Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Observe the diagram. Which one of the following statement is correct? |
|
Answer» B can REDUCE `AO_(2)` |
|
| 2. |
Observe the compound 'M' If in this compound X=Total number of asymmetric C^(***) atoms Y= Number of similar asymmetric C^(***) atoms Z= Number of optically active stereoisomers W= Number of optically inactive isomers R= Number of geometrical orientations in space Report your answer as : X +Y +Z+ W+ R |
|
Answer» |
|
| 3. |
Ozone reacts with dry Iodine to form an oxide having ______ Oxygen atoms in its molecule. |
|
Answer» |
|
| 4. |
O_3reacts with CH_2 = CH_2to form ozonide. On hydrolysis it forms ... |
|
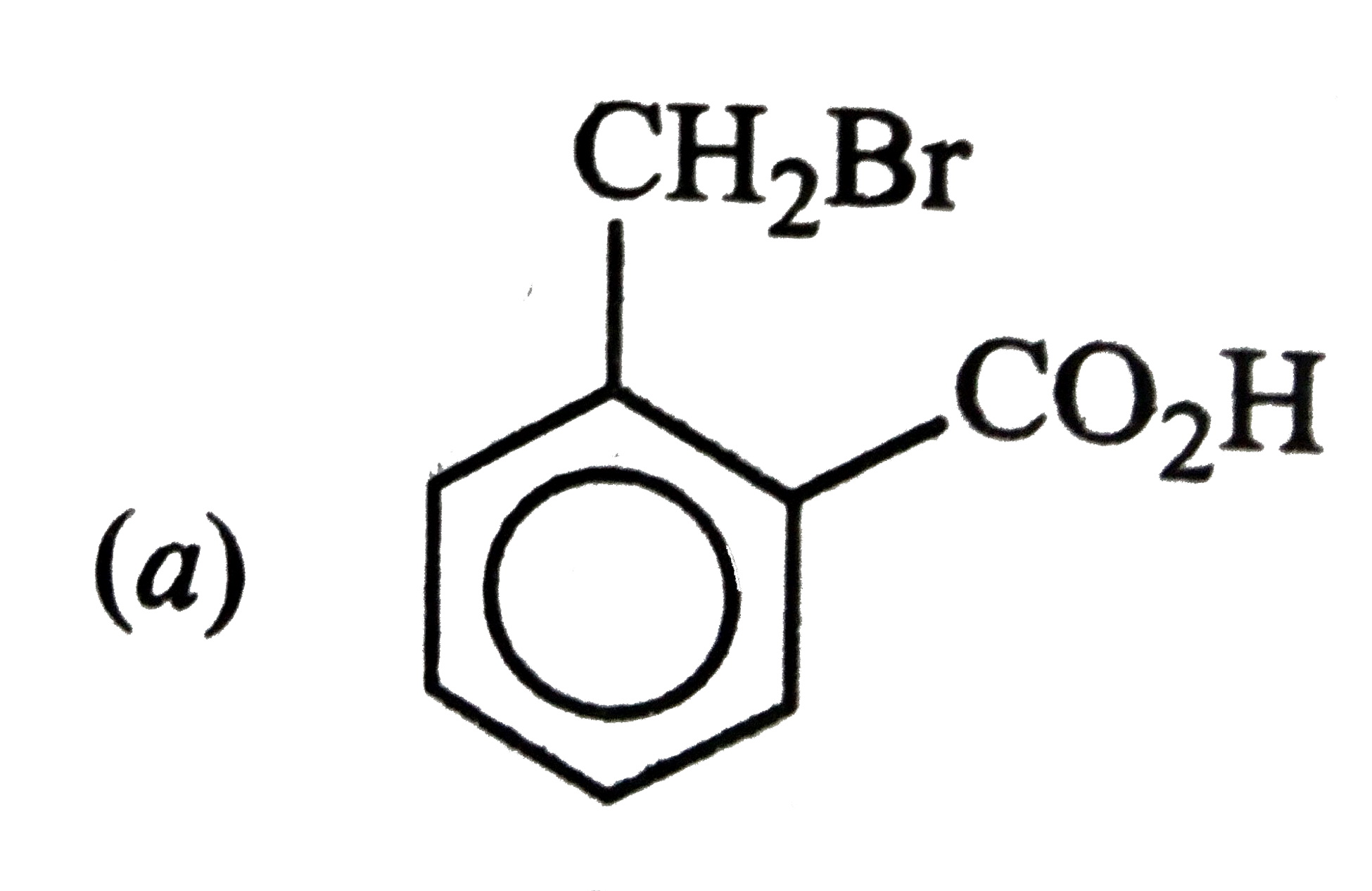
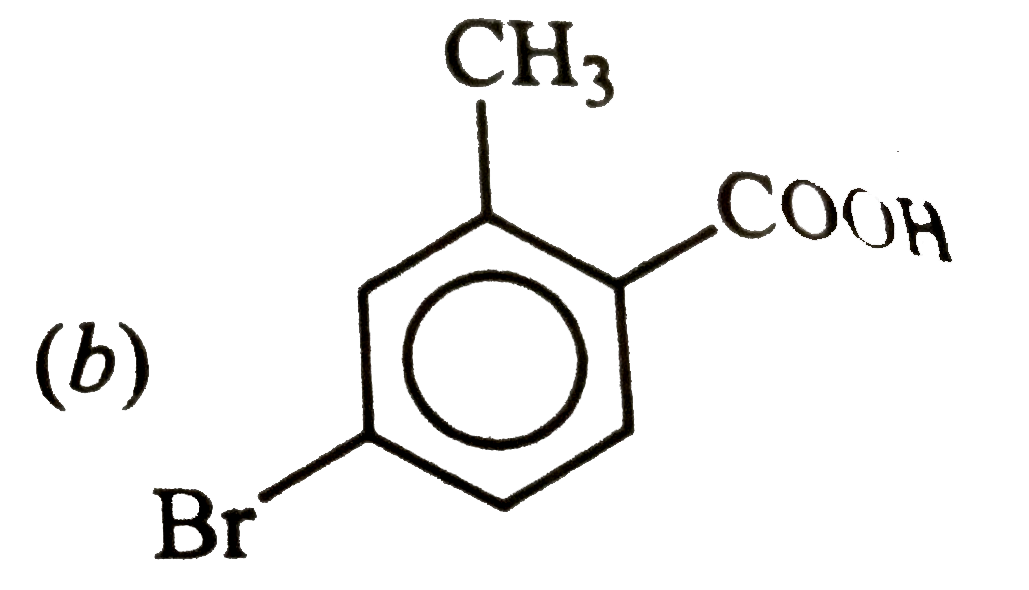
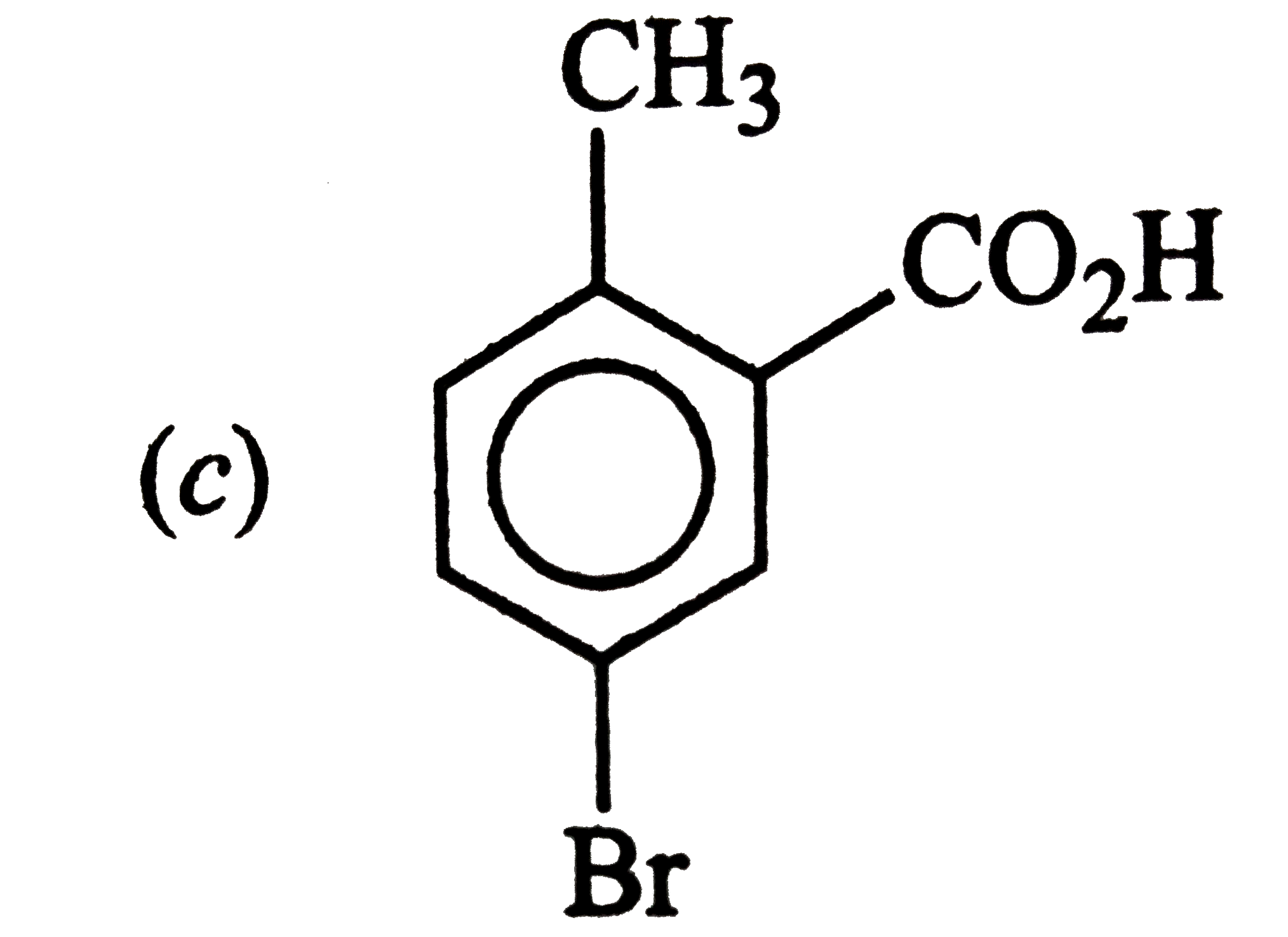
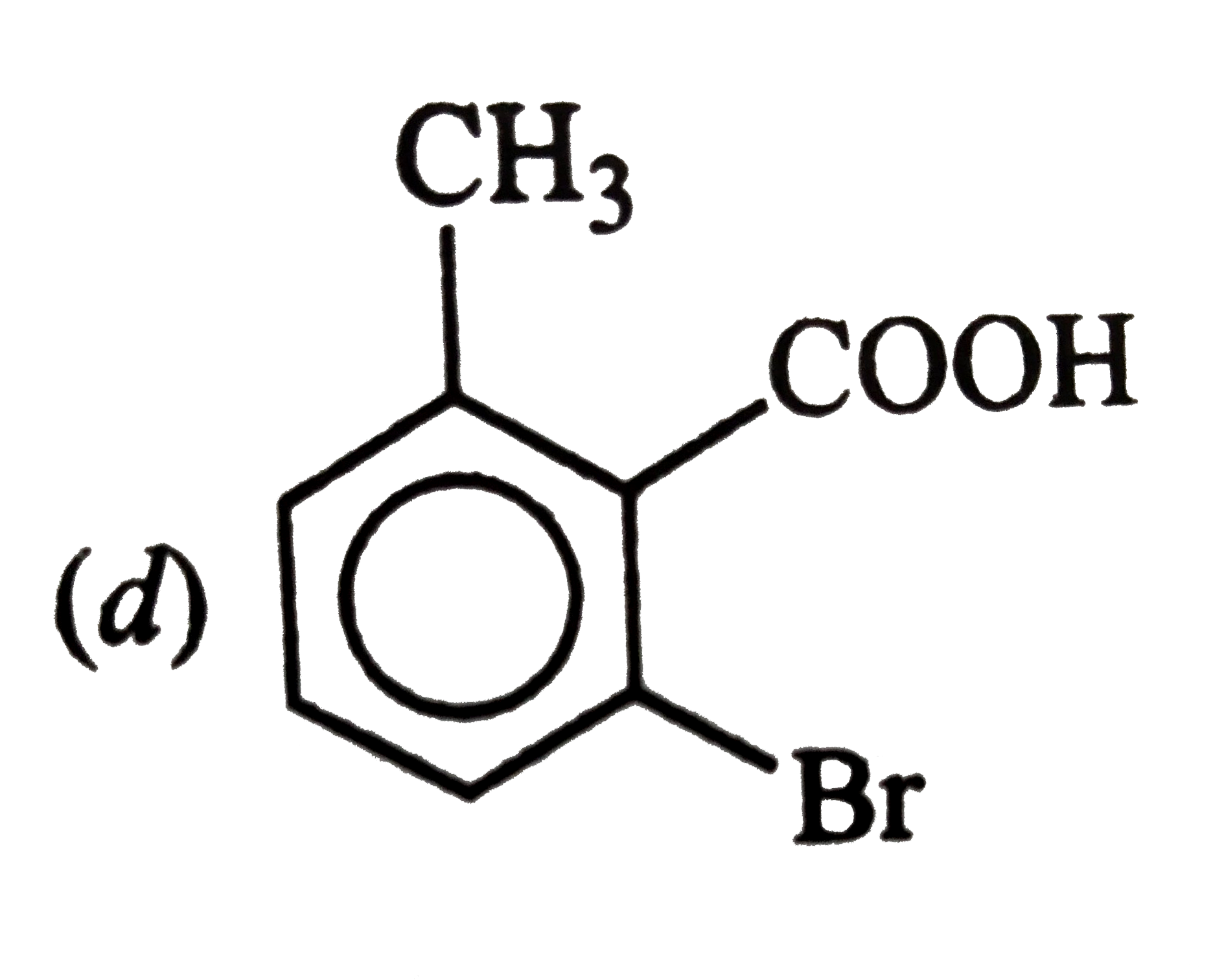
Answer» ETHYLENE oxide |
|
| 5. |
O_(3) reacts with CH_(2)=CH_(2) to form ozonide. On hydrolysis it forms __________. |
|
Answer» ETHYLENE OXIDE |
|
| 6. |
O_3 is soluble in |
|
Answer» WATER |
|
| 7. |
O_3 is oxidising agent |
| Answer» Answer :A | |
| 8. |
O_3 does not give ozonide with |
|
Answer» 2-Butene |
|
| 9. |
O_3 is made from O_2 : |
|
Answer» By OXIDATION at high temperature |
|
| 10. |
O_3 is absorbed by caustic soda.is it true or false ? |
| Answer» SOLUTION :TURPENTINE OIL | |
| 11. |
O_2^(2-) is isoelectronic with |
|
Answer» `H_2` |
|
| 12. |
O_2 required for complete oxidation of 1 litre of ethane at NTP is: |
|
Answer» 3.5 litre |
|
| 13. |
O_2 molecule is |
|
Answer» PARAMAGNETIC |
|
| 14. |
O_(2-) ions are arranged in ccp in spinel structure. A^(2+) ions occupy 1//8 of TV_(s) and B^(o+) ions occupy half of OV . The void volume of unit cell = 0.11 Å. Find the value of A ? |
|
Answer» VOID volume `= 0.22//"unit"` volume of unit CELL ` `0.11 Å = 0.22 rArr A = 2` |
|
| 15. |
O_2 is bubbled through water at 293K, assuming that O_2 exerts a partial pressure of 0.98 bar, the solubility of O_2 in gm.L^(-1) is (Henry's law constant = 34 k bar) |
|
Answer» 0.025 |
|
| 16. |
O_2 is denser than air and therefore it is collected in: |
|
Answer» SPIRIT |
|
| 17. |
O_2 is absorbed by lime water. Is it true or false? |
| Answer» SOLUTION :ALKALINE PYROGALLOL | |
| 18. |
"o-Xylene"overset(HNO_(3))(to)Xoverset("phenol")underset(H_(2)SO_(4))(to)Y. The product is |
|
Answer» PHTHALIC acid 
|
|
| 19. |
o- xylene on oxidation gives |
|
Answer» PHTHALIC ACID |
|
| 20. |
o-Toluic acid on reaction with Br_(2)+Fe gives |
|
Answer»
|
|
| 22. |
o-Phenylenediamine has a structure |
|
Answer»
 the divalent group `(C_(6)H_(4))` is CALLED as PHENYLENE group. |
|
| 23. |
o-nitrophenol is steam volatile due to ________ hydrogen bonding. |
| Answer» SOLUTION :INTRAMOLECULAR | |
| 24. |
o-nitrophenol is more volatile than p-nitrophenol it is due to |
|
Answer» Intermolecular HYDROGEN BONDING in o- nitrophenol |
|
| 25. |
o-nitrophenol can be easily stream distilled whereas p-nitrophenol cannot be. This is because of : |
|
Answer» Strong intermolecular hydrogen BONDING in o-nitrophenol |
|
| 26. |
O-nitro phenol is slightly soluble in water where as P-nitro phenol is more soluble. Give reason. |
| Answer» Solution :O-nitro phenol is slightly SOLUBLE in WATER and more volatile due to intra MOLECULAR hydrogen bonding, WHEREAS P-nitro phenol is more soluble in water and less volatile due to INTERMOLECULAR hydrogen bonding. | |
| 27. |
o-Methoxybromobenzene is treated with sodamide and then with ammonia. The product formed is |
|
Answer» o-Methoxyaniline 
|
|
| 28. |
o-nitro phenol has lower boiling point than p-nitro phenol. Why? |
| Answer» Solution :p-nitro PHENOL SHOWS inter MOLECULAR H bonding. So it has high BOILING point. | |
| 29. |
o-Chlorotoluene reacts with sodamide in liquid NH_(3) to give o-toluidine and m-toluidine. Which of the following is an intermediate in the above reaction? |
|
Answer»
|
|
| 30. |
O+H_(2)NOH toA overset(H^(+))toB THe compound ‘B' is |
|
Answer»
|
|
| 31. |
o- fluorotoluene reacts with sodium amide to form- |
|
Answer» <P>only o-TOLUIDINE |
|
| 32. |
o- and p-Nitrophenols can be separated by_____. |
| Answer» SOLUTION :STEAM DISTILLATION | |
| 33. |
O_(3) is prepared by subjecting 0, to silent electric discharge. The favourable conditions for the formation of ozone according to Le-chatlier's principle are |
|
Answer» LOW TEMPERATURE, low pressure |
|
| 34. |
O^(-2) or S ^(-2) formation is endothermic because |
|
Answer» More STABILITY of `O^(-2) or S ^(-2)` |
|
| 35. |
O - 18 isotope of oxygen will have |
|
Answer» 18 PROTONS |
|
| 36. |
O |
|
Answer» Which of the following CATALYST is used in preparation of HIGH density polythene ? |
|
| 37. |
Nylons polyesters and cotton all possess strength due to : |
|
Answer» INTERMOLECULAR H-bonding |
|
| 38. |
Nylon threads are made of |
|
Answer» POLYETHYLENE POLYMER |
|
| 39. |
Nylons, polyesters and cotton all possess strength due to : |
|
Answer» INTERMOLECULAR H-bonding |
|
| 40. |
Nylon salt is known as |
|
Answer» tetramethylene diammonium ADIPATE |
|
| 41. |
Nylon thread contains the polymer |
|
Answer» polyamide |
|
| 43. |
Nylon threads are made of : |
|
Answer» Polythylene POLYMER |
|
| 45. |
Nylon is of which type fibre ? |
| Answer» Solution :Polyamide | |
| 46. |
Nylon is not a |
|
Answer» Condensation polymer |
|
| 47. |
Nylon is generic name for all synthetic fibre forming |
|
Answer» polysters |
|
| 48. |
Nylon is classified as a |
|
Answer» POLYOLEFIN |
|
| 49. |
Nylon 66, polyethylene, PVC, teflon. |
| Answer» SOLUTION :Nylon 66. It is a CONDENSATION polymer whereas others are ADDITION POLYMERS. | |
| 50. |
Nylon is anexample of |
|
Answer» polyamide |
|