Q,R,S),(1,2,4,3):}` |
|
| 41. |
PCI_5 reacts with propanone, to give |
|
Answer» VIE dichloride |
|
| 42. |
PCI_(5) is less stable. Why? |
|
Answer» SOLUTION :`PCI_(5)` molecule has trigonal bipyramidal structure. The three EQUITORIAL bonds are equivalent. The `P-Cl` bond length in equitorial POSITION is 202 pm. The twoaxial bonds (240 pm) are longer. The axial bonds suffer more repulsion as compared to theequatorial bonds. HENCE, `PCI_(5)` is less stable. |
|
| 43. |
PCI_(5) is known but NCI_(5) is not known. Or Nitrogen does not form pentahalide. |
Answer» Solution :ELECTRONIC configuration of P is `1 s^(2)2 s^(2) 2 s^(6) 3 s^(2)" "3 P_(x)^(1) 3P_(y)^(1)3P_(z)^(1)3d^(0)`. Thus, P has emptly 3d orbitals to  which the 3S ELECTRON can be excited to have five half-filled orbitals needed for formation of `PCI_(5)`. Thus, `PCl_(5)`, is known. In contrast, electronic configuration of N is `1s^(2)2s^(2)2P_(x)^(1)2P_(y)^(1)2P_(z)^(1)`. Since the valence shell of nitrogen has n = 2, therefore, it cannot have d-orbitals. However, if one of the 2s electrons is excited to 3s orbital, five  half-filled orbitals needed to form `NCI_(5)`can still be obtained. But such an excitation is thermodynamically not favourable since the energy needed for excitation is more than the energy expected to be generated during the formation of TWO additional P-CI bonds. Therefore, nitrogen doesnot form `NCI_(5)`. In other words, `NCI_(5)` is UNKNOWN. |
|
| 44. |
PCI_(5) is ionic in nature in the solid state. Give reasons. Or Solid phosphorus pentachloride behaves as an ionic compound. Explain. |
|
Answer» SOLUTION :`PCI_(5)` conducts electricity in the MOLTEN state. This MEANS that in the SOLID state, it exists as `[PCI_(4)]^(+) [PCI_(6)]^(-)` in which the cation is tetrahedral and the anion is octahedral `2 PCI_(5) rarr [PCI_(4)]^(+) [PCI_(6)]^(-)` On MELTING, these ions become free to move and hence `PCI_(5)` conducts electricity in the molten state. |
|
| 45. |
PCl_(5)+Cl^(-) to PCl_(6)^(-). The wrong statement regarding the above equation is |
|
Answer» Hybridisation of P CHANGES from `sp^3d` to `sp^3d^2` |
|
| 47. |
PCl_(3) is covalent. It fumes in moisture and its aqueous solution is electrical conductor. Why? |
|
Answer» Solution :`PCl_(3)` hydrolyses in moisture (or in water) giving fumes of HYDROGEN CHLORIDE. `PCl_(3)+3H_(2)OrarrH_(3)PO_(3)+3HCl` In aqueous SOLUTIONS, HCI ionises and HENCE acts as ELECTRICAL conductor. |
|
| 48. |
PbSO_(4) is soluble in (a) _____ due to formation of (b) _____. |
|
Answer» |
|
| 49. |
PbS can be separated from ZnS by electrostatic separation method. The property utilized in this method is |
|
Answer» PBS is a GOOD CONDUCTOR and ZnS is a poor conductor of ELECTRICITY |
|